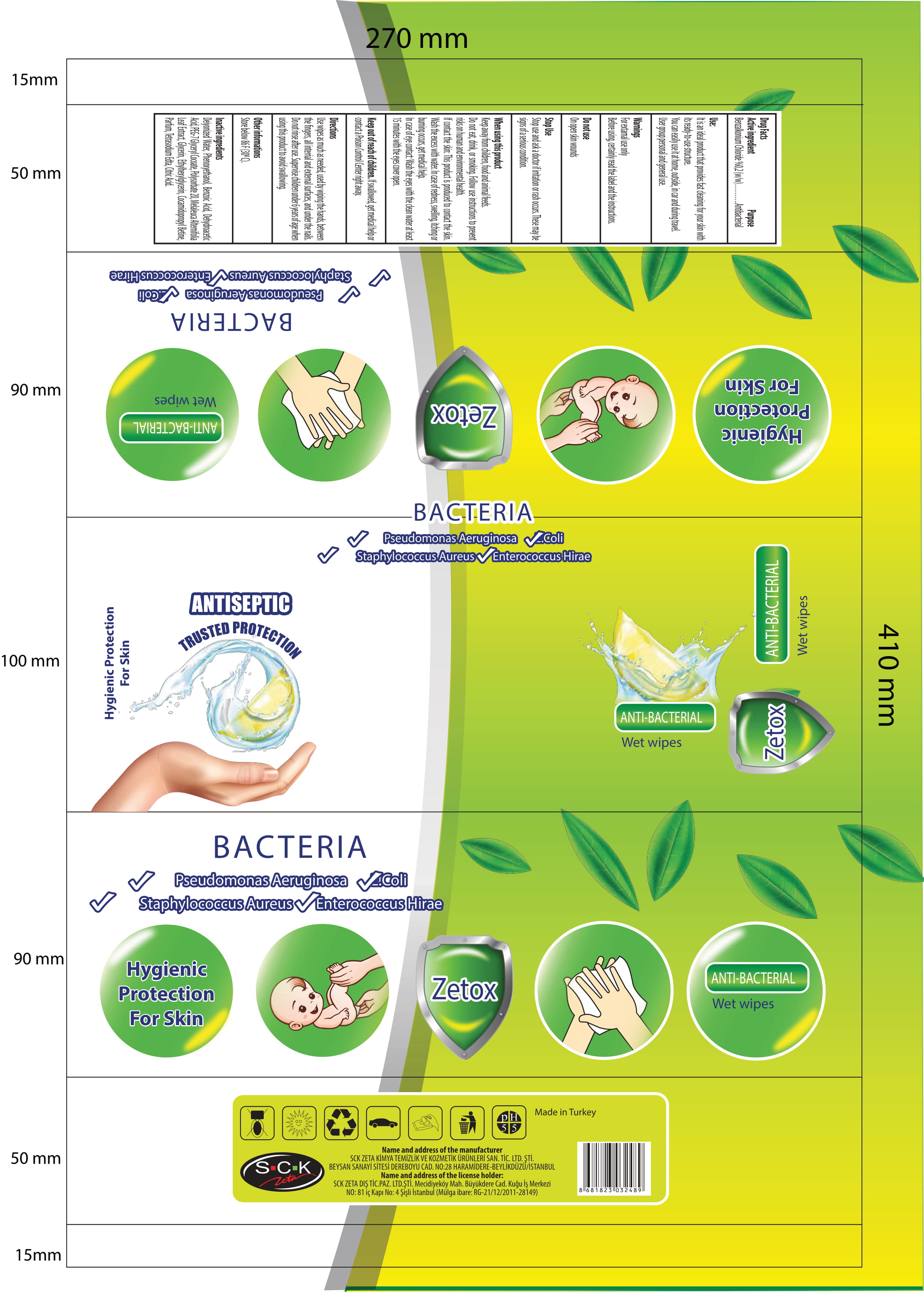
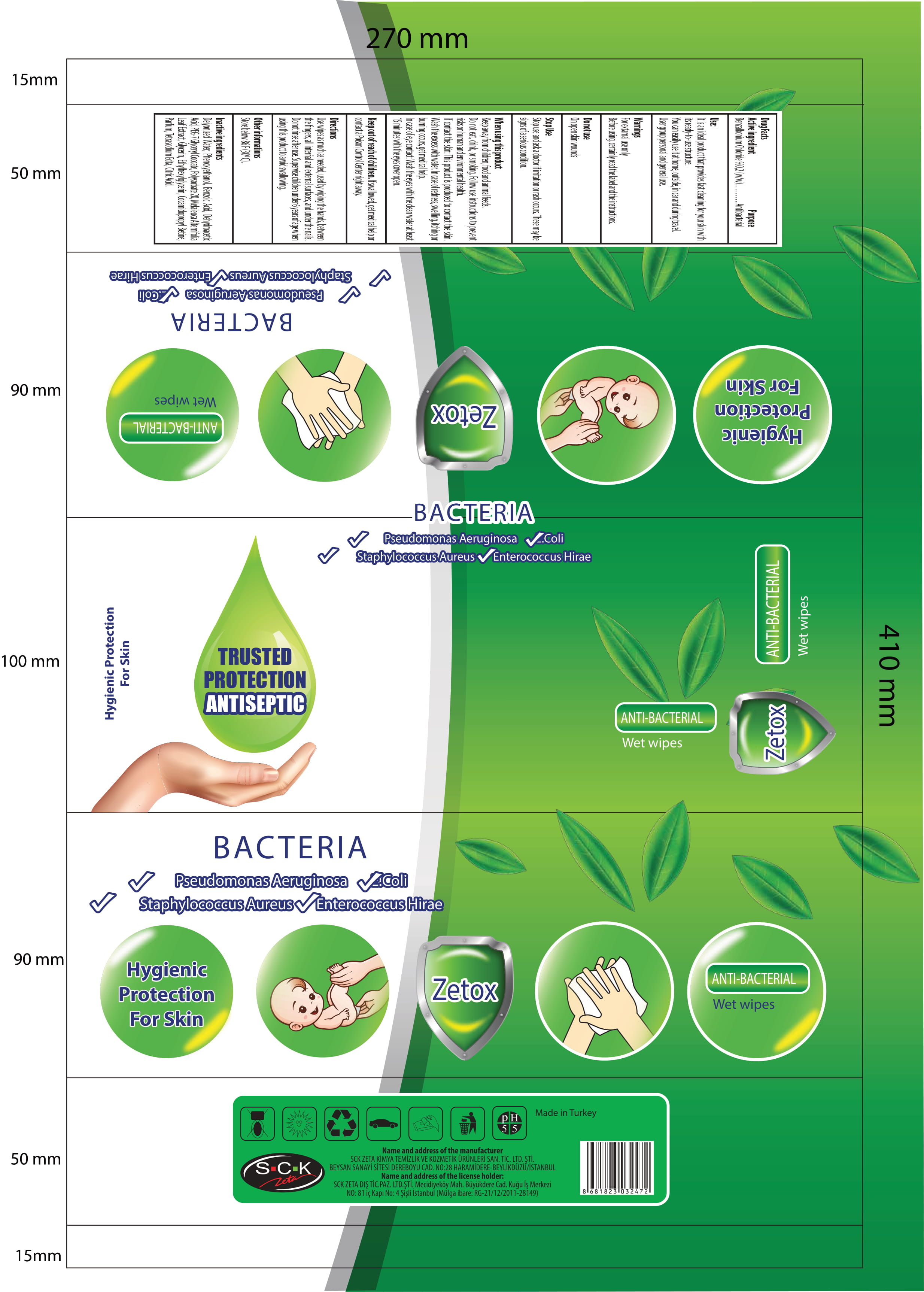
Label: ZETOX ANTIBACTERIAL WET WIPES- benzalkonium chloride cloth
-
Contains inactivated NDC Code(s)
NDC Code(s): 86818-004-01, 86818-004-02, 86818-004-03, 86818-004-04, view more86818-004-05, 86818-004-06 - Packager: SCK ZETA DIS TICARET
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 23, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
SPL UNCLASSIFIED SECTION
This is a hand sanitizer manufactured according to the Temporary Policy for Preparation of Certain Hand Sanitizer Products During the Public Health Emergency (CoViD-19); Guidance for Industry.
The hand sanitizer is manufactured using only the following United States Pharmacopoeia (USP) grade ingredients in the preparation of the product (percentage in final product formulation) consistent with World Health Organization (WHO) recommendations:
- Benzalkonium Chloride 0.2 % ( w/w )
- Glycerin
- Phenoxyethanol
- Water
- Benzoic Acid
- Etidronate Tetrasodium
- Peg-7 Glyceryl Cocoate
- Polysorbate 20
- Melaleuca Alternifolia Leaf
- Ethylhexylglycerin
- Cocamidopropyl Betaine
- Anhydrous Citric Acid
- Dehydroacetic Acid The firm does not add other active or inactive ingredients. Different or additional ingredients may impact the quality and potency of the product.
- Active Ingredient(s)
- Purpose
- Use
- Warnings
- Do not use
-
WHEN USING
