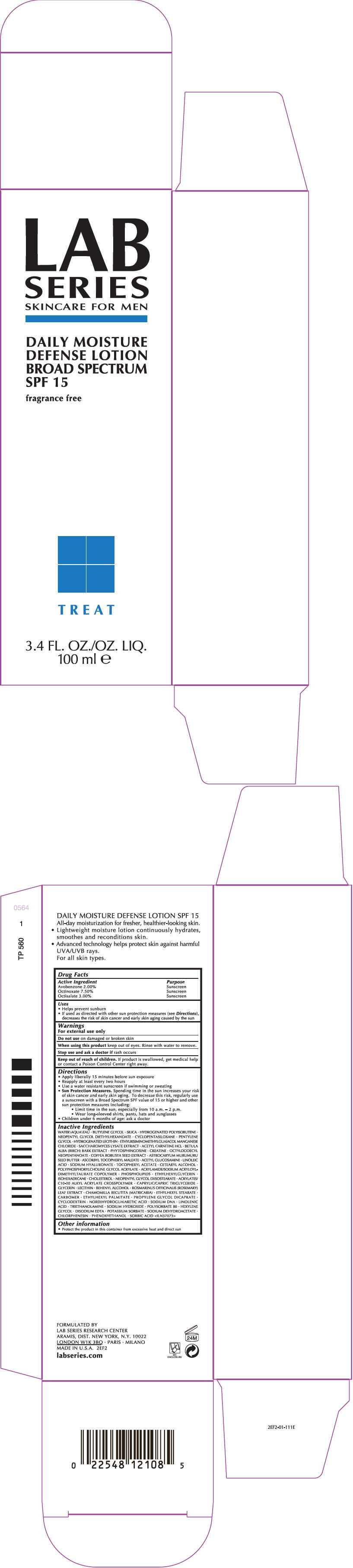
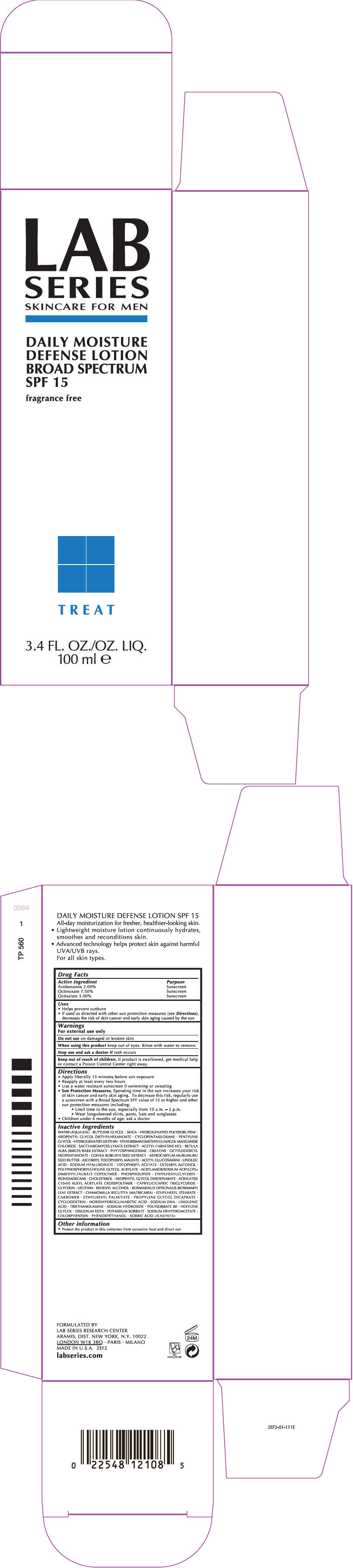
Label: LAB SERIES SKINCARE FOR MEN DAILY MOISTURE DEFENSE BROAD SPECTRUM SPF 15- avobenzone, octinoxate, and octisalate lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 42472-887-01 - Packager: ARAMIS INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 7, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- Helps prevent sunburn
- If used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
- Apply liberally 15 minutes before sun exposure
- Reapply at least every two hours
- Use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- Limit time in the sun, especially from 10 a.m. – 2 p.m.
