Label: BUTEQUINE- phenylbutazone paste
- NDC Code(s): 17030-100-60
- Packager: Vetoquinol USA, Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Animal Drug Application
Drug Label Information
Updated November 16, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
DESCRIPTION: Butequine™ paste is a synthetic, non-hormonal anti-inflammatory, antipyretic compound useful in the management of inflammatory conditions. The apparent analgesic effect is probably related mainly to the compound's anti-inflammatory properties. Chemically, phenylbutazone is 4-butyl-1, 2-diphenyl-3, 5-pyrazolidinedione. It is a pyrazalone derivative, entirely unrelated to the steroid hormones, and has the following structural formula:
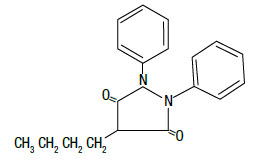
- VETERINARY INDICATIONS
- CONTRAINDICATIONS
- WARNINGS
-
PRECAUTIONS
PRECAUTIONS: Stop medication at the first sign of gastrointestinal upset, jaundice, or blood dyscrasia. Authenticated cases of agranulocytosis associated with the drug have occurred in man; fatal reactions, although rare, have been reported in dogs after long-term therapy. To guard against the possibility, conduct routine blood counts at weekly intervals during the early phase of therapy and at intervals of two weeks thereafter. Any significant fall in the total white blood cell count, relative decrease in granulocytes, or black or tarry stools should be regarded as a signal for immediate cessation of therapy and institution of appropriate counter-measures. In the treatment of inflammatory conditions associated with infections, specific anti-infective therapy is required.
- DOSAGE & ADMINISTRATION
-
Guidelines to Successful Therapy:
- Important - To avoid overdosing and to ensure the correct first dose, move ring on plunger to the appropriate dosage position before administration (see Figure 1). The edge of the ring closest to the syringe barrel should be aligned to the gram/dosage mark on the plunger.
- When administering Butequine™ paste the oral cavity should be empty. Set ring on plunger to deliver the appropriate dosage based on weight (see Figure 1). Deposit paste on the back of tongue by depressing plunger.
- Use a relatively high dose for the first 48 hours (not to exceed 4 g daily), then reduce gradually to a maintenance dose. Maintain lowest dose capable of producing desired clinical response.
- Response to Butequine™ paste therapy is prompt, usually occuring within 24 hours. If no significant clinical response is evident after 5 days, reevaluate diagnosis and therapeutic approach.
- Many chronic conditions will respond to Butequine™ paste therapy, but discontinuance of treatment may result in recurrence of symptoms.
- STORAGE AND HANDLING
- HOW SUPPLIED
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
-
CONTACT INFORMATION:
To report suspected adverse events, contact Vetoquinol USA, Inc. at 1-800-835-9496. For technical assistance or to obtain a copy of the Safety Data Sheet (SDS), contact Vetoquinol USA, Inc. at 1-800-267-5707. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDAVETS or online at www.fda.gov/reportanimalae
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 60 mL Syringe Carton
-
INGREDIENTS AND APPEARANCE
BUTEQUINE
phenylbutazone pasteProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:17030-100 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLBUTAZONE (UNII: GN5P7K3T8S) (PHENYLBUTAZONE - UNII:GN5P7K3T8S) PHENYLBUTAZONE 1 g in 3 mL Product Characteristics Color Score Shape Size Flavor APPLE Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:17030-100-60 1 in 1 CARTON 1 60 mL in 1 SYRINGE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200266 01/15/2020 Labeler - Vetoquinol USA, Inc. (106824209) Establishment Name Address ID/FEI Business Operations Nantong Jinghua Pharmaceutical Co., Ltd 421303119 API MANUFACTURE Establishment Name Address ID/FEI Business Operations Med-Pharmex, Inc 025353699 MANUFACTURE Establishment Name Address ID/FEI Business Operations Vetoquinol N.-A. INC 202919940 ANALYSIS, API MANUFACTURE, LABEL, MANUFACTURE, PACK