When using this product Keep out of reach of children, food and animal feeds. Do not eat, dirnk and smoking. Follow use instructions to prevent risks on human and environmental health.If contact the skin: This product is produced to contact the skin. Wash the excess with water. In case of redness, swelling, itching or burning occurs, get medical help.In case of eye contact: wash the eyes with the clean water at least 15 minutes with the eyes cover open. If swallowed shake the mouth with water. Do not induce vomiting. Immediately apply to the doctor and show him the label of the product.
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ZETOX ANTIBACTERIAL WET WIPES
benzalkonium chloride clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:86818-004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.2 g in 100 g Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) GLYCERIN (UNII: PDC6A3C0OX) PHENOXYETHANOL (UNII: HIE492ZZ3T) WATER (UNII: 059QF0KO0R) BENZOIC ACID (UNII: 8SKN0B0MIM) DEHYDROACETIC ACID (UNII: 2KAG279R6R) PEG-7 GLYCERYL COCOATE (UNII: VNX7251543) POLYSORBATE 20 (UNII: 7T1F30V5YH) MELALEUCA ALTERNIFOLIA LEAF (UNII: G43C57162K) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) ETIDRONATE TETRASODIUM (UNII: CZZ9T1T1X4) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:86818-004-02 120 in 1 PACKAGE 03/30/2020 1 NDC:86818-004-01 0.74 g in 1 PATCH; Type 0: Not a Combination Product 2 NDC:86818-004-04 100 in 1 PACKAGE 03/30/2020 2 NDC:86818-004-03 0.74 g in 1 PATCH; Type 0: Not a Combination Product 3 NDC:86818-004-06 80 in 1 PACKAGE 03/30/2020 3 NDC:86818-004-05 0.74 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 03/30/2020 Labeler - SCK ZETA DIS TICARET (356290986) Registrant - SCK ZETA DIS TICARET (356290986) Establishment Name Address ID/FEI Business Operations SCK ZETA DIS TICARET 356290986 manufacture(86818-004)