- Wear long-sleeved shirts, pants, hats and sunglasses
- Children under 6 months of age: ask a doctor
-
Inactive Ingredients
WATER\AQUA\EAU • BUTYLENE GLYCOL • SILICA • HYDROGENATED POLYISOBUTENE • NEOPENTYL GLYCOL DIETHYLHEXANOATE • CYCLOPENTASILOXANE • PENTYLENE GLYCOL • HYDROGENATED LECITHIN • ETHYLBISIMINOMETHYLGUAIACOL MANGANESE CHLORIDE • SACCHAROMYCES LYSATE EXTRACT • ACETYL CARNITINE HCL • BETULA ALBA (BIRCH) BARK EXTRACT • PHYTOSPHINGOSINE • CREATINE • OCTYLDODECYL NEOPENTANOATE • COFFEA ROBUSTA SEED EXTRACT • ASTROCARYUM MURUMURU SEED BUTTER • ASCORBYL TOCOPHERYL MALEATE • ACETYL GLUCOSAMINE • LINOLEIC ACID • SODIUM HYALURONATE • TOCOPHERYL ACETATE • CETEARYL ALCOHOL • POLYPHOSPHORYLCHOLINE GLYCOL ACRYLATE • ACRYLAMIDE/SODIUM ACRYLOYLDIMETHYLTAURATE COPOLYMER • PHOSPHOLIPIDS • ETHYLHEXYLGLYCERIN • ISOHEXADECANE • CHOLESTEROL • NEOPENTYL GLYCOL DIISOSTEARATE • ACRYLATES/C10-30 ALKYL ACRYLATE CROSSPOLYMER • CAPRYLIC/CAPRIC TRIGLYCERIDE • GLYCERIN • LECITHIN • BEHENYL ALCOHOL • ROSMARINUS OFFICINALIS (ROSEMARY) LEAF EXTRACT • CHAMOMILLA RECUTITA (MATRICARIA) • ETHYLHEXYL STEARATE • CARBOMER • ETHYLHEXYL PALMITATE • PROPYLENE GLYCOL DICAPRATE • CYCLODEXTRIN • NORDIHYDROGUAIARETIC ACID • SODIUM DNA • LINOLENIC ACID • TRIETHANOLAMINE • SODIUM HYDROXIDE • POLYSORBATE 80 • HEXYLENE GLYCOL • DISODIUM EDTA • POTASSIUM SORBATE • SODIUM DEHYDROACETATE • CHLORPHENESIN • PHENOXYETHANOL • SORBIC ACID <ILN37073>
- Other information
- PRINCIPAL DISPLAY PANEL - 100 ml Tube Carton
-
INGREDIENTS AND APPEARANCE
LAB SERIES SKINCARE FOR MEN DAILY MOISTURE DEFENSE BROAD SPECTRUM SPF 15
avobenzone, octinoxate, and octisalate lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:42472-887 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 0.0212 g in 100 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.0795 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 0.0318 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) NEOPENTYL GLYCOL DIETHYLHEXANOATE (UNII: U68ZV6W62C) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) PENTYLENE GLYCOL (UNII: 50C1307PZG) ETHYLBISIMINOMETHYLGUAIACOL MANGANESE CHLORIDE (UNII: SM5YJ88LTU) SACCHAROMYCES LYSATE (UNII: R85W246Z1C) ACETYLCARNITINE HYDROCHLORIDE (UNII: NDW10MX58T) BETULA PUBESCENS BARK (UNII: 3R504894L9) PHYTOSPHINGOSINE (UNII: GIN46U9Q2Q) CREATINE (UNII: MU72812GK0) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) ROBUSTA COFFEE BEAN (UNII: V5032728L7) ASTROCARYUM MURUMURU SEED BUTTER (UNII: 12V64UPU6R) ASCORBYL TOCOPHERYL MALEATE (UNII: D2G6259XR5) N-ACETYLGLUCOSAMINE (UNII: V956696549) LINOLEIC ACID (UNII: 9KJL21T0QJ) HYALURONATE SODIUM (UNII: YSE9PPT4TH) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) ISOHEXADECANE (UNII: 918X1OUF1E) CHOLESTEROL (UNII: 97C5T2UQ7J) NEOPENTYL GLYCOL DIISOSTEARATE (UNII: 4M6OQ34JWW) GLYCERIN (UNII: PDC6A3C0OX) DOCOSANOL (UNII: 9G1OE216XY) ROSEMARY (UNII: IJ67X351P9) MATRICARIA CHAMOMILLA (UNII: G0R4UBI2ZZ) ETHYLHEXYL STEARATE (UNII: EG3PA2K3K5) ETHYLHEXYL PALMITATE (UNII: 2865993309) PROPYLENE GLYCOL DICAPRATE (UNII: U783H9JHWY) MASOPROCOL (UNII: 7BO8G1BYQU) LINOLENIC ACID (UNII: 0RBV727H71) TROLAMINE (UNII: 9O3K93S3TK) SODIUM HYDROXIDE (UNII: 55X04QC32I) POLYSORBATE 80 (UNII: 6OZP39ZG8H) HEXYLENE GLYCOL (UNII: KEH0A3F75J) EDETATE DISODIUM (UNII: 7FLD91C86K) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SODIUM DEHYDROACETATE (UNII: 8W46YN971G) CHLORPHENESIN (UNII: I670DAL4SZ) PHENOXYETHANOL (UNII: HIE492ZZ3T) SORBIC ACID (UNII: X045WJ989B) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42472-887-01 1 in 1 CARTON 01/01/2005 1 100 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 01/01/2005 Labeler - ARAMIS INC. (042918826) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COMPANY, THE 828534516 RELABEL(42472-887) , REPACK(42472-887) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD 244669714 MANUFACTURE(42472-887) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD. 202952982 MANUFACTURE(42472-887) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER N.V. 370151326 MANUFACTURE(42472-887) Establishment Name Address ID/FEI Business Operations LEN-RON MANUFACTURING DIVISION OF ARAMIS INC 809771152 MANUFACTURE(42472-887) Establishment Name Address ID/FEI Business Operations NORTHTEC INC 943871157 RELABEL(42472-887) , REPACK(42472-887) Establishment Name Address ID/FEI Business Operations PADC 1 949264774 RELABEL(42472-887) , REPACK(42472-887) Establishment Name Address ID/FEI Business Operations WHITMAN LABORATORIES, LTD. 216866277 MANUFACTURE(42472-887) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD 255175580 MANUFACTURE(42472-887)