Label: CLOBEX- clobetasol propionate spray
- NDC Code(s): 0299-3849-01, 0299-3849-02, 0299-3849-04
- Packager: Galderma Laboratories, L.P.
- Category: HUMAN PRESCRIPTION DRUG LABEL
Drug Label Information
Updated February 2, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use CLOBEX® (clobetasol propionate) Spray, 0.05% safely and effectively. See full prescribing information for CLOBEX® (clobetasol propionate) Spray, 0.05%. CLOBEX spray, for topical use. Initial U.S. Approval: 1985
INDICATIONS AND USAGE
CLOBEX® (clobetasol propionate) Spray, 0.05% is a corticosteroid indicated for the topical treatment of moderate to severe plaque psoriasis affecting up to 20% body surface area (BSA) in patients 18 years of age or older (1.1)
Limitations of Use:DOSAGE AND ADMINISTRATION
- Not for oral, ophthalmic, or intravaginal use. (1.2)
- Sprayed directly onto the affected skin areas twice daily and rub in gently. (2)
- The total dosage should not exceed 50 g (59 mL or 2 fluid ounces) per week. Do not use more than 26 sprays per application or 52 sprays per day. (2)
- Treatment beyond 2 weeks should be limited to localized lesions of moderate to severe plaque psoriasis that have not sufficiently improved after the initial 2 weeks of treatment with CLOBEX® Spray, 0.05%. (2)
- Do not use for more than 4 weeks. (2)
DOSAGE FORMS AND STRENGTHS
Spray, 0.05% w/w (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
- Clobetasol propionate is a highly potent topical corticosteroid that has been shown to suppress the HPA axis at the lowest doses tested. (5.1)
- Cushing’s syndrome, hyperglycemia, and unmasking of latent diabetes mellitus can also result from systemic absorption of topical corticosteroids. (5.1)
- Systemic absorption may require periodic evaluation for HPA axis suppression. Modify use if HPA axis suppression develops. (5.1)
- Children may be more susceptible to systemic toxicity from use of topical corticosteroids. (5.1,8.4)
- CLOBEX Spray may increase the risk of cataract and glaucoma. If visual symptoms occur, consider referral to an ophthalmologist. (5.2)
- Local adverse reactions with topical corticosteroids may occur more frequently with the use of occlusive dressings and higher potency corticosteroids, including clobetasol propionate (5.3)
- CLOBEX® Spray is flammable, keep away from heat or flame. (5.6)
ADVERSE REACTIONS
In controlled, clinical trials with CLOBEX® Spray, 0.05%, the most common adverse reactions (incidence > 2%) were burning, pruritus, nasopharyngitis, upper respiratory tract infection. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Galderma Laboratories, L.P. at 1-866-735-4137 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 2/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Indication
1.2 Limitations of Use
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Effects on the Endocrine System
5.2 Ophthalmic Adverse Reactions
5.3 Local Adverse Reactions with Topical Corticosteroids.
5.4 Allergic Contact Dermatitis
5.5 Concomitant Skin Infections
5.6 Flammable Contents
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Pediatric Use
8.4 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
17.1 Information for Patients
17.2 Instructions to the Pharmacist
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Indication
CLOBEX® Spray, 0.05% is a super-high potent topical corticosteroid formulation indicated for the treatment of moderate to severe plaque psoriasis affecting up to 20% body surface area (BSA) in patients 18 years of age or older.
Patients should be instructed to use CLOBEX® Spray, 0.05% for the minimum amount of time necessary to achieve the desired results see [Dosage and Administration (2)].
Use in patients under 18 years of age is not recommended because safety has not been established and because numerically high rates of HPA axis suppression were seen with other clobetasol propionate topical formulations. [see Use in Specific Populations (8.4)].
-
2 DOSAGE AND ADMINISTRATION
CLOBEX® Spray, 0.05% is for topical use only, and not for ophthalmic, oral or intravaginal use.
CLOBEX® Spray, 0.05% should be sprayed directly onto the affected skin areas twice daily and rubbed in gently and completely.
The total dosage should not exceed 50 g (59 mL or 2 fluid ounces) per week because of the potential for the drug to suppress the hypothalamic-pituitary-adrenal (HPA) axis. Do not use more than 26 sprays per application or 52 sprays per day.
