Label: DIAZOXIDE suspension
- NDC Code(s): 0254-1010-19
- Packager: Par Pharmaceutical, Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated October 2, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Diazoxide Oral Suspension, USP is a nondiuretic benzothiadiazine derivative taken orally for the management of symptomatic hypoglycemia. The suspension contains 50 mg of diazoxide, USP in each milliliter and has a chocolate-mint flavor; alcohol content is approximately 7.29%. Other ingredients: Sorbitol solution, chocolate mint type flavor, propylene glycol, magnesium aluminum silicate, carboxymethycellulose sodium, sodium benzoate, methylparaben, poloxamer 188, propylparaben, FD&C Red No. 40, FD&C Yellow No. 6 and purified water. Hydrochloric acid may be added to adjust pH.
Diazoxide has the following structural formula:
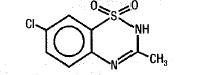
Diazoxide is 7-chloro-3-methyl-2H-1,2,4-benzothiadiazine 1,1-dioxide with the empirical formula C8H7CIN2O2S and the molecular weight 230.7. It is a white powder practically insoluble to sparingly soluble in water.
-
CLINICAL PHARMACOLOGY
Diazoxide administered orally produces a prompt dose-related increase in blood glucose level, due primarily to an inhibition of insulin release from the pancreas, and also to an extrapancreatic effect. The hyperglycemic effect begins within an hour and generally lasts no more than eight hours in the presence of normal renal function.
Diazoxide decreases the excretion of sodium and water, resulting in fluid retention which may be clinically significant.
The hypotensive effect of diazoxide on blood pressure is usually not marked with the oral preparation. This contrasts with the intravenous preparation of diazoxide (see ADVERSE REACTIONS).
Other pharmacologic actions of diazoxide include increased pulse rate; increased serum uric acid levels due to decreased excretion; increased serum levels of free fatty acids’ decreased chloride excretion; decreased para-aminohippuric acid; (PAH) clearance with no appreciable effect on glomerular filtration rate.
The concomitant administration of a benzothiazide diuretic may intensify the hyperglycemic and hyperuricemic effects of diazoxide. In the presence of hypokalemia, hyperglycemic effects are also potentiated.
Diazoxide-induced hyperglycemia is reversed by the administration of insulin or tolbutamide. The inhibition of insulin release by diazoxide is antagonized by alpha-adrenergic blocking agents.
Diazoxide is extensively bound (more than 90%) to serum proteins, and is excreted in the kidneys. The plasma half-life following I.V. administration is 28 ± 8.3 hours. Limited data on oral administration revealed a half-life of 24 and 36 hours in two adults. In four children aged four months to six years, the plasma half-life varied from 9.5 to 24 hours on long-term oral administration. The half-life may be prolonged following overdosage, and in patients with impaired renal function.
-
INDICATIONS & USAGE
Diazoxide oral suspension is useful in the management of hypoglycemia due to hyperinsulinism associated with the following conditions:
Adults: Inoperable islet cell adenoma or carcinoma, or extrapancreatic malignancy.
Infants and Children: Leucine sensitivity, islet cell hyperplasia, nesidioblastosis, extrapancreatic malignancy, islet cell adenoma, or adenomatosis. Diazoxide oral suspension may be used preoperatively as a temporary measure, and postoperatively, if hypoglycemia persists.
Diazoxide oral suspension should be used only after a diagnosis of hypoglycemia due to one of the above conditions has been definitely established. When other specific medical therapy or surgical management either has been unsuccessful or is not feasible, treatment with diazoxide oral suspension should be considered.
- CONTRAINDICATIONS
-
WARNINGS
The antidiuretic property of diazoxide may lead to significant fluid retention, which in patients with compromised cardiac reserve, may precipitate congestive heart failure. The fluid retention will respond to conventional therapy with diuretics.
It should be noted that concomitantly administered thiazides may potentiate the hyperglycemic and hyperuricemic actions of diazoxide (See DRUG INTERACTIONS and ANIMAL PHARMACOLOGY AND/OR TOXICOLOGY).
