Label: ACETAMINOPHEN DIPHENHYDRAMINE HCL tablet
- NDC Code(s): 21130-354-02, 21130-354-08
- Packager: BETTER LIVING BRANDS LLC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 18, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
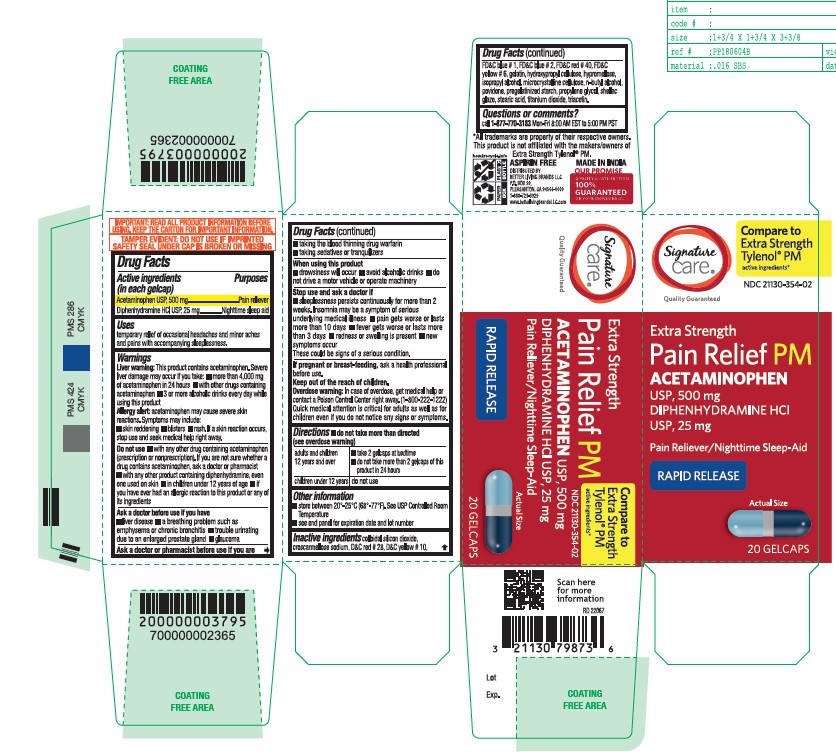
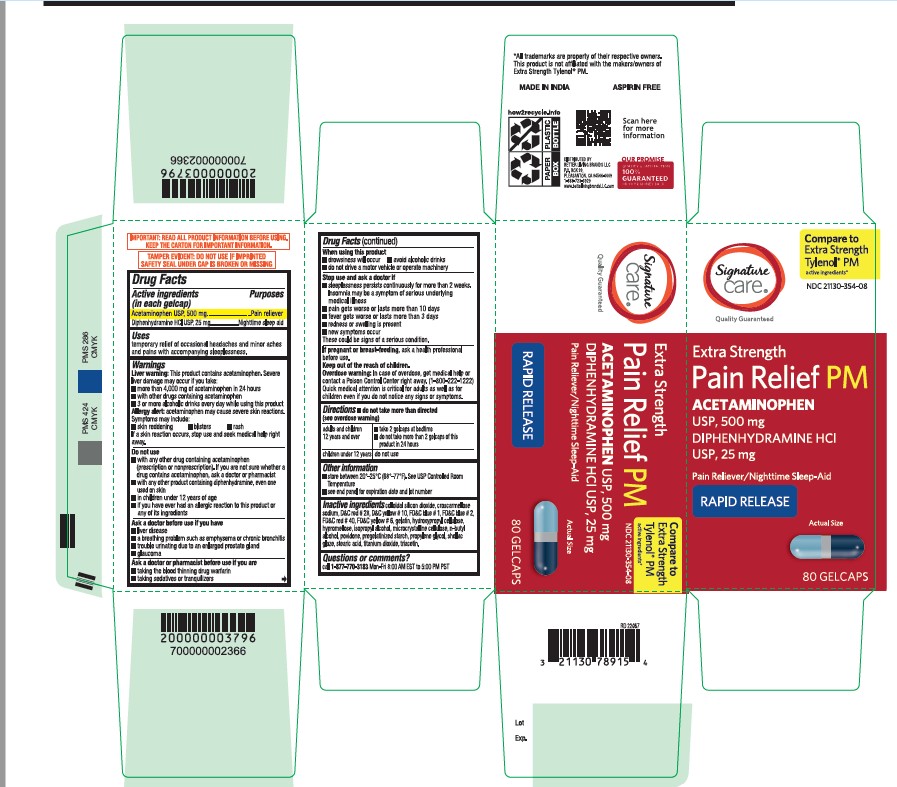
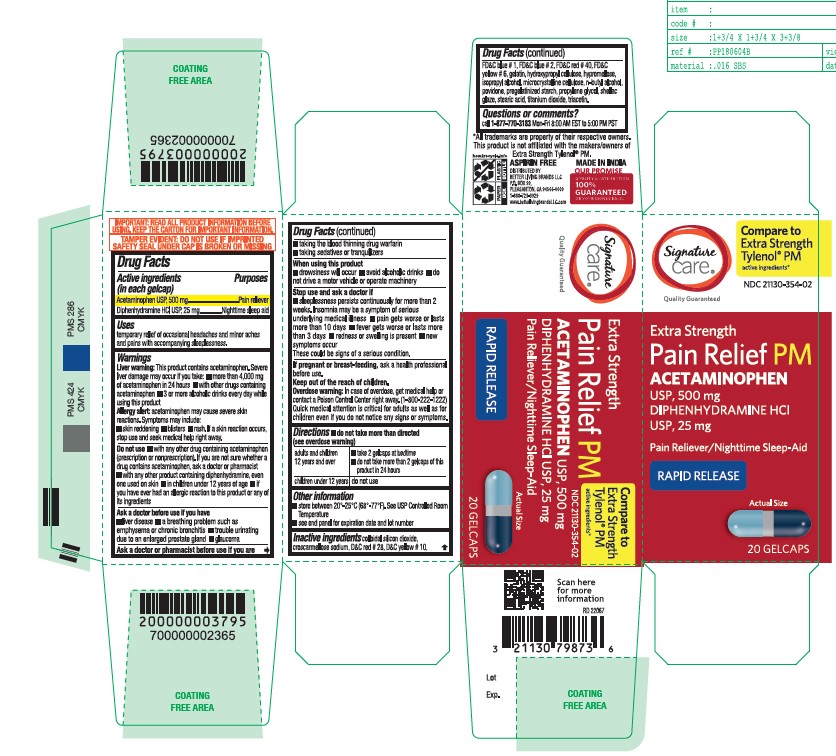
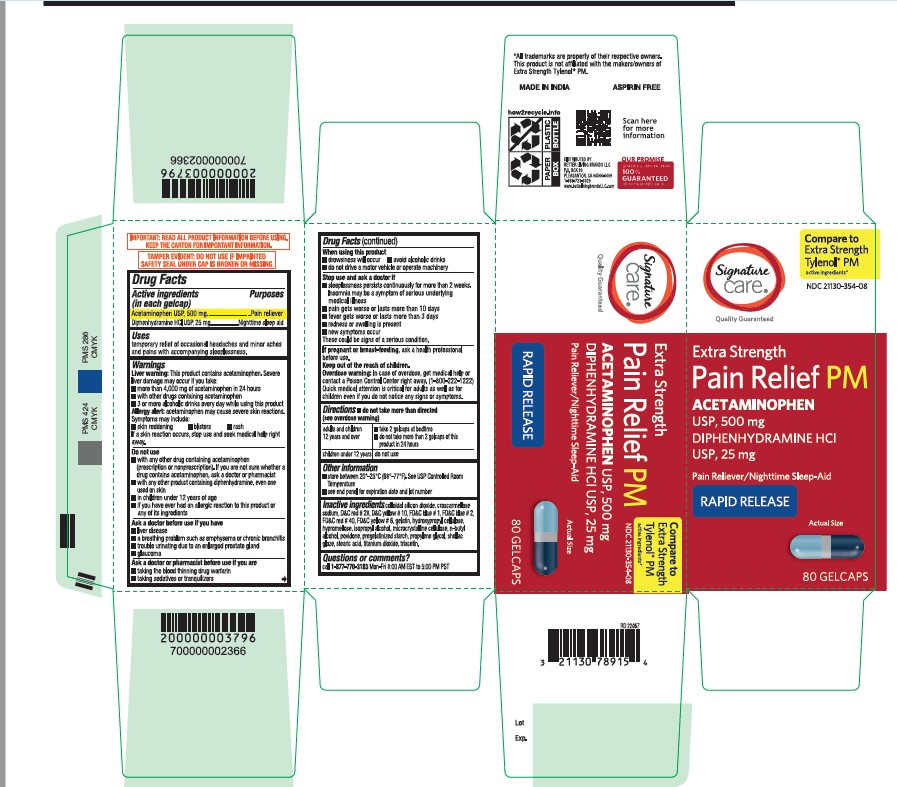
- Active ingredient
- Purpose
- Uses
- Liver warning
- Allergy alert
-
Do not use
■ with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
■ with any other product containing diphenhydramine, even one used on skin
■ in children under 12 years of age
■ if you have ever had an allergic reaction to this product or any of its ingredients - Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- When using this product
-
Stop use and ask a doctor if
■ sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of serious underlying medical illness.
■ pain gets worse or lasts more than 10 days
■ fever gets worse or lasts more than 3 days
■ redness or swelling is present
■ new symptoms occur