CLOBEX® Spray, 0.05% contains a topical corticosteroid; therefore treatment should be limited to 4 weeks. Therapy should be discontinued when control has been achieved. Treatment beyond 2 weeks should be limited to localized lesions of moderate to severe plaque psoriasis that have not sufficiently improved after the initial 2 weeks of treatment with CLOBEX® Spray, 0.05%. If no improvement is seen within 2 weeks, reassessment of diagnosis may be necessary. Before prescribing for more than 2 weeks, any additional benefits of extending treatment to 4 weeks should be weighed against the risk of HPA axis suppression.
Unless directed by physician, CLOBEX® Spray, 0.05% should not be used with occlusive dressings.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Effects on the Endocrine System
Clobetasol propionate is a highly potent topical corticosteroid that has been shown to suppress the HPA axis at the lowest doses tested.
Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal (HPA) axis suppression with the potential for clinical glucocorticosteroid insufficiency. This may occur during treatment or upon withdrawal of the topical corticosteroid.
In studies evaluating the potential for hypothalamic-pituitary-adrenal (HPA) axis suppression, using the Cosyntropin Stimulation Test, CLOBEX® Spray, 0.05% demonstrated rates of suppression that were comparable after 2 and 4 weeks of twice-daily use (19% and 15-20%, respectively), in adult patients with moderate to severe plaque psoriasis (≥ 20% BSA). In these studies, HPA axis suppression was defined as serum cortisol level ≤18 μg/dL 30-min post cosyntropin stimulation [see Clinical Pharmacology (12)].
Because of the potential for systemic absorption, use of topical corticosteroids may require that patients be periodically evaluated for HPA axis suppression. Factors that predispose a patient using a topical corticosteroid to HPA axis suppression include the use of more potent steroids, use over large surface areas, use over prolonged periods, use under occlusion, use on an altered skin barrier, and use in patients with liver failure.
An ACTH stimulation test may be helpful in evaluating patients for HPA axis suppression. If HPA axis suppression is documented, an attempt should be made to gradually withdraw the drug, to reduce the frequency of application, or to substitute a less potent steroid. Manifestations of adrenal insufficiency may require supplemental systemic corticosteroids. Recovery of HPA axis function is generally prompt and complete upon discontinuation of topical corticosteroids.
Cushing’s syndrome, hyperglycemia, and unmasking of latent diabetes mellitus can also result from systemic absorption of topical corticosteroids.
Use of more than one corticosteroid-containing product at the same time may increase the total systemic corticosteroid exposure.
Pediatric patients may be more susceptible to systemic toxicity from use of topical corticosteroids [see Use in Specific Populations (8.4)].
5.2 Ophthalmic Adverse Reactions
Use of topical corticosteroids, including CLOBEX Spray, may increase the risks of glaucoma and posterior subcapsular cataract. Glaucoma and cataracts have been reported in postmarketing experience with the use of topical corticosteroid products, including topical clobetasol products [see Adverse Reactions (6.2)].
Avoid contact of CLOBEX Spray with eyes. Advise patients to report any visual symptoms and consider referral to an ophthalmologist for evaluation.
5.3 Local Adverse Reactions with Topical Corticosteroids.
The following additional local adverse reactions have been reported with topical corticosteroids. They may occur more frequently with the use of occlusive dressings and higher potency corticosteroids, including clobetasol propionate. These reactions are listed in an approximate decreasing order of occurrence: folliculitis, acneiform eruptions, hypopigmentation, perioral dermatitis, allergic contact dermatitis, secondary infection, striae and miliaria.
5.4 Allergic Contact Dermatitis
Allergic contact dermatitis to any component of topical corticosteroids is usually diagnosed by a failure to heal rather than a clinical exacerbation. Clinical diagnosis of allergic contact dermatitis can be confirmed by patch testing.
5.5 Concomitant Skin Infections
In the presence of dermatological infections, the use of an appropriate antifungal or antibacterial agent should be instituted. If a favorable response does not occur promptly, use of CLOBEX® Spray, 0.05% should be discontinued until the infection has been adequately controlled.
-
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
In controlled, clinical trials with CLOBEX® Spray, 0.05%, the most common adverse reaction was burning at the site of application [40% of subjects treated with CLOBEX® Spray, 0.05% and 47% of subjects treated with Spray Vehicle]. Other commonly reported adverse reactions for CLOBEX® Spray, 0.05% and Spray Vehicle, respectively, are noted in Table 1.