Ketoacidosis and nonketotic hyperosmolar coma have been reported in patients treated with recommended doses of diazoxide oral suspension usually during intercurrent illness. Prompt recognition and treatment are essential (See OVERDOSAGE), and prolonged surveillance following the acute episode is necessary because of the long drug half-life of approximately 30 hours. The occurrence of these serious events may be reduced by careful education of patients regarding the need for monitoring the urine for sugar and ketones and for prompt reporting of abnormal findings and unusual symptoms to the physician. Transient cataracts occurred in association with hyperosmolar coma in an infant, and subsided on correction of the hyper-osmolarity. Cataracts have been observed in several animals receiving daily doses of intravenous or oral diazoxide.
The development of abnormal facial features in four children treated chronically (>4 years) with diazoxide oral suspension for hypoglycemia hyperinsulinism in the same clinic has been reported.
Pulmonary Hypertension in Neonates and Infants
There have been postmarketing reports of pulmonary hypertension occurring in infants and neonates treated with diazoxide. The cases were reversible upon discontinuation of the drug. Monitor patients, especially those with risk factors for pulmonary hypertension, for respiratory distress and discontinue diazoxide if pulmonary hypertension is suspected.
-
PRECAUTIONS
GENERAL PRECAUTIONS
Treatment with diazoxide oral suspension should be initiated under close clinical supervision, with careful monitoring of blood glucose and clinical response until the patient’s condition has stabilized. This usually requires several days. If not effective in two to three weeks, the drug should be discontinued.
Prolonged treatment requires regular monitoring of the urine for sugar and ketones, especially under stress conditions, with prompt reporting of any abnormalities to the physician. Additionally, blood sugar levels should be monitored periodically by the physician to determine the need for dose adjustment.
The effects of diazoxide on the hematopoietic system and the level of serum uric acid should be kept in mind; the latter should be considered particularly in patients with hyperuricemia or a history of gout.
In some patients, higher blood levels have been observed with the oral suspension than with the capsule formulation of diazoxide. Dosage should be adjusted as necessary in individual patients if changed from one formulation to the other.
Since the plasma half-life of diazoxide is prolonged in patients with impaired renal function, a reduced dosage should be considered. Serum electrolyte levels should also be evaluated for such patients.
The antihypertensive effect of other drugs may be enhanced by diazoxide oral suspension and this should be kept in mind when administering it concomitantly with antihypertensive agents.
Because of the protein binding, administration of diazoxide oral suspension with coumarin or its derivatives may require reduction in the dosage of the anticoagulant, although there has been no reported evidence of excessive anticoagulant effect. In addition, diazoxide may possibly displace bilirubin from albumin; this should be kept in mind particularly when treating newborns with increased bilirubinemia.
Pulmonary hypertension has been reported in neonates and young infants treated with diazoxide. (see WARNINGS)
INFORMATION FOR PATIENTS
During treatment with diazoxide oral suspension the patient should be advised to consult regularly with the physician and to cooperate in the periodic monitoring of his condition by laboratory tests. In addition, the patient should be advised:
- to take the drug on a regular schedule as prescribed, not to skip doses, not to take extra doses;
- not to use this drug with other medications unless this is done with the physician’s advice;
- not to allow anyone else to take this medication;
- to follow dietary instructions;
- to report promptly any adverse effects (i.e., increased urinary frequency, increased thirst, fruity breath odor);
- to report pregnancy or to discuss plans for pregnancy.
LABORATORY TESTS
The following procedures may be especially important in patient monitoring (not necessarily inclusive); blood glucose determinations (recommended at periodic intervals in patients taking diazoxide orally for treatment of hypoglycemia, until stabilized); blood urea nitrogen (BUN) determinations and creatinine clearance determinations; hematocrit determinations; platelet count determinations; total and differential leukocyte counts; serum aspartate aminotransferase (AST) level determinations; serum uric acid level determinations; and urine testing for glucose and ketones (in patients being treated with diazoxide for hypoglycemia, semiquantitative estimation of sugar and ketones in serum performed by the patient and reported to the physician provides frequent and relatively inexpensive monitoring of the condition).
