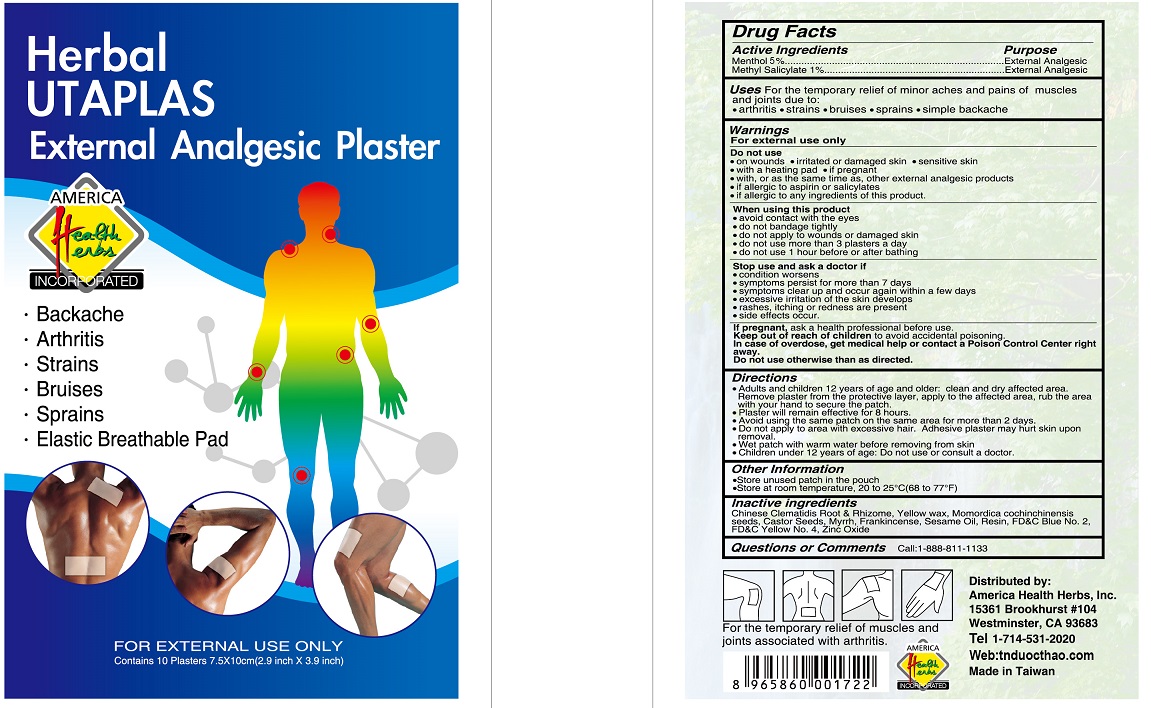
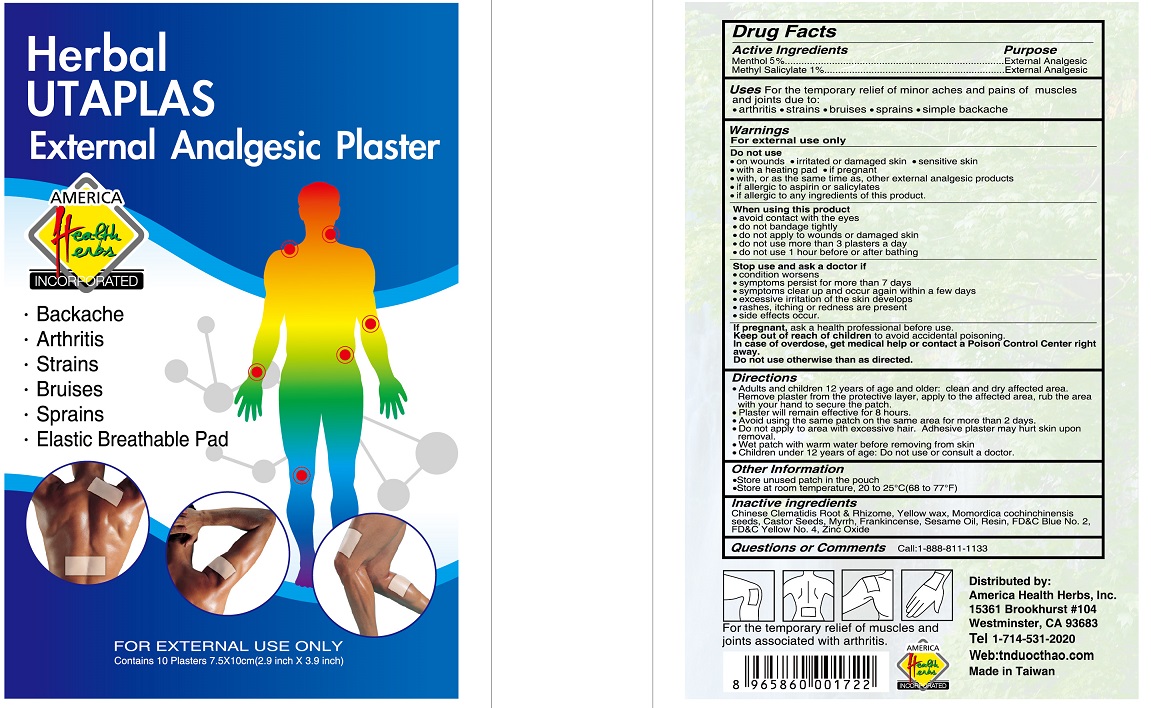
Label: HERBAL UTAPLAS EXTERNAL ANALGESIC PLASTER- menthol, methyl salicylate patch
- NDC Code(s): 82198-0016-1
- Packager: Big 5 Nutrition LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Purpose
- Uses
-
Warnings
For external use only
Do not use
● on wounds ● irritated or damaged skin ● sensitive skin
● with a heating pad ● if pregnant
● with, or as the same time as, other external analgesic products
● if allergic to aspirin or salicylates
● if allergic to any ingredients of this product.When using this product
● avoid contact with the eyes
● do not bandage tightly
● do not apply to wounds or damaged skin
● do not use more than 3 plasters a day
● do not use 1 hour before or after bathing -
Directions
● Adults and children 12 years of age and older: clean and dry affected area.
Remove plaster from the protective layer, apply to the affected area, rub the area
with your hand to secure the patch.
● Plaster will remain effective for 8 hours.
● Avoid using the same patch on the same area for more than 2 days.
● Do not apply to area with excessive hair. Adhesive plaster may hurt skin upon
removal.
● Wet patch with warm water before removing from skin
● Children under 12 years of age: Do not use or consult a doctor. - Other Information
- Inactive ingredients