Table 1 - Commonly Occurring Adverse Reactions (≥1% Incidence) Clobetasol Propionate Vehicle Spray Adverse Reaction 0.05% Spray (N=120) (N=120) System Organ Class General disorders and administration site conditions 50 (42%) 56 (47%) Application site burning 48 (40%) 56 (47%) Application site dryness 2 (2%) 0 (0%) Application site irritation 1 (1%) 0 (0%) Application site pain 1 (1%) 2 (2%) Application site pigmentation changes 1 (1%) 0 (0%) Application site pruritus 4 (3%) 3 (3%) Infections and infestations 17 (14%) 12 (10%) Nasopharyngitis 6 (5%) 3 (3%) Pharyngitis streptococcal 1 (1%) 0 (0%) Upper respiratory tract infection 10 (8%) 2 (2%) Skin and subcutaneous tissue disorders 4 (3%) 2 (2%) Eczema asteatotic 2 (2%) 0 (0%) Most local adverse reactions were rated as mild to moderate and they are not affected by age, race or gender.
Systemic absorption of topical corticosteroids has produced hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome, hyperglycemia, and glucosuria in some patients.
6.2 Postmarketing Experience
Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been identified during post-approval use of CLOBEX® Spray, 0.05%.
Skin: Burning, pruritus, erythema, pain, irritation, rash, peeling, urticaria, and contact dermatitis.
Ophthalmic adverse reactions of blurred vision, cataracts, glaucoma, increased intraocular pressure, and central serous chorioretinopathy have been reported with the use of topical corticosteroids.
-
8 USE IN SPECIFIC POPULATIONS
There are no available data on CLOBEX® Spray use in pregnant women to identify a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
Observational studies suggest an increased risk of low birthweight in infants with the maternal use of potent or very potent topical corticosteroids (see Data). Advise pregnant women that CLOBEX® Spray may increase the risk of having a low birth weight infant and to use CLOBEX® Spray on the smallest area of skin and for the shortest duration possible.
Animal reproduction studies have not been conducted with CLOBEX® Spray. In an animal reproduction study, subcutaneous administration of clobetasol propionate to pregnant rats at doses greater than 12.5 μg/kg/day during the period of organogenesis caused an increase in malformations (increased incidence of umbilical hernia) [see Data]. The available data do not allow calculation of relevant comparisons between the systemic exposure of clobetasol propionate in animal studies to the systemic exposure that would be expected in humans after topical use of CLOBEX® Spray.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data
Human Data
Available observational studies in pregnant women did not identify a drug-associated risk of major birth defects, preterm delivery, or fetal mortality with the use of topical corticosteroids of any potency. However, when the dispensed amount of potent or very potent topical corticosteroids exceeded 300 g during the entire pregnancy, maternal use was associated with an increased risk of low birth weight in infants.
Animal Data
Clobetasol propionate is absorbed percutaneously, and when administered subcutaneously it caused malformations in both the rabbit and the mouse.Clobetasol propionate has greater potential for adverse developmental effects than steroids that are less potent.
The effect of clobetasol propionate on pregnancy outcome and development of offspring was studied in the rat. Clobetasol propionate was administered subcutaneously to female rats twice daily (0, 12.5, 25, and 50 μg/kg/day) from day 7 of presumed gestation through day 25 of lactation or day 24 presumed gestation for those rats that did not deliver a litter. The maternal no-observed-adverse-effect-level (NOAEL) for clobetasol propionate was less than 12.5 μg/kg/day due to reduced body weight gain and feed consumption during the gestation period. The reproductive NOAEL in the dams was 25 μg/kg/day based on prolonged delivery at 50 μg/kg/day. The NOAEL for viability and growth in the offspring was 12.5 μg/kg/day based on incidence of stillbirths, reductions in pup body weights on days 1 and 7 of lactation, increased pup mortality, increases in the incidence of umbilical hernia, and increases in the incidence of pups with cysts on the kidney at higher dose levels during the preweaning period. The weights of the epididymides and testes were significantly reduced at higher dosages. Despite these changes, there were no effects on the mating and fertility of the offspring.
8.2 Lactation
Risk Summary
There is no information regarding the presence of clobetasol propionate in human milk or its effects on the breastfed infant or on milk production. It is not known whether topical administration of clobetasol propionate could result in sufficient systemic absorption to produce detectable quantities in human milk. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CLOBEX® Spray and any potential adverse effects on the breastfed infant from CLOBEX® Spray or from the underlying maternal condition.
Clinical Considerations
To minimize potential exposure to the breastfed infant via breast milk, use CLOBEX® Spray on the smallest area of skin and for the shortest duration possible while breastfeeding. Advise breastfeeding women not to apply CLOBEX® Spray directly to the nipple and areola to avoid direct infant exposure [see Use in Specific Populations (8.4)].
