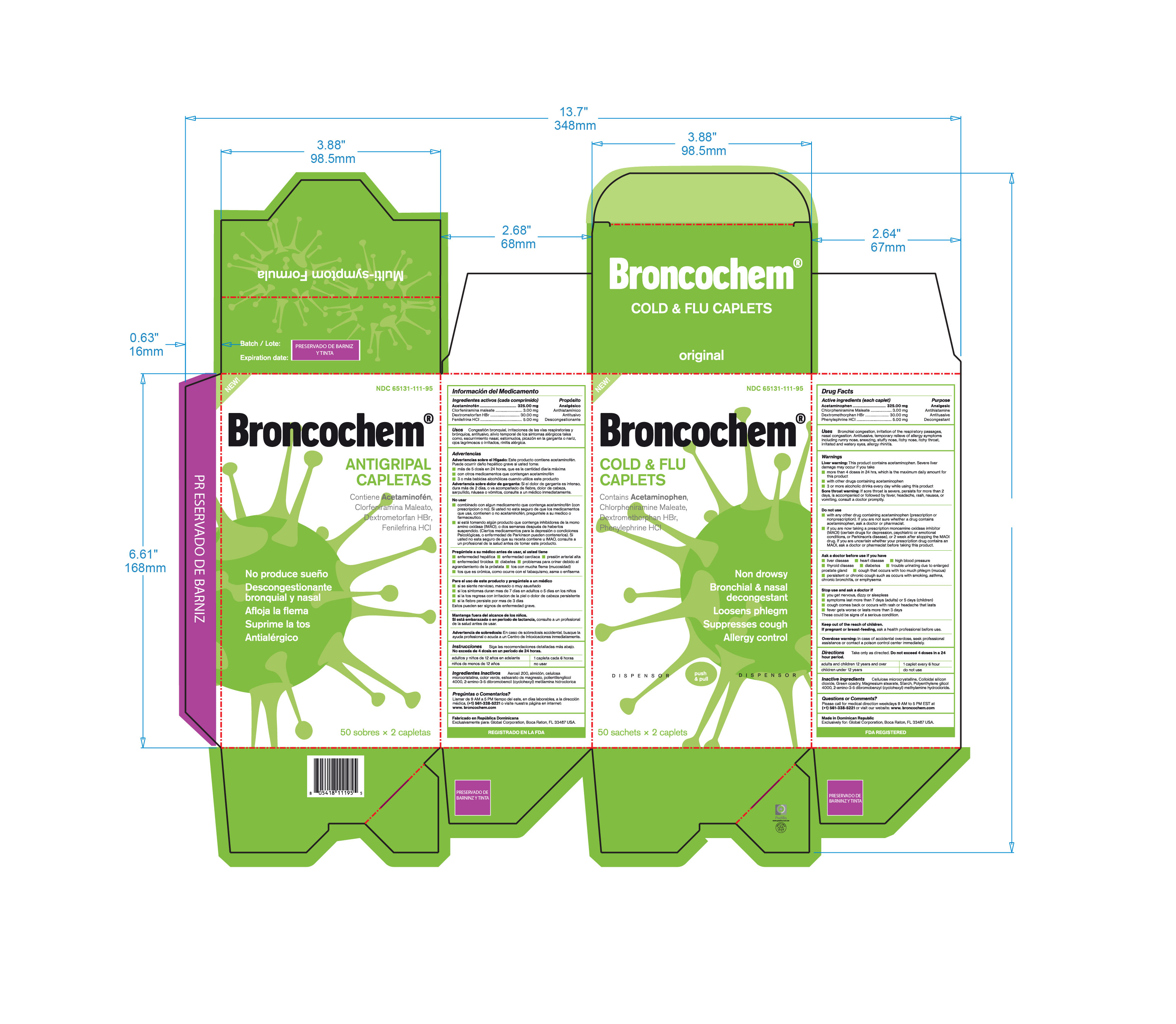
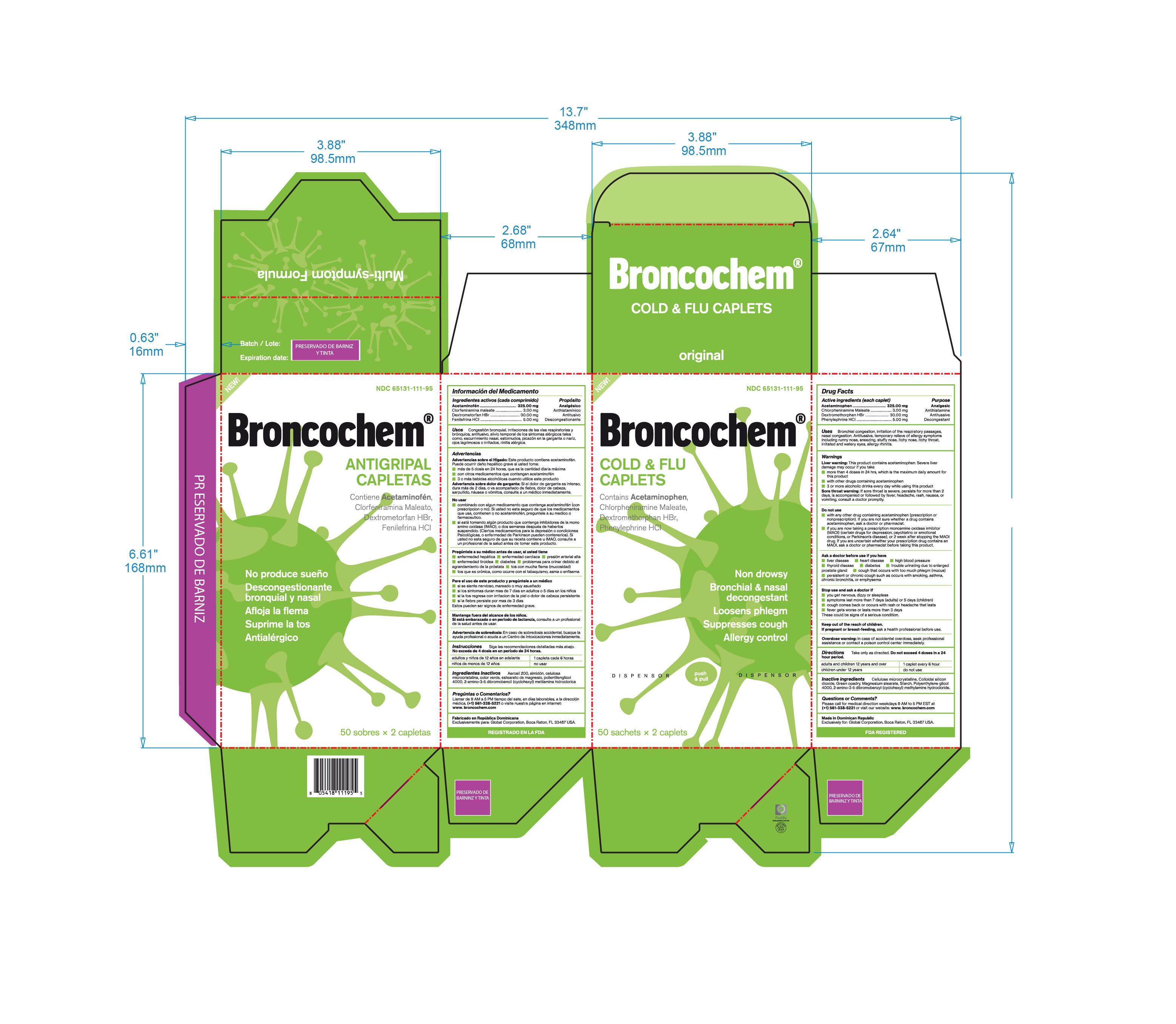
Label: BRONCOCHEM COLD AND FLU CAPLETS- acetaminophen-dextromethorphan hbr-phenylephrine hcl-chlorpheniramine maleate tablet, coated
- NDC Code(s): 65131-111-21, 65131-111-95
- Packager: LABOTORIO MAGNACHEM INTERNATIONAL SRL
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 19, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Warnings
Unless directed by a physician do not take this product if persistent or chronic cough such as occurs with, smoking, asthma, chronic bronchitis, emphysema, or if cough is accompained by excessive phlem (mucus). Likewise if you have heart disease, diabetes, or difficulty in urination due to enlargement of the prostate gland, do not take this product unless directed by a physician.
If nervousness, dizziness, or sleeplessness occurs, discontinue use or consult a doctor.
If symptoms do not improve within 7 days or are accompanied by fever, consult a doctor.
A persistent cough may be a sign of a serius condition. If cough or other symptoms persist or not improve with any drug, if you are pregnant or nursing a baby, seeh the advice of a health professional before using this product.
Unless directed by a doctor, do not take this product if you are presently taking another product containing pseudoephedryne HCl, or if you are taking this product precaution must be taken when driving a motor vehicle or operating machinery.
- Active Ingredients
- Purpose
- Keep Out Of The Reach Of Children
- Indications and Usage
- Indications and Usage
-
Drug Interactions