DRUG INTERACTIONS
Since diazoxide is highly bound to serum proteins, it may displace other substances which are also bound to protein, such as bilirubin or coumarin and its derivatives, resulting in higher blood levels of these substances. Concomitant administration of oral diazoxide and diphenylhydantoin may result in a loss of seizure control. These potential interactions must be considered when administering diazoxide capsules or suspension.
The concomitant administration of thiazides or other commonly used diuretics may potentiate the hyperglycemic and hyperuricemic effects of diazoxide.
DRUG & OR LABORATORY TEST INTERACTIONS
The hyperglycemic and hyperuricemic effects of diazoxide preclude proper assessment of these metabolic states. Increased renin secretion, IgG concentrations and decreased cortisol secretions have also been noted. Diazoxide inhibits glucagon-stimulated insulin release and causes a false-negative insulin response to glucagon.
CARCINOGENESIS & MUTAGENESIS & IMPAIRMENT OF FERTILITY
No long-term animal dosing study has been done to evaluate the carcinogenic potential of diazoxide. No laboratory study of mutagenic potential or animal study of effects on fertility has been done.
Reproduction studies using the oral preparation in rats have revealed increased fetal resorptions and delayed parturition, as well as fetal skeletal anomalies; evidence of skeletal and cardiac teratogenic effects in rabbits has been noted with intravenous administration. The drug has also been demonstrated to cross the placental barrier in animals and to cause degeneration of the fetal pancreatic beta cells (See ANIMAL PHARMACOLOGY AND/OR TOXICOLOGY). Since there are no adequate data on fetal effects of this drug when given to pregnant women, safety in pregnancy has not been established. When the use of diazoxide oral suspension is considered, the indications should be limited to those specified above for adults (See INDICATIONS AND USAGE), and the potential benefits to the mother must be weighed against possible harmful effects to the fetus.
Non-teratogenic effects:
Diazoxide crosses the placental barrier and appears in cord blood. When given to the mother prior to delivery of the infant, the drug may produce fetal or neonatal hyperbilirubinemia, thrombocytopenia, altered carbohydrate metabolism, and possibly other side effects that have occurred in adults.
Alopecia and hypertrichosis lanuginosa have occurred in infants whose mothers received oral diazoxide during the last 19 to 60 days of pregnancy.
LABOR & DELIVERY
Since intravenous administration of the drug during labor may cause cessation of uterine contractions, and administration of oxytocic agents may be required to reinstate labor, caution is advised in administering diazoxide oral suspension at that time.
NURSING MOTHERS
Information is not available concerning the passage of diazoxide in breast milk. Because many drugs are excreted in human milk and because of the potential for adverse reactions from diazoxide in nursing infants, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
-
ADVERSE REACTIONS
Frequent and Serious:
Sodium and fluid retention is most common in young infants and in adults and may precipitate congestive heart failure in patients with compromised cardiac reserve. It usually responds to diuretic therapy (See DRUG INTERACTIONS).
Infrequent but Serious:
Diabetic ketoacidosis and hyperosmolar nonketotic coma may develop very rapidly. Conventional therapy with insulin and restoration of fluid and electrolyte balance is usually effective if instituted promptly. Prolonged surveillance is essential in view of the long half-life of diazoxide oral suspension (See OVERDOSAGE).
Other frequent adverse reactions:
Hirsutism of the lanugo type, mainly on the forehead, back and limbs, occurs most commonly in children and women and may be cosmetically unacceptable. It subsides on discontinuation of the drug.
Hyperglycemia or glycosuria may require reduction in dosage in order to avoid progression to ketoacidosis or hyperosmolar coma.
Gastrointestinal intolerance may include anorexia, nausea, vomiting, abdominal pain, ileus, diarrhea, transient loss of taste.
Tachycardia, palpitations, increased levels of serum uric acid are common.
Thrombocytopenia with or without purpura may require discontinuation of the drug. Neutropenia is transient, is not associated with increased susceptibility to infection, and ordinarily does not require discontinuation of the drug. Skin rash, headache, weakness, and malaise may also occur.
Other adverse reactions which have been observed are:
Cardiovascular: Hypotension occurs occasionally, which may be augmented by thiazide diuretics given concurrently. A few cases of transient hypertension, for which no explanation is apparent, have been noted. Chest pain has been reported rarely. Pulmonary hypertension has been reported in neonates and young infants (see WARNINGS).