8.3 Pediatric Use
Use in patients under 18 years of age is not recommended, because safety has not been established and because numerically high rates of HPA axis suppression were seen with other clobetasol propionate topical formulations. Safety and effectiveness in pediatric patients treated with CLOBEX® Spray, 0.05% have not been established [see Warnings and Precautions (5.1)].
Because of higher ratio of skin surface area to body mass, pediatric patients are at a greater risk than adults of HPA axis suppression and Cushing’s syndrome when they are treated with topical corticosteroids. They are therefore also at greater risk of glucocorticosteroid insufficiency during and/or after withdrawal of treatment.
Adverse effects including striae have been reported with inappropriate use of topical corticosteroids in infants and children.HPA axis suppression, Cushing’s syndrome, linear growth retardation, delayed weight gain, and intracranial hypertension have been reported in children receiving topical corticosteroids. Manifestations of adrenal suppression in children include low plasma cortisol levels and absence of response to ACTH stimulation. Manifestations of intracranial hypertension include bulging fontanelles, headaches, and bilateral papilledema.
8.4 Geriatric Use
Clinical studies of CLOBEX® Spray, 0.05% did not include sufficient numbers of patients aged 65 and over to adequately determine whether they respond differently than younger patients. In two randomized, vehicle controlled clinical trials, 21 of the 240 patients (9%) were over the age of 65. In general, dose selection for an elderly patient should be made with caution, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
-
10 OVERDOSAGE
Topically applied CLOBEX® Spray, 0.05% can be absorbed in sufficient amount to produce systemic effects [see Warnings and Precautions (5.1)].
-
11 DESCRIPTION
CLOBEX® (clobetasol propionate) Spray, 0.05% contains clobetasol propionate, a synthetic fluorinated corticosteroid, for topical use. The corticosteroids constitute a class of primarily synthetic steroids used topically as anti-inflammatory and antipruritic agents. Clobetasol propionate is 21-chloro-9-fluoro-11β, 17-dihydroxy-16β-methylpregna-1,4-diene-3,20-dione 17-propionate, with the empirical formula C25H32CIFO5, and a molecular weight of 466.97 (CAS Registry Number 25122-46-7).
The following is the chemical structure:
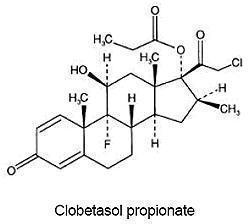
Clobetasol propionate is a white to almost white crystalline powder that is practically insoluble in water. Each gram of CLOBEX® Spray, 0.05% contains 0.5 mg of clobetasol propionate, in a clear, colorless liquid composed of alcohol, isopropyl myristate, sodium lauryl sulfate, and undecylenic acid.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corticosteroids play a role in cellular signaling, immune function, inflammation, and protein regulation; however, the precise mechanism of action in corticosteroid responsive dermatoses is unknown.
12.2 Pharmacodynamics
Vasoconstrictor Assay
CLOBEX® Spray, 0.05% is in the super-high range of potency as demonstrated in a vasoconstrictor study in healthy subjects when compared with other topical corticosteroids. However, similar blanching scores do not necessarily imply therapeutic equivalence.
Hypothalamic-Pituitary-Adrenal (HPA) Axis Suppression
The effect of CLOBEX® Spray, 0.05% on hypothalamic-pituitary-adrenal (HPA) axis function was investigated in adults in two studies. In the first study, patients with plaque psoriasis covering at least 20% of their body applied CLOBEX® Spray, 0.05% twice daily for up to 4 weeks. 15% (2 out of 13) of patients displayed adrenal suppression after 4 weeks of use based on the Cosyntropin Stimulation Test. The laboratory suppression was transient; all subjects returned to normal after cessation of drug use. In the second study, patients with plaque psoriasis covering at least 20% of their body applied CLOBEX® Spray, 0.05% twice daily for either 2 or 4 weeks. 19% (4 out of 21) of patients treated for 2 weeks and 20% (3 out of 15) of patients treated for 4 weeks displayed adrenal suppression at the end of treatment based on the Cosyntropin Stimulation Test. The laboratory suppression was transient; all subjects returned to normal after cessation of drug use. In these studies, HPA axis suppression was defined as serum cortisol level ≤ 18 μg/dL 30-min post cosyntropin (ACTH 1-24) stimulation [see Warnings and Precautions (5)].
12.3 Pharmacokinetics
The extent of percutaneous absorption of topical corticosteroids is determined by many factors, including the vehicle, the integrity of the epidermal barrier and occlusion.
