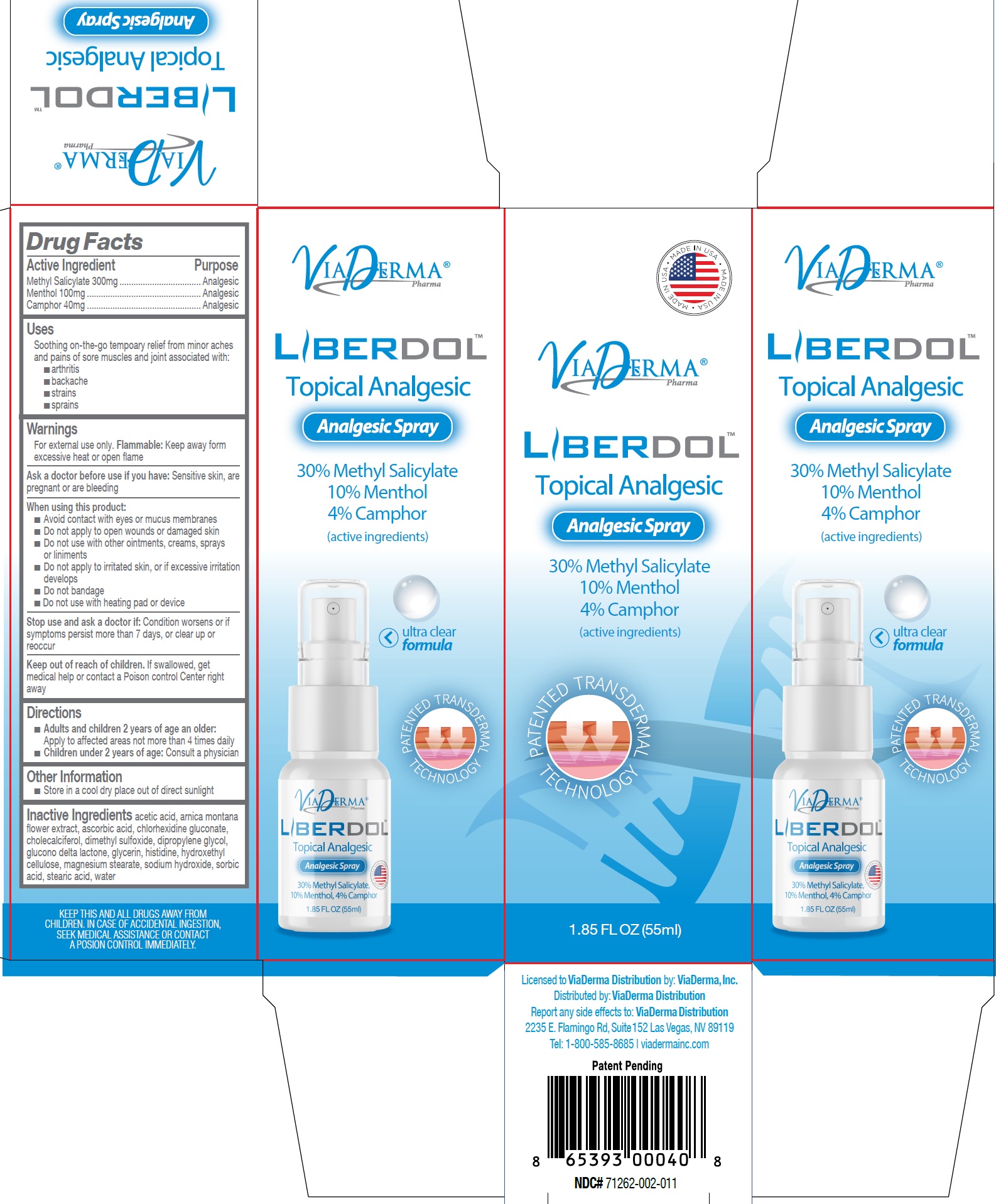
Label: LIBERDOL TOPICAL ANALGESIC (methyl salicylate, menthol, camphor- synthetic spray
- NDC Code(s): 71262-008-55
- Packager: ViaDerma Distribution, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredient
- Uses
-
Warnings
For external use only. : Keep away form excessive heat or open flame Flammable
When using this product:
- Avoid contact with eyes or mucus membranes
- Do not apply to open wounds or damaged skin
- Do not use with other ointments, creams, sprays or liniments
- Do not apply to irritated skin, or if excessive irritation develops
- Do not bandage
- Do not use with heating pad or device
- Directions
- Other Information
- Inactive Ingredients
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
LIBERDOL TOPICAL ANALGESIC
methyl salicylate, menthol, camphor (synthetic) sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71262-008 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYL SALICYLATE (UNII: LAV5U5022Y) (SALICYLIC ACID - UNII:O414PZ4LPZ) METHYL SALICYLATE 300 mg in 1 mL MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 100 mg in 1 mL CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 40 mg in 1 mL Inactive Ingredients Ingredient Name Strength ACETIC ACID (UNII: Q40Q9N063P) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) ASCORBIC ACID (UNII: PQ6CK8PD0R) CHLORHEXIDINE GLUCONATE (UNII: MOR84MUD8E) CHOLECALCIFEROL (UNII: 1C6V77QF41) DIMETHYL SULFOXIDE (UNII: YOW8V9698H) DIPROPYLENE GLYCOL (UNII: E107L85C40) GLUCONOLACTONE (UNII: WQ29KQ9POT) GLYCERIN (UNII: PDC6A3C0OX) HISTIDINE (UNII: 4QD397987E) HYDROXYETHYL CELLULOSE, UNSPECIFIED (UNII: T4V6TWG28D) MAGNESIUM STEARATE (UNII: 70097M6I30) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBIC ACID (UNII: X045WJ989B) STEARIC ACID (UNII: 4ELV7Z65AP) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71262-008-55 1 in 1 CARTON 06/01/2021 1 55 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 06/01/2021 Labeler - ViaDerma Distribution, Inc (081113521)