Label: CETIRIZINE HYDROCHLORIDE tablet
-
Contains inactivated NDC Code(s)
NDC Code(s): 67296-1085-3 - Packager: RedPharm Drug
- This is a repackaged label.
- Source NDC Code(s): 16571-402
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated January 20, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
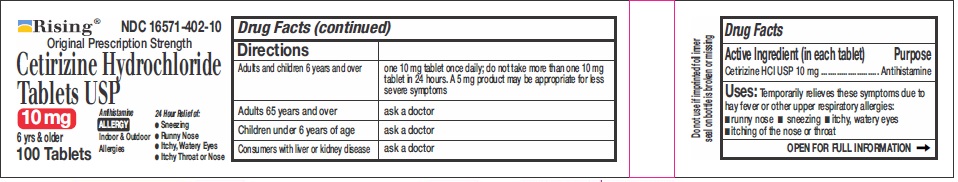
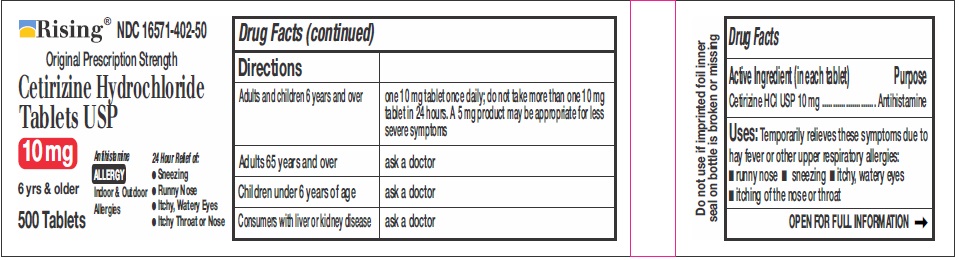
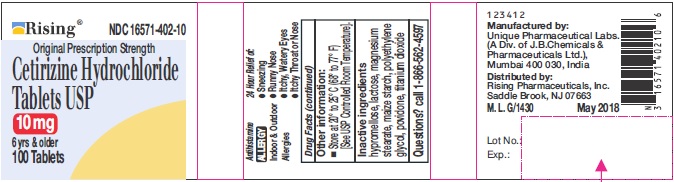
- ACTIVE INGREDIENTS
- PURPOSE
- USES
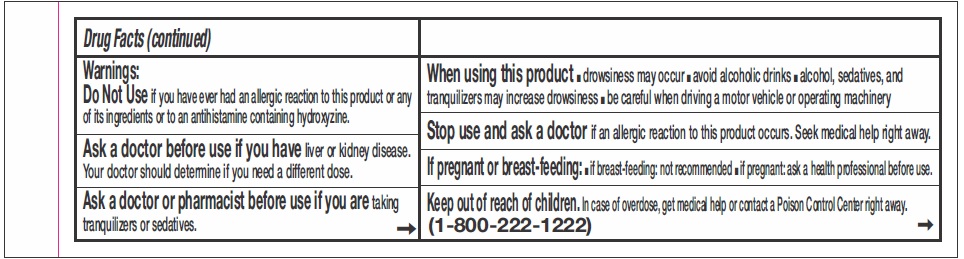
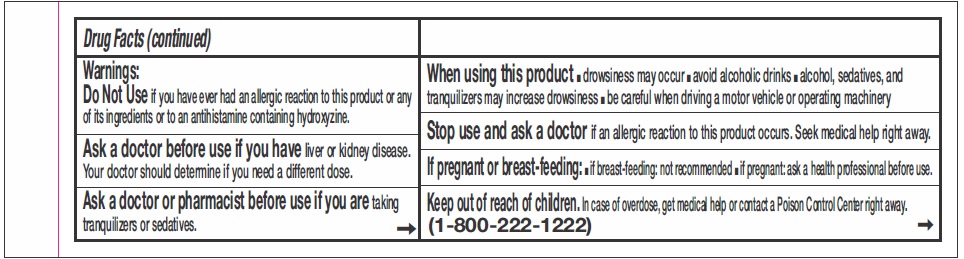
- WARNINGS
- ASK DOCTOR
- ASK DOCTOR/PHARMACIST
- WHEN USING THIS PRODUCT
- STOP USE
- IF PREGNANT OR BREAST FEEDING:
- KEEP OUT OF REACH OF CHILDREN
-
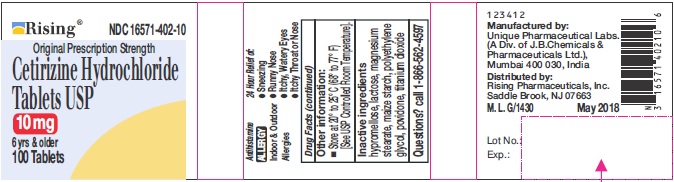
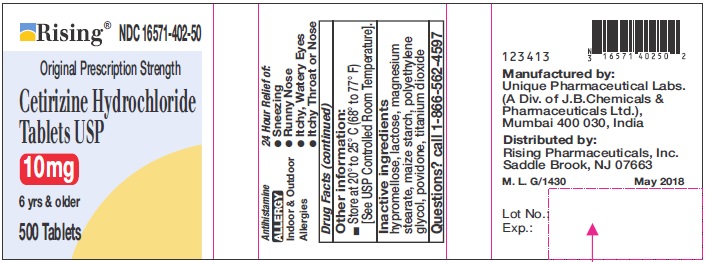
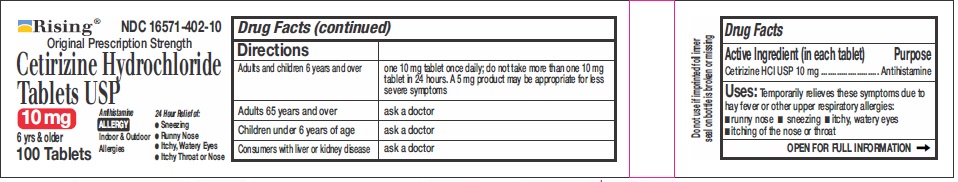
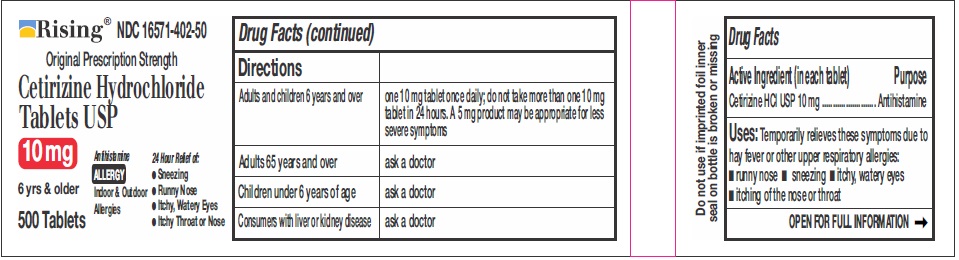
DIRECTIONS
Adults and children 6
years and overone 10 mg tablet once daily, do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms. Adults 65 years and over Ask a doctor Children under 6 years of age Ask a doctor Consumers with liver or kidney disease Ask a doctor - OTHER INFORMATION