Topical corticosteroids can be absorbed from normal intact skin. Inflammation and other disease processes in the skin may increase percutaneous absorption.
There are no human data regarding the distribution of corticosteroids to body organs following topical application. Nevertheless, once absorbed through the skin, topical corticosteroids are handled through metabolic pathways similar to systemically administered corticosteroids. They are metabolized, primarily in the liver, and are then excreted by the kidneys. In addition, some corticosteroids and their metabolites are also excreted in the bile.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Clobetasol propionate was not carcinogenic to rats when topically applied for 2 years at concentrations up to 0.005% which corresponded to doses up to 11 μg/kg/day.
Clobetasol propionate was negative in the in vitro mammalian chromosomal aberration test and in the in vivo mammalian erythrocyte micronucleus test
The effect of subcutaneously administered clobetasol propionate on fertility and general reproductive toxicity was studied in rats at doses of 0, 12.5, 25, and 50 μg/kg/day. Males were treated beginning 70 days before mating and females beginning 15 days before mating through day 7 of gestation. A dosage level of less than 12.5 μg/kg/day clobetasol propionate was considered to be the NOAEL for paternal and maternal general toxicity based on decreased weight gain and for male reproductive toxicity based on increased weights of fluid-filled seminal vesicles. The female reproductive NOAEL was 12.5 μg/kg/day based on reduction in the numbers of estrous cycles during the pre-cohabitation period and an increase in the number of nonviable embryos at higher doses. -
14 CLINICAL STUDIES
The efficacy of CLOBEX® Spray, 0.05% in psoriasis has been demonstrated in two randomized, vehicle controlled clinical trials, which were identical in design. The studies were conducted in patients aged 18 years and older with moderate to severe plaque psoriasis. Patients were treated twice daily for up to 4 weeks with either CLOBEX® Spray, 0.05% or vehicle spray.
Patients were evaluated on their Overall Disease Severity, a 5-point scale based on scaling, erythema, and plaque elevation that classified subjects as clear, almost clear, mild, moderate, or severe/very severe. Only patients classified as moderate or severe/very severe at baseline were enrolled in the studies. The median percent body surface area (BSA) at baseline was 6% for the two studies. The numbers of patients scored as clear or almost clear at Weeks 2 and 4 are presented in Table 2.
Table 2: Number of Patients Clear or Almost Clear on Overall Disease Severity Sale at Weeks 2 & 4 Study 1 Study 2 Clobex®
N=60Vehicle
N=60Clobex®
N=60Vehicle
N=60Week 2 Clear
Almost Clear1 (2%)
32 (53%)0 (0%)
1 (2%)0 (0%)
28 (47%)0 (0%)
0 (0%)Week 4 Clear
Almost Clear15 (25%)
32 (53%)0 (0%)
2 (3%)18 (30%)
31 (52%)0 (0%)
1 (2%) -
16 HOW SUPPLIED/STORAGE AND HANDLING
CLOBEX® Spray, 0.05% is a clear, colorless liquid, supplied in a white HDPE bottle with a white polypropylene cap and white LDPE liner in the following sizes:
2 fl oz/59 mL NDC 0299-3849-02
4.25 fl oz/125 mL NDC 0299-3849-04
Storage: Keep tightly closed. Store under controlled room temperature conditions 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C and 30°C (59°F and 86°F). Do not freeze, refrigerate or store above 30°C. Spray is flammable; avoid heat, flame or smoking when using this product.
-
17 PATIENT COUNSELING INFORMATION
[See FDA-approved patient labeling (Patient Information)]
17.1 Information for Patients
Patients using topical corticosteroids should receive the following information and instructions:
- This medication is to be used as directed by the physician and should not be used longer than the prescribed time period.
- This medication should not be used for any disorder other than that for which it was prescribed.
- Do not use other corticosteroid-containing products while using CLOBEX® Spray, 0.05% unless directed by your physician.
- The treated skin area should not be bandaged, otherwise covered, or wrapped so as to be occlusive unless directed by the physician.
- Patients should wash their hands after applying the medication.
- Advise patients to report any visual symptoms to their healthcare providers.
- Patients should report any signs of local or systemic adverse reactions to the physician.
- Patients should inform their physicians that they are using CLOBEX® Spray, 0.05% if surgery is contemplated.
- If you go to another doctor for illness, injury or surgery, tell that doctor you are using CLOBEX® Spray, 0.05%.
- This medication is for external use only. It should not be used on the face, underarms, or groin area. Also avoid contact with the eyes and lips.
