Label: COLDCALM- onion, apis mellifera, atropa belladonna, eupatorium perfoliatum flowering top, gelsemium sempervirens root, strychnos nux-vomica seed, phytolacca americana root, pulsatilla vulgaris, dichromate ion tablet
- NDC Code(s): 0220-9048-04, 0220-9048-29, 0220-9048-91
- Packager: Laboratoires Boiron
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated March 23, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
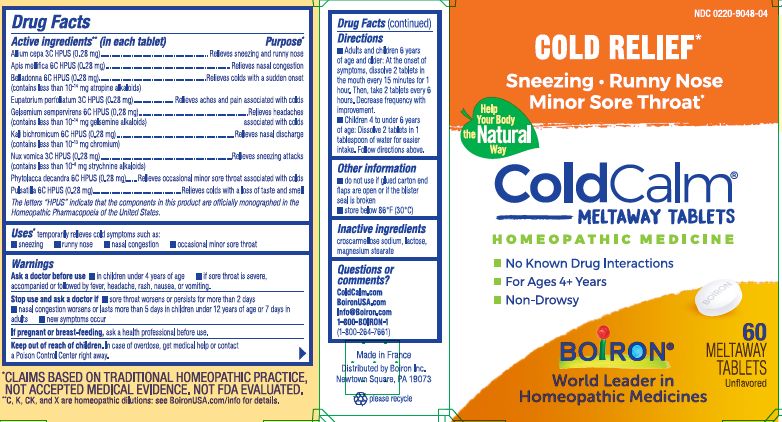
ACTIVE INGREDIENT
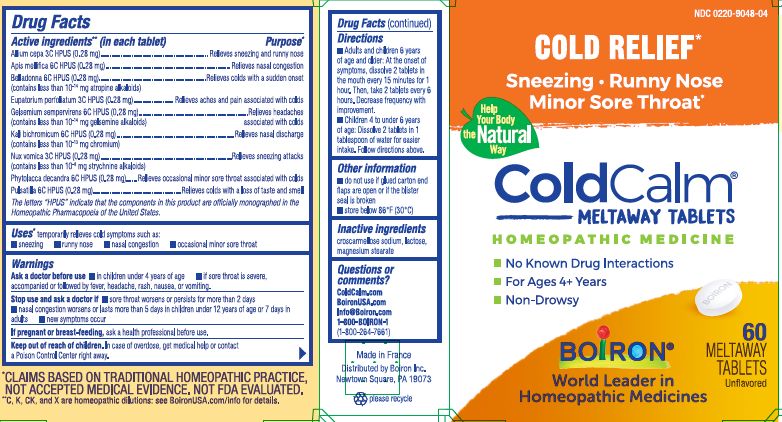
Active ingredients** (in each tablet)
Allium cepa 3C HPUS (0.28 mg)
Apis mellifica 6C HPUS (0.28 mg)
Belladonna 6C HPUS (0.28 mg) (contains less than 10 -14 mg atropine alkaloids)
Eupatorium perfoliatum 3C HPUS (0.28 mg)
Gelsemium sempervirens 6C HPUS (0.28 mg) (contains less than 10 -14 gelsemine alkaloids)
Kali bichromicum 6C HPUS (0.28 mg) (contains less than 10 -13 mg chromium)
Nux vomica 3C HPUS (0.28 mg) (contains less than 10 -8 mg strychnine alkaloids)
Phytolacca decandra 6C HPUS (0.28 mg)
Pulsatilla 6C HPUS (0.28 mg)
The letters "HPUS" indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United States.
-
PURPOSE
Purpose*
Allium cepa 3C HPUS ... Relieves sneezing and runny nose
Apis mellifica 6C HPUS ... Relieves nasal congestion
Belladonna 6C HPUS ... Relieves colds with a sudden onset
Eupatorium perfoliatum 3C HPUS ... Relieves aches and pain associated with colds
Gelsemium sempervirens 6C HPUS ... Relieves headaches associated with colds
Kali bichromicum 6C HPUS ... Relieves nasal discharge
Nux vomica 3C HPUS .... Relieves sneezing attacks
Phytolacca decandra 6C HPUS ... Relieves occasional minor sore throat associated with colds
Pulsatilla 6C HPUS ... Relieves colds with a loss of taste and smell
- INDICATIONS & USAGE
- WARNINGS
- ASK DOCTOR
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
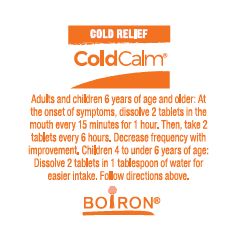
Directions
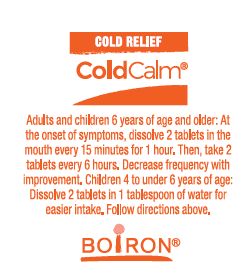
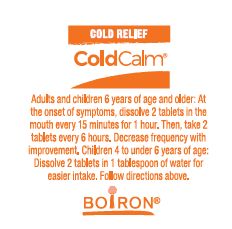
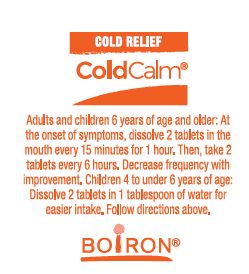
- Adults and children 6 years of age and older: At the onset of symptoms, dissolve 2 tablets in mouth every 15 minutes for 1 hour. Then, take 2 tablets every 6 hours. Decrease frequency with improvement.