Do not use if you are taking a prescription monoamine oxidase inhibitor (MAOI), (Certain drugs for depression, psychiatric or emotional conditions, or Parkinsons disease) or for 2 weeks after stopping that MAOI drug, if you are uncertain whether your prescription drug contains an MAOI, consult a health professional before taking this product
- Inactive Ingredients
- Package Label Principal Display Panel
- Package Label Principal Display Panel
-
INGREDIENTS AND APPEARANCE
BRONCOCHEM COLD AND FLU CAPLETS
acetaminophen-dextromethorphan hbr-phenylephrine hcl-chlorpheniramine maleate tablet, coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65131-111 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 30 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE 5 mg CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 3 mg Inactive Ingredients Ingredient Name Strength CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) 250 mg STARCH, PREGELATINIZED CORN (UNII: O8232NY3SJ) 142 mg SILICON DIOXIDE (UNII: ETJ7Z6XBU4) 2 mg MAGNESIUM STEARATE (UNII: 70097M6I30) 2.8 mg PEG-80 STEARATE (UNII: 1X2OAT3BFK) 1.6 mg Product Characteristics Color green Score no score Shape capsule Size 18mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65131-111-21 12 in 1 BOX 12/30/2016 1 NDC:65131-111-95 50 in 1 BOX 1 2 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 01/01/2010 Labeler - LABOTORIO MAGNACHEM INTERNATIONAL SRL (871446100) Registrant - LABORATORIO MAGNACHEM INTERNATIONAL SRL (871446100) Establishment Name Address ID/FEI Business Operations LABORATORIO MAGNACHEM INTERNATIONAL SRL 871446100 manufacture(65131-111)