- As with other corticosteroids, therapy should be discontinued when control is achieved. If no improvement is seen within 2 weeks, contact the physician.
- Advise a woman to use CLOBEX Spray on the smallest area of skin and for the shortest duration possible while pregnant or breastfeeding. Advise breastfeeding women not to apply CLOBEX Spray directly to the nipple and areola to avoid direct infant exposure
- Patients should not use more than 50 g (59 mL or 2 fl.oz.) per week of CLOBEX® Spray, 0.05%.
- Do not use more than 26 sprays per application or 52 sprays per day.
- This medication is flammable; avoid heat, flame or smoking when applying this product.
-
PATIENT INFORMATION CLOBEX® (KLO-bex) (clobetasol propionate) Spray
Important: For use on skin only. Do not get CLOBEX® Spray near or in your mouth, eyes or vagina. Read the Patient Information that comes with CLOBEX® Spray before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment.
What is CLOBEX® Spray?
CLOBEX® Spray is a prescription corticosteroid medicine used to treat adults with moderate to severe plaque psoriasis that affects up to 20% of the body’s skin surface. CLOBEX® Spray is for use on the skin only (topical).
- CLOBEX® Spray should only be used for the shortest amount of time needed to treat your plaque psoriasis.
- Do not use more than 26 sprays for each application or more than 52 sprays in 1 day.
- You should not apply more than 59 mL (2 fluid ounces) of CLOBEX® Spray to your skin in 1 week.
You should not use CLOBEX® Spray:
• on your face, under arms (armpits), or groin areas
• if you have thinning of the skin (atrophy) at the treatment site
• to treat rosacea or a rash around the mouth (perioral dermatitis)
It is not known if CLOBEX® Spray is safe and effective in children under 18 years of age. CLOBEX® Spray should not be used in children under 18 years of age.What should I tell my doctor before using CLOBEX® Spray?
Before you use CLOBEX® Spray, tell your doctor if you:
• have a skin infection. You may need medicine to treat the skin infection before you use CLOBEX® Spray.
• plan to have surgery.
• have any other medical conditions.
• are pregnant or plan to become pregnant. It is not known if CLOBEX® Spray will harm your unborn baby.
• are breast-feeding or plan to breast-feed. It is not known if CLOBEX® Spray passes into your breast milk. Talk to your doctor about the best way to feed your baby if you use CLOBEX® Spray.Tell your doctor about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements. Especially tell your doctor if you take other corticosteroid medicines by mouth or use other products on your skin that contain corticosteroids. Ask your doctor or pharmacist if you are not sure.
Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.How should I use CLOBEX® Spray?
• Use CLOBEX® Spray exactly as your doctor tells you to use it.
• Your doctor should tell you how much CLOBEX® Spray to use and where to apply it.
• CLOBEX® Spray is for use on skin only. Do not get CLOBEX® Spray near or in your eyes, mouth, or vagina.
• You should not use CLOBEX® Spray on your face, underarms (armpits), or groin areas.
• Apply CLOBEX® Spray 2 times each day.
• Apply only enough CLOBEX® Spray to cover the affected skin areas. Rub in gently and completely.
• Wash your hands after using CLOBEX® Spray.
• Throw away any unused CLOBEX® Spray.
• Do not bandage or cover your treated areas unless your doctor tells you to.
• Tell your doctor if your skin condition is not getting better after 2 weeks of using CLOBEX®Spray. Your doctor may tell you to apply CLOBEX® Spray to certain areas of your skin for up to 2 more weeks if needed. You should not use CLOBEX® Spray for more than 4 weeks unless your doctor tells you to. This can increase your risk of serious side effects.
See the "Instructions for Use" at the end of the Patient Information for detailed information on the right way to apply CLOBEX® Spray.
What should I avoid while using CLOBEX®Spray?
• CLOBEX® Spray is flammable. Avoid heat, flames or smoking while applying CLOBEX® Spray to your skin.What are the possible side effects of CLOBEX® Spray?
• Clobex® Spray can pass through your skin. Too much Clobex® Spray passing through your skin can cause your adrenal glands to stop working. Your doctor may do blood tests to check how well your adrenal glands are working.
The most common side effects with CLOBEX® Spray include:
• burning at treated site
• upper respiratory tract infection
• runny nose
• sore throat
• dry, itchy, and reddened skin
If you go to another doctor for illness, injury or surgery, tell that doctor you are using Clobex® Spray. Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of CLOBEX® Spray. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. You may also report side effects to Galderma Laboratories, L.P. at 1-866-735-4137
How should I store CLOBEX®Spray?
