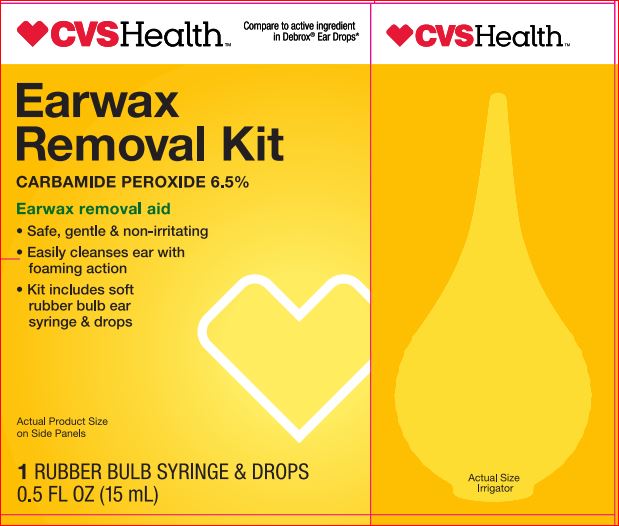
Label: EARWAX REMOVAL KIT- carbamide peroxide 6.5% kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 69842-039-01, 69842-224-07 - Packager: CVS
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 2, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient Purpose
- PURPOSE
- Uses
-
Warnings
Ask doctor before use if you have • ear drainage or discharge • ear pain • irritation or rash in ear • dizziness • an injury or perforation (hole) of the eardrum • recently had ear surgeryStop and ask doctor before use if • you need to use for more than four days • excessive earwax remains after use of this product
-
DOSAGE & ADMINISTRATION
DirectionsFor use in the ear only.
Adults and children over 12 years of age:
• tilt head sideways and place 5 to 10 drops into ear
• tip of applicator should not enter ear canal
• keep drops in ear for several minutes by keeping head tilted or placing cotton in the ear
• use twice daily for up to 4 days if needed, or as directed by a doctor
• any wax remaining after treatment may be removed by gently flushing the ear with warm water, using a soft rubber bulb ear syringe
Children under 12 years of age: consult a doctor. - INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EARWAX REMOVAL KIT
carbamide peroxide 6.5% kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69842-224 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-224-07 1 in 1 KIT 12/24/2014 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 15 mL Part 1 of 1 EAR WAX RMVL CVS
carbamide peroxide 6.5% liquidProduct Information Item Code (Source) NDC:69842-039 Route of Administration AURICULAR (OTIC) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Carbamide Peroxide (UNII: 31PZ2VAU81) (HYDROGEN PEROXIDE - UNII:BBX060AN9V) Carbamide Peroxide 6.5 g in 100 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) Glycerin (UNII: PDC6A3C0OX) Propylene Glycol (UNII: 6DC9Q167V3) Sodium Lauroyl Sarcosinate (UNII: 632GS99618) Sodium Stannate (UNII: NJ7C1V83KG) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69842-039-01 1 in 1 CARTON 1 15 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part344 12/24/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part344 12/24/2014 Labeler - CVS (062312574) Registrant - Product Quest Mfg. (927768135) Establishment Name Address ID/FEI Business Operations Product Quest Mfg. 927768135 manufacture(69842-039) , label(69842-039) , pack(69842-224)