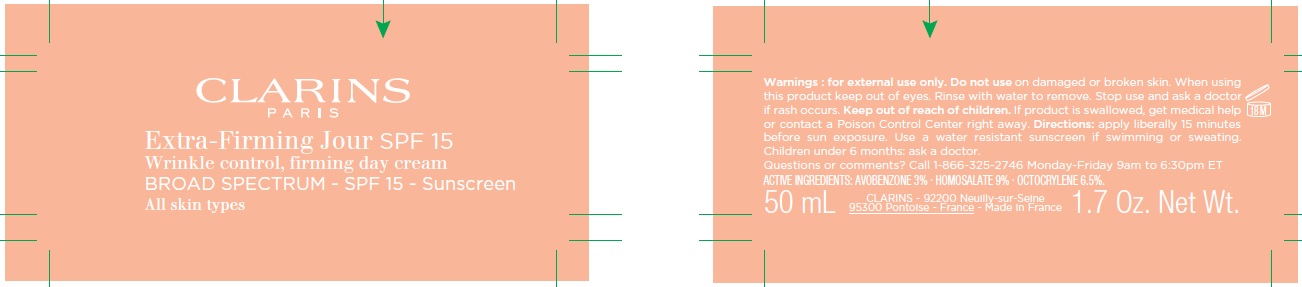
Label: CLARINS - EXTRA-FIRMING JOUR SPF 15 - WRINKLE, FIRMING DAY ALL SKIN TYPES - BROAD SPECTRUM SPF 15 SUNSCREEN- avobenzone, homosalate, octocrylene cream
-
NDC Code(s):
58668-9918-1,
58668-9918-2,
58668-9918-3,
58668-9918-4, view more58668-9918-5, 58668-9918-6
- Packager: Laboratoires Clarins
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 8, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug facts
- Active ingredients
- Uses:
- Warnings:
-
Directions:
• apply liberally 15 minutes before sun exposure • apply to all skin exposed to the sun • children under 6 months: Ask a doctor
• Spending time in the sun in creases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with broad spectrum SPF of 15 or higher and other sun protection measures including: • limit time in the sun, especially from 10 a.m. – 2 p.m. • wear long-sleeved shirts, pants, hats and sunglasses • reapply at least every 2 hours • use a water resistant sun screen if swimming or sweating Sun Protection Measures.
-
Inactive ingredients:
AQUA/WATER/EAU . GLYCERIN . BUTYLOCTYL SALICYLATE . BUTYROSPERMUM PARKII (SHEA) BUTTER . DIMETHICONE . C12-15 ALKYL BENZOATE . CETEARYL ALCOHOL . METHYL METHACRYLATE CROSSPOLYMER . POTASSIUM CETYL PHOSPHATE . PARFUM/FRAGRANCE . CETEARYL GLUCOSIDE . DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER . PHENOXYETHANOL . BUTYLENE GLYCOL . ACRYLATES/C10-30 ALKYL ACRYLATE CROSSPOLYMER . CAPRYLYL GLYCOL . TROMETHAMINE . AVENA SATIVA (OAT) KERNEL EXTRACT . DISODIUM EDTA . TOCOPHERYL ACETATE . XANTHAN GUM . DIMETHICONOL . TOCOPHEROL . ANIGOZANTHOS FLAVIDUS EXTRACT . CAPRYLIC/CAPRIC TRIGLYCERIDE . BETAINE . SILICA . SORBITOL . DIOSPYROS MESPILIFORMIS LEAF EXTRACT . MALTODEXTRIN . SANICULA EUROPAEA EXTRACT . SODIUM BENZOATE . KALANCHOE PINNATA LEAF EXTRACT . BALANITES ROXBURGHII SEED OIL . MITRACARPUS SCABER EXTRACT . CITRIC ACID . MALPIGHIA EMARGINATA (ACEROLA) SEED EXTRACT . LAPSANA COMMUNIS FLOWER/LEAF/STEM EXTRACT . FURCELLARIA LUMBRICALIS EXTRACT . CI 15985/YELLOW 6 . POTASSIUM SORBATE . MARIS SAL/SEA SALT/SEL MARIN. [S3095A]
- Other information:
- Questions or comments?
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
CLARINS - EXTRA-FIRMING JOUR SPF 15 - WRINKLE, FIRMING DAY ALL SKIN TYPES - BROAD SPECTRUM SPF 15 SUNSCREEN
avobenzone, homosalate, octocrylene creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:58668-9918 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 90 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 65 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) SHEA BUTTER (UNII: K49155WL9Y) DIMETHICONE (UNII: 92RU3N3Y1O) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) POTASSIUM CETYL PHOSPHATE (UNII: 03KCY6P7UT) CETEARYL GLUCOSIDE (UNII: 09FUA47KNA) PHENOXYETHANOL (UNII: HIE492ZZ3T) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) CAPRYLYL GLYCOL (UNII: 00YIU5438U) TROMETHAMINE (UNII: 023C2WHX2V) OAT (UNII: Z6J799EAJK) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) XANTHAN GUM (UNII: TTV12P4NEE) TOCOPHEROL (UNII: R0ZB2556P8) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) BETAINE (UNII: 3SCV180C9W) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SORBITOL (UNII: 506T60A25R) DIOSPYROS MESPILIFORMIS LEAF (UNII: 0R6X2JX6J1) MALTODEXTRIN (UNII: 7CVR7L4A2D) SODIUM BENZOATE (UNII: OJ245FE5EU) KALANCHOE PINNATA LEAF (UNII: 3R963LO08T) BALANITES ROXBURGHII SEED OIL (UNII: CZ67V4ZW0I) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) MALPIGHIA EMARGINATA SEED (UNII: 1X7L93686M) LAPSANA COMMUNIS FLOWERING TOP (UNII: T9B77O7PZA) FURCELLERAN (UNII: 30QS0PF14U) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SEA SALT (UNII: 87GE52P74G) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:58668-9918-1 1 in 1 CARTON 10/05/2017 1 50 mL in 1 JAR; Type 0: Not a Combination Product 2 NDC:58668-9918-2 1 in 1 CARTON 10/05/2017 12/15/2019 2 50 mL in 1 JAR; Type 0: Not a Combination Product 3 NDC:58668-9918-3 1 in 1 CARTON 10/05/2017 12/15/2019 3 15 mL in 1 TUBE; Type 0: Not a Combination Product 4 NDC:58668-9918-4 1 in 1 CARTON 10/05/2017 12/15/2019 4 5 mL in 1 TUBE; Type 0: Not a Combination Product 5 NDC:58668-9918-5 1 in 1 CARTON 10/05/2017 12/15/2019 5 2 mL in 1 BAG; Type 0: Not a Combination Product 6 NDC:58668-9918-6 1 in 1 CARTON 10/26/2017 12/15/2019 6 2 mL in 1 BAG; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 10/05/2017 Labeler - Laboratoires Clarins (266317555) Establishment Name Address ID/FEI Business Operations Laboratoires Clarins 266317555 manufacture(58668-9918)