- Children 4 to under 6 years of age: Dissolve 2 tablets in 1 tablespoon of water for easier intake. Follow directions above.
-
SPL UNCLASSIFIED SECTION
- do not use if glued carton end flaps are open or if the blister seal is broken
- store below 86° F (30° C)
Contains 20 meltaway tablets unflavored
Contains 60 meltaway tablets unflavored
Contains 120 meltaway tablets unflavored
No Known Drug Interactions
For Ages 4+ Years
Non-Drowsy
Cold Relief*
Sneezing, Runny Nose, Minor Sore Throat*
*CLAIMS BASED ON TRADITIONAL HOMEOPATHIC PRACTICE, NOT ACCEPTED MEDICAL EVIDENCE. NOT FDA EVALUATED.
*C,K,CK, and X are homeopathic dilutions: see BoironUSA.com/info for details. - INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
COLDCALM
onion, apis mellifera, atropa belladonna, eupatorium perfoliatum flowering top, gelsemium sempervirens root, strychnos nux-vomica seed, phytolacca americana root, pulsatilla vulgaris, dichromate ion tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0220-9048 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ONION (UNII: 492225Q21H) (ONION - UNII:492225Q21H) ONION 3 [hp_C] APIS MELLIFERA (UNII: 7S82P3R43Z) (APIS MELLIFERA - UNII:7S82P3R43Z) APIS MELLIFERA 6 [hp_C] ATROPA BELLADONNA (UNII: WQZ3G9PF0H) (ATROPA BELLADONNA - UNII:WQZ3G9PF0H) ATROPA BELLADONNA 6 [hp_C] EUPATORIUM PERFOLIATUM FLOWERING TOP (UNII: 1W0775VX6E) (EUPATORIUM PERFOLIATUM FLOWERING TOP - UNII:1W0775VX6E) EUPATORIUM PERFOLIATUM FLOWERING TOP 3 [hp_C] GELSEMIUM SEMPERVIRENS ROOT (UNII: 639KR60Q1Q) (GELSEMIUM SEMPERVIRENS ROOT - UNII:639KR60Q1Q) GELSEMIUM SEMPERVIRENS ROOT 6 [hp_C] DICHROMATE ION (UNII: 9LKY4BFN2V) (DICHROMATE ION - UNII:9LKY4BFN2V) DICHROMATE ION 6 [hp_C] STRYCHNOS NUX-VOMICA SEED (UNII: 269XH13919) (STRYCHNOS NUX-VOMICA SEED - UNII:269XH13919) STRYCHNOS NUX-VOMICA SEED 3 [hp_C] PHYTOLACCA AMERICANA ROOT (UNII: 11E6VI8VEG) (PHYTOLACCA AMERICANA ROOT - UNII:11E6VI8VEG) PHYTOLACCA AMERICANA ROOT 6 [hp_C] PULSATILLA VULGARIS (UNII: I76KB35JEV) (PULSATILLA VULGARIS - UNII:I76KB35JEV) PULSATILLA VULGARIS 6 [hp_C] Inactive Ingredients Ingredient Name Strength CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) LACTOSE (UNII: J2B2A4N98G) MAGNESIUM STEARATE (UNII: 70097M6I30) Product Characteristics Color white Score no score Shape ROUND (BOIRON) Size 9mm Flavor Imprint Code ; Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0220-9048-04 60 in 1 BLISTER PACK; Type 0: Not a Combination Product 01/01/1995 2 NDC:0220-9048-29 120 in 1 BLISTER PACK; Type 0: Not a Combination Product 12/09/2020 3 NDC:0220-9048-91 20 in 1 BLISTER PACK; Type 0: Not a Combination Product 07/14/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 01/01/1995 Labeler - Laboratoires Boiron (282560473) Registrant - Boiron Inc. (014892269) Establishment Name Address ID/FEI Business Operations Boiron 282560473 manufacture(0220-9048)