There have been postmarketing reports of pericardial effusion in patients without structural heart disease; the majority of cases occurred in pediatric patients and infants.
Gastrointestinal: There have been postmarketing reports of necrotizing enterocolitis; the majority of cases occurred in infants with underlying co-morbid conditions.
Hematologic: eosinophilia; decreased hemoglobin / hematocrit; excessive bleeding, decreased IgG.
Hepato-renal: increased AST, alkaline phosphatase; azotemia, decreased creatinine clearance, reversible nephrotic syndrome, decreased urinary output, hematuria, albuminuria. Neurologic: anxiety, dizziness, insomnia, polyneuritis, paresthesia, pruritus, extrapyramidal signs.
Ophthalmologic: transient cataracts, subconjunctival hemorrhage, ring scotoma, blurred vision, diplopia, lacrimation. Skeletal, integumentary; monilial dermatitis, herpes, advance in bone age; loss of scalp hair. Systemic: fever, lymphadenopathy. Other; gout acute pancreatitis/pancreatic necrosis, galactorrhea, enlargement of lump in breast.
-
OVERDOSAGE
An overdosage of diazoxide oral suspension causes marked hyperglycemia which may be associated with ketoacidosis. It will respond to prompt insulin administration and restoration of fluid and electrolyte balance. Because of the drug’s long half-life (approximately 30 hours), the symptoms of overdosage require prolonged surveillance for periods up to seven days until the blood sugar level stabilizes within the normal range. One investigator reported successful lowering of diazoxide blood levels by peritoneal dialysis in one patient and by hemodialysis in another.
-
DOSAGE & ADMINISTRATION
Patients should be under close clinical observation when treatment with diazoxide oral suspension is initiated. The clinical response and blood glucose level should be carefully monitored until the patient’s condition has stabilized satisfactory; in most instances, this may be accomplished in several days. If administration of diazoxide oral suspension is not effective after two or three weeks, the drug should be discontinued.
The dosage of diazoxide oral suspension must be individualized based on the severity of the hypoglycemic condition and the blood glucose level and clinical response of the patient. The dosage should be adjusted until the desired clinical and laboratory effects are produced with the least amount of the drug. Special care should be taken to assure accuracy of dosage in infants and young children.
Adults and children:
The usual daily dosage is 3 to 8 mg/kg, divided into two or three equal doses every 8 or 12 hours. In certain instances, patients with refractory hypoglycemia may require higher dosages. Ordinarily, an appropriate starting dosage is 3 mg/kg/day, divided into three equal doses every 8 hours. Thus, an average adult would receive a starting dosage of approximately 200 mg daily.
Infants and newborns:
The usual daily dosage is 8 to 15 mg/kg divided into two or three equal doses every 8 to 12 hours. An appropriate starting dosage is 10 mg/kg/day, divided into three equal doses every 8 hours.
-
ANIMAL PHARMACOLOGY & OR TOXICOLOGY SECTION
Oral diazoxide in the mouse, rat, rabbit, dog, pig, and monkey produces a rapid and transient rise in blood glucose levels. In dogs, increased blood glucose is accompanied by increased free fatty acids, lactate, and pyruvate in the serum. In mice, a marked decrease in liver glycogen and an increase in the blood urea nitrogen level occur.
In acute toxicity studies the LD50 for oral diazoxide suspension is >5000 mg/kg in the rat, >522 mg/kg in the neonatal rat, between 1900 and 2572 mg/kg in the mouse, and 219 mg/kg in the guinea pig. Although the oral LD50 was not determined in the dog, a dosage of up to 500 mg/kg was well tolerated.