These could be signs of a serious condition. - If pregnant or breast-feeding
- Keep out of the reach of children
- Overdose warning
- do not take more than directed (see overdose warning)
- Other information
-
Inactive Ingrdients
colloidal silicon dioxide, croscarmellose sodium, D&C red#28, D&C yellow#10, FD&C blue#1, FD&C blue #2, FD&C red #40, FD&C yellow #6, gelatin, hydroxypropyl cellulose, hypromellose, isopropyl alcohol, microcrystalline cellulose, n-butyl alcohol, povidone, pregelatinized starch, propylene glycol, shellac glaze, stearic acid, titanium dioxide, triacetin
- Questions or Comments?
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ACETAMINOPHEN DIPHENHYDRAMINE HCL
acetaminophen diphenhydramine hcl tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21130-354 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 500 mg DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 25 mg Inactive Ingredients Ingredient Name Strength GELATIN (UNII: 2G86QN327L) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) D&C RED NO. 28 (UNII: 767IP0Y5NH) BUTYL ALCOHOL (UNII: 8PJ61P6TS3) FD&C RED NO. 40 (UNII: WZB9127XOA) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) STEARIC ACID (UNII: 4ELV7Z65AP) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TRIACETIN (UNII: XHX3C3X673) HYPROMELLOSE 2208 (100 MPA.S) (UNII: B1QE5P712K) STARCH, CORN (UNII: O8232NY3SJ) SHELLAC (UNII: 46N107B71O) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) ISOPROPYL ALCOHOL (UNII: ND2M416302) POVIDONE K30 (UNII: U725QWY32X) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYDROXYPROPYL CELLULOSE, UNSPECIFIED (UNII: 9XZ8H6N6OH) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) Product Characteristics Color gray ((Encapsulated gray color tablets with dark blue opaque and light blue opaque hard gelatin shells)) Score no score Shape OVAL Size 20mm Flavor Imprint Code G3 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21130-354-02 20 in 1 BOTTLE; Type 0: Not a Combination Product 12/12/2022 2 NDC:21130-354-08 80 in 1 BOTTLE; Type 0: Not a Combination Product 12/12/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 12/12/2022 Labeler - BETTER LIVING BRANDS LLC. (009137209)