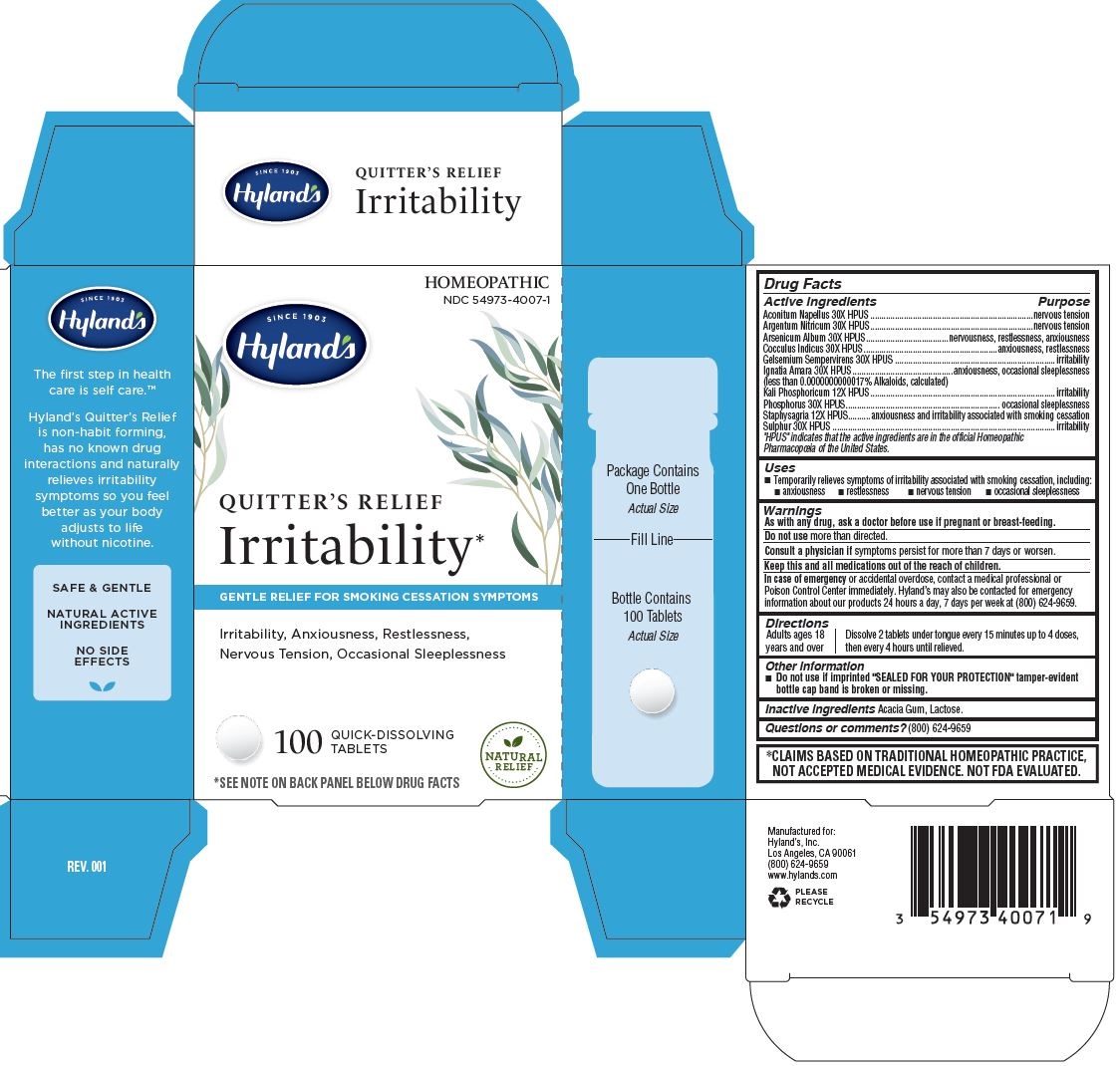
Label: QUITTERS RELIEF IRRITABILITY- aconitum napellus whole, anamirta cocculus seed, arsenic trioxide, delphinium staphisagria seed, dibasic potassium phosphate, gelsemium sempervirens root, phosphorus, silver nitrate and sulfur tablet
- NDC Code(s): 54973-4007-1
- Packager: Hyland's Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 13, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- PURPOSE
-
Drug Facts
Active ingredients
Active ingredients
Purpose
Aconitum Napellus 30X HPUS
nervous tension
Argentum Nitricum 30X HPUS
nervous tension
Arsenicum Album 30X HPUS
nervousness, restlessness, anxiousness
Cocculus Indicus 30X HPUS
anxiousness, restlessness
Gelsemium Sempervirens 30X HPUS
irritability
Ignatia Amara 30X HPUS
(less than 0.0000000000017% Alkaloids, calculated)
anxiousness, occasional sleeplessness
Kali Phosphoricum 12X HPUS
irritability
Phosphorus 30X HPUS
occasional sleeplessness
Staphysagria 12X HPUS
anxiousness and irritability associated with smoking cessation
Sulphur 30X HPUS
irritability
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- PACKAGE LABEL - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
QUITTERS RELIEF IRRITABILITY
aconitum napellus whole, anamirta cocculus seed, arsenic trioxide, delphinium staphisagria seed, dibasic potassium phosphate, gelsemium sempervirens root, phosphorus, silver nitrate and sulfur tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54973-4007 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SILVER