In subacute oral toxicity studies, diazoxide at 400 mg/kg in the rat produced growth retardation, edema, increases in liver and kidney weights, and adrenal hypertrophy. Daily dosages up to 1080 mg/kg for three months produced hyperglycemia, an increase in liver weight and an increase in mortality. In dogs given oral diazoxide at approximately 40 mg/kg/day for one month, no biologically significant gross or microscopic abnormalities were observed. Cataracts, attributed to markedly disturbed carbohydrate metabolism, have been observed in a few dogs given repeated daily doses of oral or intravenous diazoxide. The lenticular changes resembled those which occur experimentally in animals with increased blood glucose levels. In chronic toxicity studies, rats given a daily dose of 200 mg/kg diazoxide for 52 weeks had a decrease in weight gain and an increase in heart, liver, adrenal and thyroid weights. Mortality in drug-treated and control groups was not different. Dogs treated with diazoxide at dosages of 50, 100, and 200 mg/kg/day for 82 weeks had higher blood glucose levels than controls. Mild bone marrow stimulation and increased pancreas weights were evident in the drug-treated dogs; several developed inguinal hernias, one had a testicular seminoma, and another had a mass near the penis. Two females had inguinal mammary swellings. The etiology of these changes was not established. There was no difference in mortality between drug-treated and control groups. In a second chronic oral toxicity study, dogs given milled diazoxide at 50, 100, and 200 mg/kg/day had anorexia and severe weight loss, causing death in a few. Hematologic, biochemical, and histologic examination did not indicate any cause of death other than inanition. After one year of treatment, there is no evidence of herniation or tissue swelling in any of the dogs.
When diazoxide was administered at high dosages concomitantly with either chlorothiazide to rats or trichlormethiazide to dogs, increased toxicity was observed. In rats, the combination was nephrotoxic; epithelial hyperplasia was observed in the collecting tubules. In dogs, a diabetic syndrome was produced which resulted in ketosis and death. Neither of the drugs given alone produced these effects.
