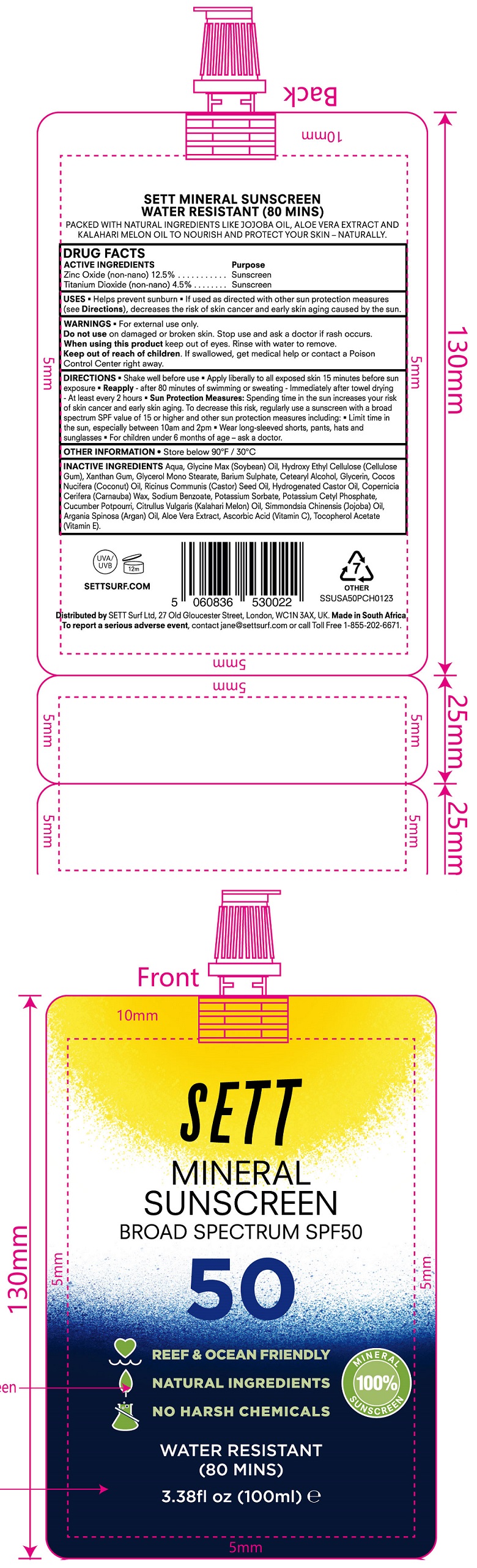
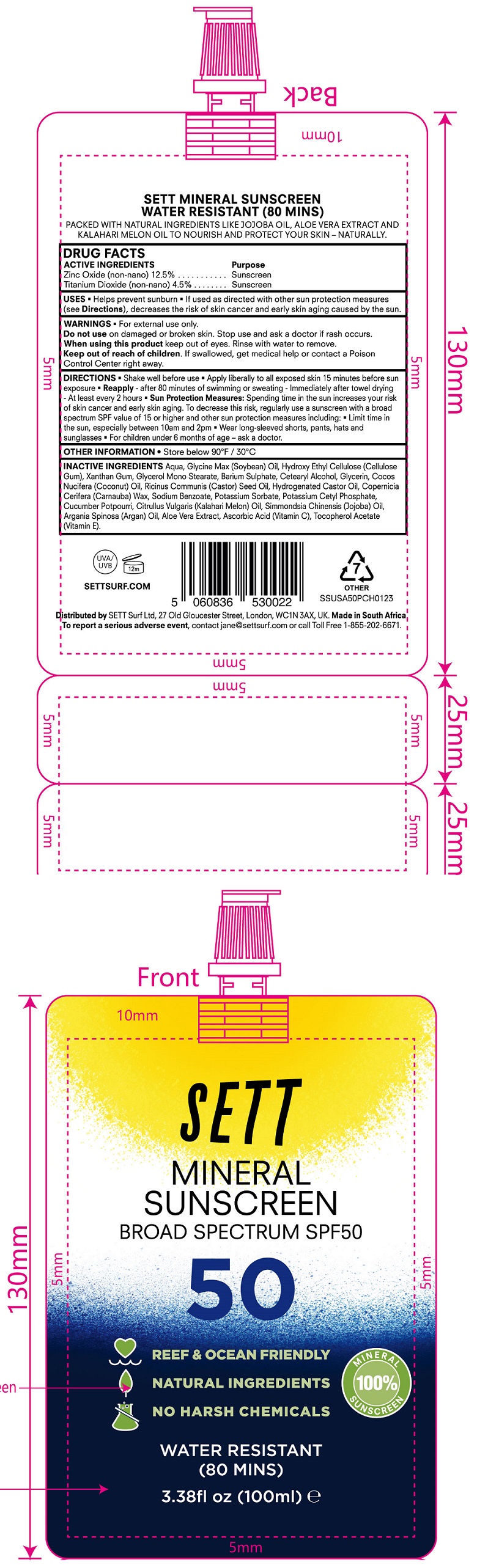
Label: SETT MINERAL SUNSCREEN SPF 50- zinc oxide, titanium dioxide cream
- NDC Code(s): 83478-000-00
- Packager: Sett Surf, Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 6, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DRUG FACTS
- ACTIVE INGREDIENTS
- USES
- WARNINGS
-
DIRECTIONS
• Shake well before use • Apply liberally to all exposed skin 15 minutes before sun exposure • - after 80 minutes of swimming or sweating • Immediately after towel drying - At least every 2 hours • Sun Protection Measures: Spending time in the sun increaases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF value of 15 or higher and other sun protection measres including: • Limit time in the sun, especially between 10am and 2pm • Wear long-sleeved shorts, pants, hats and sunglasses • For children under 6 months of age - ask a doctor. Reapply
- OTHER INFORMATION
-
INACTIVE INGREDIENTS
Aqua, Glycine Max (Soybean) Oil, Hydroxy Ethyl Cellulose (Cellulose Gum), Xanthan Gum, Glycerol Mono Stearate, Barium Sulphate, Cetearyl Alcohol, Glycerin, Cocos Nucifera (Coconut) Oil, Ricinus Communis (Castor) Seed Oil, Hydrogenated Castor Oil, Copernicia Cerifera (Carnauba) Wax, Sodium Benzoate, Potassium Sorbate, Potassium Cetyl Phosphate, Cucumber Potpourri, Citrullus Vulgaris (Kalahari Melon) Oil, Simmondsia Chenensis (Jojoba) Oil, Argania Spinosa (Argan) Oil, Aloe Vera Extract, Ascorbic Acid (Vitamin C), Tocopherol Acetate (Vitamin E).
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
SETT MINERAL SUNSCREEN SPF 50
zinc oxide, titanium dioxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83478-000 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 125 mg in 1 mL TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 45 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SOYBEAN OIL (UNII: 241ATL177A) HYDROXYMETHYL CELLULOSE (UNII: 273FM27VK1) XANTHAN GUM (UNII: TTV12P4NEE) BARIUM SULFATE (UNII: 25BB7EKE2E) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) GLYCERIN (UNII: PDC6A3C0OX) COCONUT OIL (UNII: Q9L0O73W7L) CASTOR OIL (UNII: D5340Y2I9G) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) CARNAUBA WAX (UNII: R12CBM0EIZ) SODIUM BENZOATE (UNII: OJ245FE5EU) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) POTASSIUM CETYL PHOSPHATE (UNII: 03KCY6P7UT) JOJOBA OIL (UNII: 724GKU717M) ARGAN OIL (UNII: 4V59G5UW9X) ALOE VERA LEAF (UNII: ZY81Z83H0X) ASCORBIC ACID (UNII: PQ6CK8PD0R) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83478-000-00 100 mL in 1 POUCH; Type 0: Not a Combination Product 08/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 08/01/2023 Labeler - Sett Surf, Ltd. (225883876)