NITRATE (UNII: 95IT3W8JZE) (SILVER CATION - UNII:57N7B0K90A) SILVER NITRATE 30 [hp_X] SULFUR (UNII: 70FD1KFU70) (SULFUR - UNII:70FD1KFU70) SULFUR 30 [hp_X] ACONITUM NAPELLUS WHOLE (UNII: U0NQ8555JD) (ACONITUM NAPELLUS WHOLE - UNII:U0NQ8555JD) ACONITUM NAPELLUS WHOLE 30 [hp_X] ARSENIC TRIOXIDE (UNII: S7V92P67HO) (ARSENIC CATION (3+) - UNII:C96613F5AV) ARSENIC TRIOXIDE 30 [hp_X] ANAMIRTA COCCULUS SEED (UNII: 810258W28U) (ANAMIRTA COCCULUS SEED - UNII:810258W28U) ANAMIRTA COCCULUS SEED 30 [hp_X] DIBASIC POTASSIUM PHOSPHATE (UNII: CI71S98N1Z) (PHOSPHATE ION - UNII:NK08V8K8HR) DIBASIC POTASSIUM PHOSPHATE 12 [hp_X] GELSEMIUM SEMPERVIRENS ROOT (UNII: 639KR60Q1Q) (GELSEMIUM SEMPERVIRENS ROOT - UNII:639KR60Q1Q) GELSEMIUM SEMPERVIRENS ROOT 30 [hp_X] STRYCHNOS IGNATII SEED (UNII: 1NM3M2487K) (STRYCHNOS IGNATII SEED - UNII:1NM3M2487K) STRYCHNOS IGNATII SEED 30 [hp_X] DELPHINIUM STAPHISAGRIA SEED (UNII: 00543AP1JV) (DELPHINIUM STAPHISAGRIA SEED - UNII:00543AP1JV) DELPHINIUM STAPHISAGRIA SEED 12 [hp_X] PHOSPHORUS (UNII: 27YLU75U4W) (PHOSPHORUS - UNII:27YLU75U4W) PHOSPHORUS 30 [hp_X] Inactive Ingredients Ingredient Name Strength LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) ACACIA (UNII: 5C5403N26O) Product Characteristics Color white (Off White) Score no score Shape ROUND Size 9mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54973-4007-1 1 in 1 CARTON 12/15/2020 1 100 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 12/15/2020 Labeler - Hyland's Inc. (008316655)