- Questions or Comments
- Package Label
-
INGREDIENTS AND APPEARANCE
HERBAL UTAPLAS EXTERNAL ANALGESIC PLASTER
menthol, methyl salicylate patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:82198-0016 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL, UNSPECIFIED FORM - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 0.06 g in 1 g METHYL SALICYLATE (UNII: LAV5U5022Y) (SALICYLIC ACID - UNII:O414PZ4LPZ) METHYL SALICYLATE 0.1 g in 1 g Inactive Ingredients Ingredient Name Strength CLEMATIS CHINENSIS ROOT (UNII: 8Z18N528CU) INDIGOTINDISULFONATE SODIUM (UNII: D3741U8K7L) MOMORDICA COCHINCHINENSIS SEED (UNII: 2T87O1UPVD) MYRRH (UNII: JC71GJ1F3L) FRANKINCENSE (UNII: R9XLF1R1WM) PINUS MASSONIANA RESIN (UNII: 64S07U83T7) RICINUS COMMUNIS SEED (UNII: 7EK4SFN1TX) SESAME OIL (UNII: QX10HYY4QV) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) YELLOW WAX (UNII: 2ZA36H0S2V) ZINC OXIDE (UNII: SOI2LOH54Z) Product Characteristics Color green Score Shape RECTANGLE Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:82198-0016-1 10 in 1 BAG 01/01/2024 1 3.1 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 01/01/2024 Labeler - Big 5 Nutrition LLC (114574559) Establishment Name Address ID/FEI Business Operations TAIWAN SHUENN-AN BIOTECHNOLOGY PHARMACEUTICAL CO., LTD. 656348265 manufacture(82198-0016) , pack(82198-0016) , label(82198-0016)