• Store CLOBEX® Spray at room temperature between 68°F to 77°F (20°C to 25°C).
• Do not store CLOBEX® Spray above 86°F (30°C).
• Do not freeze or refrigerate CLOBEX® Spray.
• Keep the bottle of CLOBEX® Spray tightly closed.
• Keep away from heat or flame.
Keep CLOBEX® Spray and all medicines out of the reach of children.
General information about CLOBEX® Spray.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use CLOBEX® Spray for a condition for which it was not prescribed. Do not give CLOBEX® Spray to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about CLOBEX® Spray. If you would like more information, talk with your doctor. You can ask your pharmacist or doctor for information about CLOBEX® Spray that is written for health professionals.What are the ingredients of CLOBEX® Spray?
Active ingredient: clobetasol propionate
Inactive ingredients: alcohol, isopropyl myristate, sodium lauryl sulfate, and undecylenic acid.
Read the Instructions for Use that comes with CLOBEX® Spray before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment.
Parts of CLOBEX® Spray (See Figure A)
When you receive CLOBEX® Spray the Directional Spray Nozzle is in the “locked” position with the nozzle pointing downwards. (See Figure B)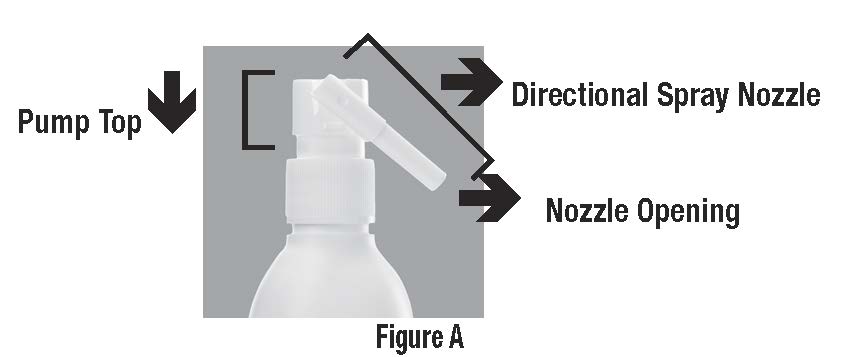

How to apply CLOBEX® Spray:
Step 1: To unlock the Directional Spray Nozzle, hold the CLOBEX® Spray bottle and sides of the Pump Top with one hand. Use your other hand to turn the Directional Spray Nozzle to either the right or the left. (See Figure C) The spray will come out through the opening at the end of the Directional Spray Nozzle.
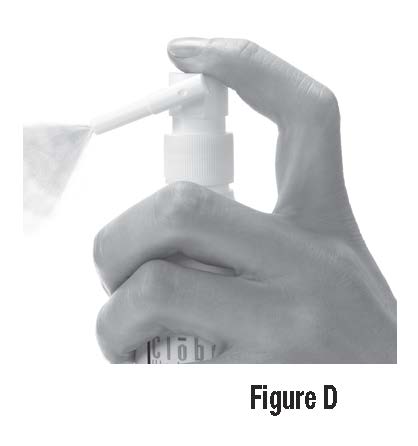
Step 2: To apply CLOBEX® Spray, point the Directional Spray Nozzle to the affected area. To spray, push down on the Pump Top. Apply CLOBEX® Spray to the affected area as instructed by your doctor. (See Figure D)
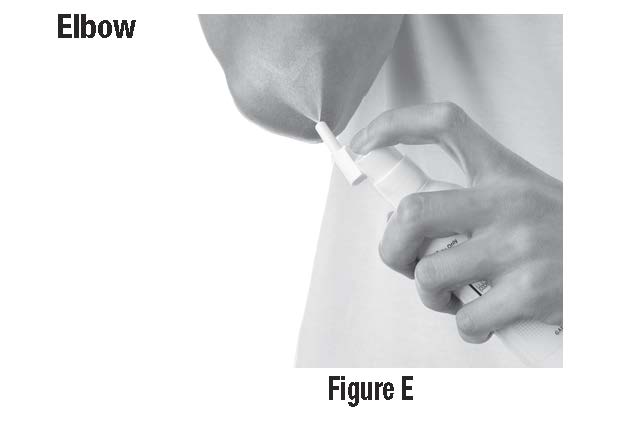
Step 3: Spray only enough CLOBEX® Spray to cover the affected area, for example, the elbow. (SeeFigure E) Rub in CLOBEX® Spray gently and completely.
