Label: TING ATHETES FOOT- undecylenic acid, triclosan cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 51769-002-14, 51769-002-28 - Packager: ALL NATURAL DYNAMICS
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 22, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
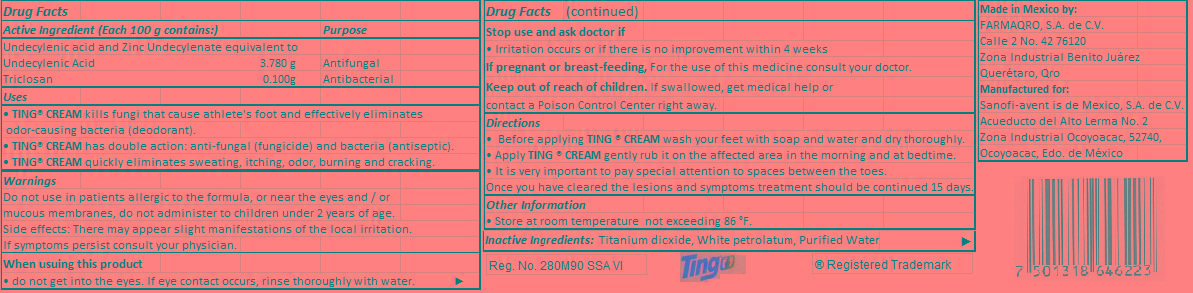
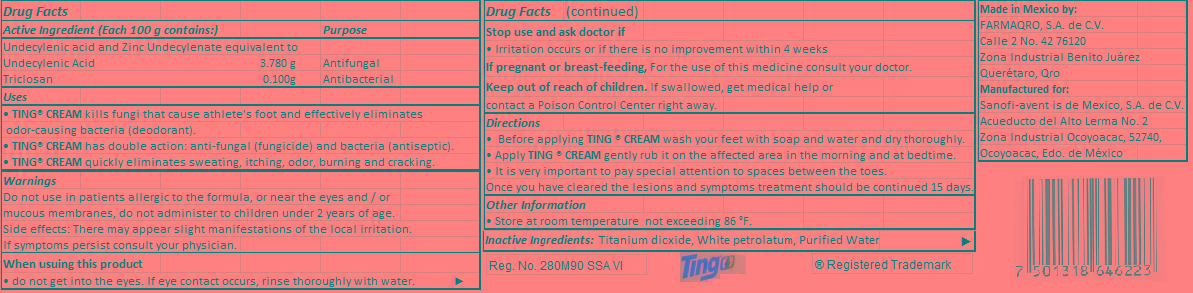
- Active Ingredient (Each 100 g contains:)
- Purpose
- Keep out of reach of children.
- Uses
- Warnings
-
Directions
• Before applying TING ® CREAM wash your feet with soap and water and dry thoroughly.
• Apply TING ® CREAM gently rub it on the affected area in the morning and at bedtime.
• It is very important to pay special attention to spaces between the toes.
Once you have cleared the lesions and symptoms treatment should be continued 15 days.
- Inactive ingredient
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TING ATHETES FOOT
undecylenic acid, triclosan creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51769-002 Route of Administration Cutaneous Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Undecylenic acid (UNII: K3D86KJ24N) (UNDECYLENIC ACID - UNII:K3D86KJ24N) Undecylenic acid 3.780 g in 100 g TRICLOSAN (UNII: 4NM5039Y5X) (TRICLOSAN - UNII:4NM5039Y5X) TRICLOSAN 0.100 g in 100 g Inactive Ingredients Ingredient Name Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) PETROLATUM (UNII: 4T6H12BN9U) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51769-002-28 28 g in 1 TUBE 2 NDC:51769-002-14 14 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333C 01/09/2013 Labeler - ALL NATURAL DYNAMICS (962732892) Establishment Name Address ID/FEI Business Operations Farmaqro, S.A. de C.V. 588161468 manufacture(51769-002)