Although the data are inconclusive, reproduction and teratology studies in several species of animals indicate that diazoxide, when administered during the critical period of embryo formation, may interfere with normal fetal development, possibly through altered glucose metabolism. Parturition was occasionally prolonged in animals treated at term. Intravenous administration of diazoxide to pregnant sheep, goats, and swine produced in the fetus an appreciable increase in blood glucose level and degeneration of the beta cells of the Islets of Langerhans. The reversibility of these effects was not studied.
-
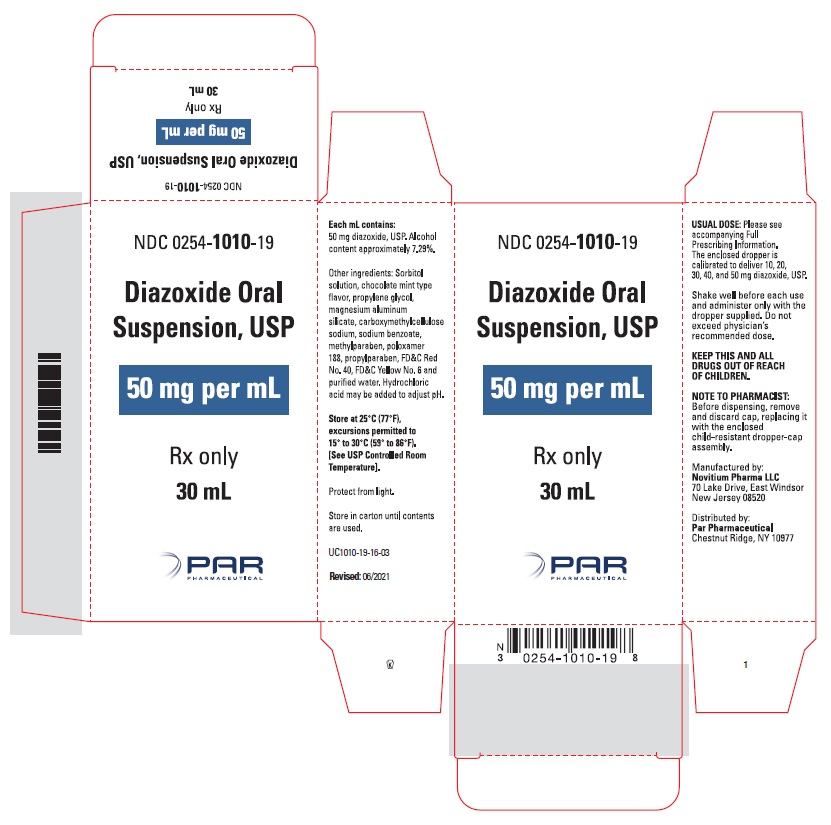
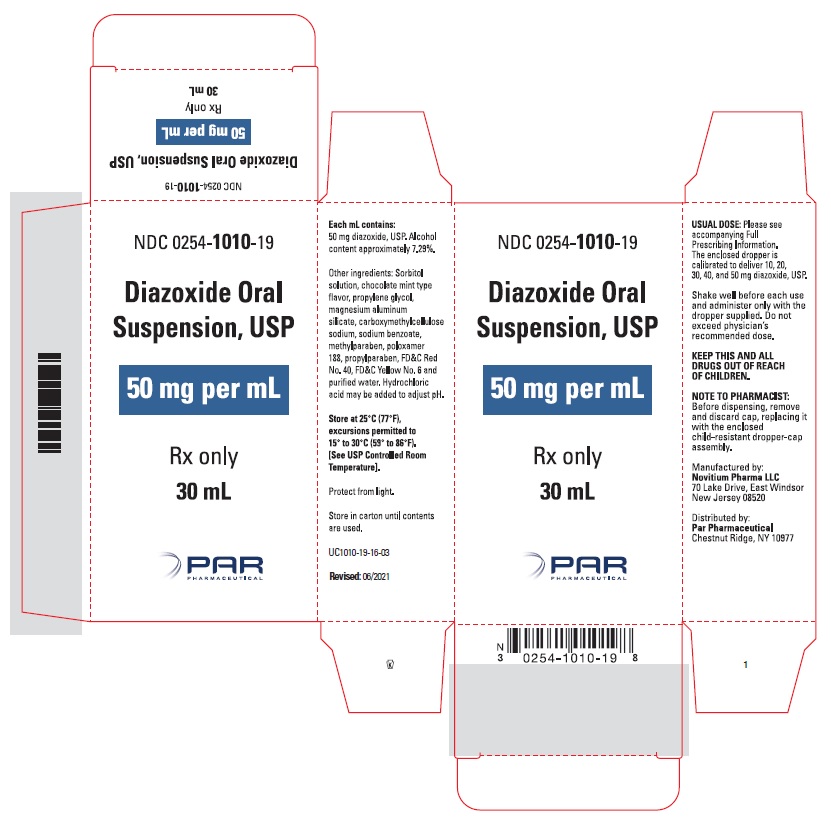
HOW SUPPLIED
Diazoxide Oral Suspension, USP 50 mg/mL, a white to light brown colored, chocolate-mint flavored suspension; bottle of 30 mL (NDC 0254-1010-19), with dropper calibrated to deliver 10, 20, 30, 40 and 50 mg diazoxide, USP.
Shake well before each use. Protect from light. Store in carton until contents are used. Store in light resistant container as defined in the USP. Store Diazoxide Oral Suspension, USP at 25°C (77°F), excursions permitted to 15°-30°C (59-86°F). [See USP Controlled Room Temperature].
Rx only
Manufactured by:
Novitium Pharma LLC
70 Lake Drive, East Windsor
New Jersey 08520
Distributed by:
Par Pharmaceutical
Chestnut Ridge, NY 10977
Revised: 09/2023
OS1010-01-16-04
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DIAZOXIDE
diazoxide suspensionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0254-1010 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIAZOXIDE (UNII: O5CB12L4FN) (DIAZOXIDE - UNII:O5CB12L4FN) DIAZOXIDE 50 mg in 1 mL Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) SODIUM BENZOATE (UNII: OJ245FE5EU) METHYLPARABEN (UNII: A2I8C7HI9T) POLOXAMER 188 (UNII: LQA7B6G8JG) PROPYLPARABEN (UNII: Z8IX2SC1OH) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) HYDROCHLORIC ACID (UNII: QTT17582CB) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) Product Characteristics Color Score Shape Size Flavor CHOCOLATE (Chocolate-mint) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0254-1010-19 1 in 1 CARTON 07/21/2020 1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210799 07/21/2020 Labeler - Par Pharmaceutical, Inc (092733690)