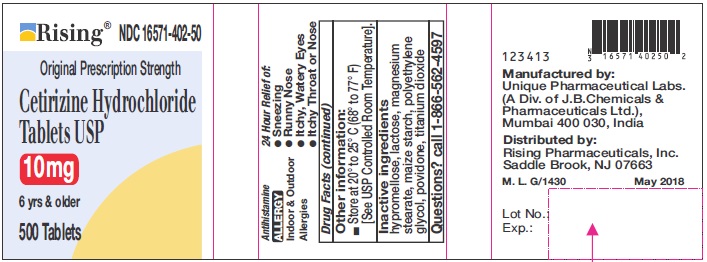
- INACTIVE INGREDIENTS
- QUESTIONS?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL-100'S COUNT
- PRINCIPAL DISPLAY PANEL-500'S COUNT
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CETIRIZINE HYDROCHLORIDE
cetirizine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67296-1085(NDC:16571-402) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CETIRIZINE HYDROCHLORIDE (UNII: 64O047KTOA) (CETIRIZINE - UNII:YO7261ME24) CETIRIZINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSES (UNII: 3NXW29V3WO) LACTOSE (UNII: J2B2A4N98G) MAGNESIUM STEARATE (UNII: 70097M6I30) STARCH, CORN (UNII: O8232NY3SJ) POLYETHYLENE GLYCOL (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white (White) Score no score Shape BULLET (Barrel Shaped) Size 8mm Flavor Imprint Code CTN;10 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67296-1085-3 30 in 1 BOTTLE; Type 0: Not a Combination Product 09/21/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077829 09/21/2020 Labeler - RedPharm Drug (828374897) Establishment Name Address ID/FEI Business Operations EPM Packaging 079124340 repack(67296-1085)