Label: OXYMETAZOLINE HYDROCHLORIDE solution
- NDC Code(s): 63868-608-01
- Packager: CHAIN DRUG MARKETING ASSOCIATION INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 1, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
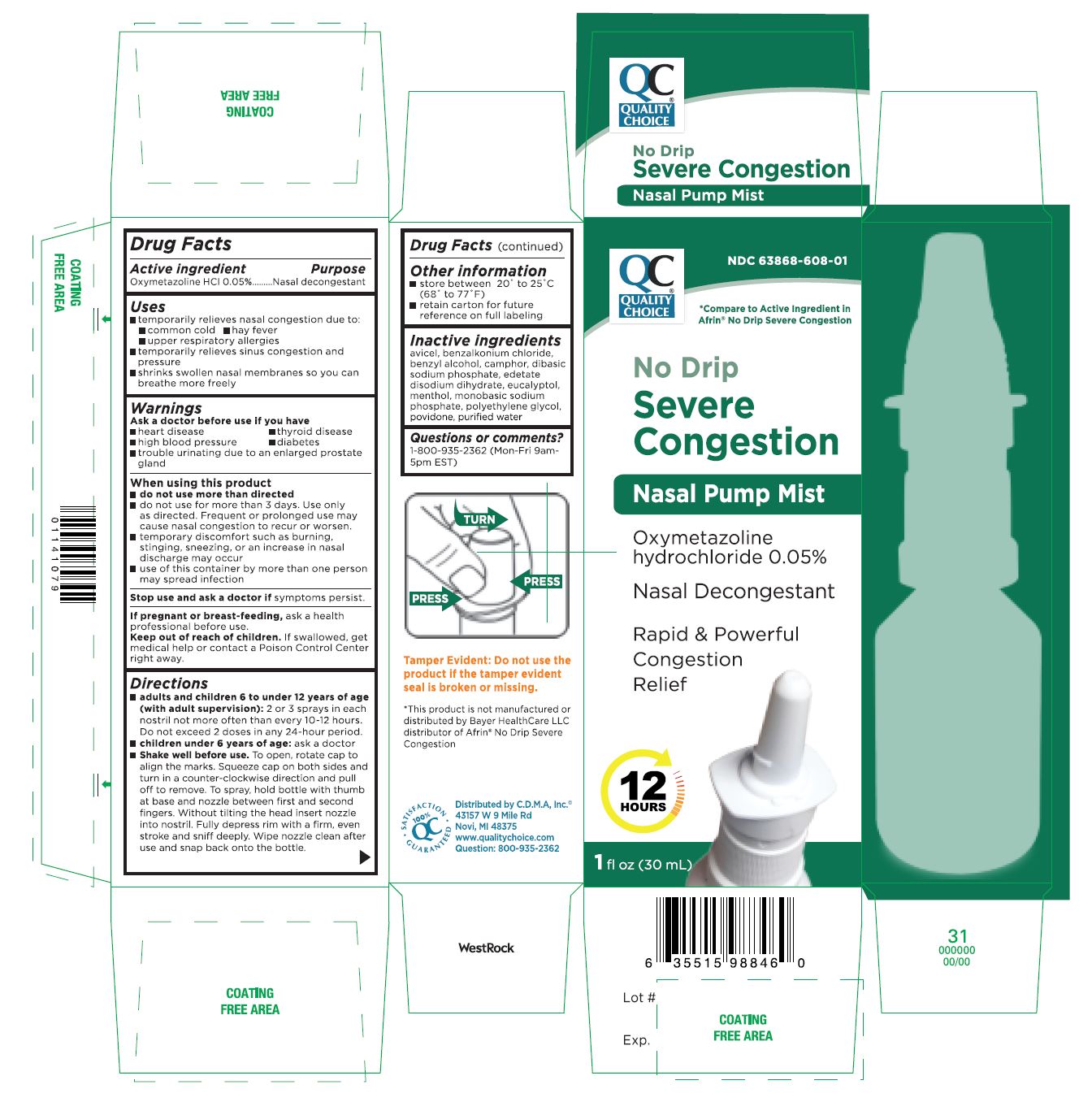
- Active Ingredient Purpose
- PURPOSE
- Uses
- Warnings
-
When using this product
- do not use more than directed
- do not use for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen.
- temporary discomfort such as burning, stinging, sneezing, or an increase in nasal discharge may occur
- use of this container by more than one person may spread infection
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
Directions
- adults and children 6 to under 12 years of age (with adult supervision): 2 or 3 sprays in each nostril not more often than every 10-12 hours. Do not exceed 2 doses in any 24-hour period.
- children under 6 years of age: ask a doctor
- Shake well before use. To open, rotate cap to align the marks. Squeeze cap on both sides and turn in a counter-clockwise direction and pull off to remove. To spray, hold bottle with thumb at base and nozzle between first and second fingers. Without tilting the head insert nozzle into nostril. Fully depress rim with a firm, even stroke and sniff deeply. Wipe nozzle clean after use and snap back onto the bottle.
- Other information
- Inactive ingredients
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
OXYMETAZOLINE HYDROCHLORIDE
oxymetazoline hydrochloride solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63868-608 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OXYMETAZOLINE HYDROCHLORIDE (UNII: K89MJ0S5VY) (OXYMETAZOLINE - UNII:8VLN5B44ZY) OXYMETAZOLINE HYDROCHLORIDE 0.05 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) BENZYL ALCOHOL (UNII: LKG8494WBH) CAMPHOR (NATURAL) (UNII: N20HL7Q941) SODIUM PHOSPHATE DIBASIC DIHYDRATE (UNII: 94255I6E2T) EDETATE DISODIUM (UNII: 7FLD91C86K) EUCALYPTOL (UNII: RV6J6604TK) MENTHOL (UNII: L7T10EIP3A) WATER (UNII: 059QF0KO0R) SODIUM PHOSPHATE, MONOBASIC (UNII: 3980JIH2SW) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE (UNII: FZ989GH94E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63868-608-01 30 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/15/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 03/15/2021 Labeler - CHAIN DRUG MARKETING ASSOCIATION INC (011920774) Registrant - Seaway Pharma Inc. (117218785) Establishment Name Address ID/FEI Business Operations Seaway Pharma Inc. 117218785 manufacture(63868-608)