• You should not apply CLOBEX® Spray to your face, underarms or groin. Avoid contact with eyes.
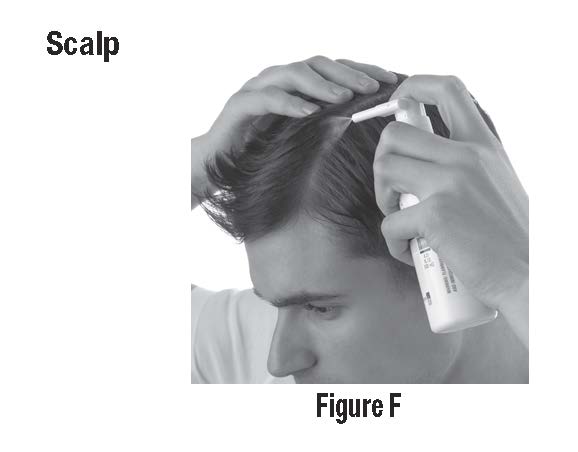
• To apply to your scalp, part your hair at the area where CLOBEX® Spray is to be applied. Spray directly on the affected area, and then rub in gently and completely. (See Figure F)
• Repeat Steps 2 and 3 to apply CLOBEX® Spray to other affected areas as instructed by your doctor
Step 4: After applying CLOBEX® Spray, return the Directional Spray Nozzle to the “locked” position. (See Figure G)

Step 5: Wash your hands after applying CLOBEX® Spray.
This Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration.
Marketed by:
GALDERMA LABORATORIES, L.P., Dallas, TX 75201 USA
Made in Canada
Revised: 02/2023
All trademarks are the property of their respective owners.
P51011-3
-
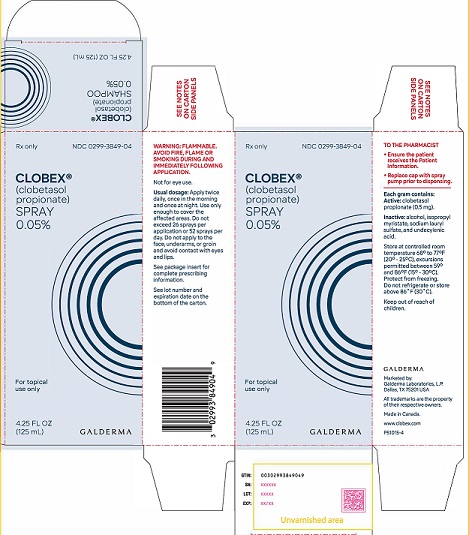
PRINCIPAL DISPLAY PANEL - 4.25 oz CARTON
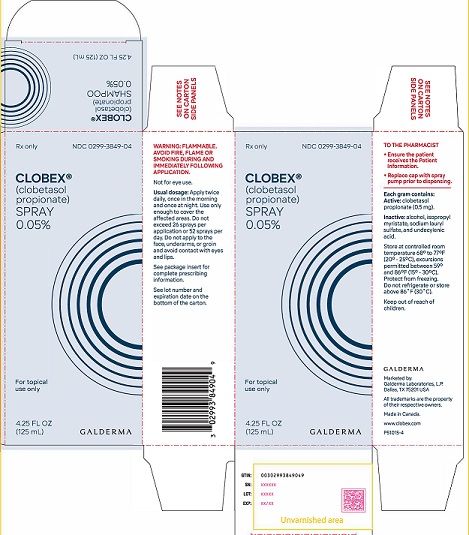
Rx Only
NDC 0299-3849-04
Clobex® (clobetasol propionate) Spray 0.05%
For topical use only
4.25 FL OZ
(125 mL)
GALDERMAWARNING: FLAMMABLE AVOID FIRE, FLAME OR SMOKING DURING AND IMMEDIATELY FOLLOWING APPLICATION
Not for eye use.
Usual dosage: Apply twice daily, once in the morning and once at night.
Use only enough to cover the affected areas. Do not exceed 26 sprays per application or 52 sprays per day. Do not apply to face, underarms, or groin and avoid contact with eyes and lips. See package insert for complete prescribing information. See lot number and expiration date on the bottom of the carton.
TO THE PHARMACIST
● Ensure the patient receives the Patient Information.
● Replace cap with spray pump prior to dispensing.
Each gram contains: Active: clobetasol propionate (0.5 mg).
Inactive: alcohol, isopropyl myristate, sodium lauryl sulfate, and undecylenic acid.
Store at controlled room temperature 68° to 77°F (20° - 25°C), excursions permitted between 59° to 86°F (15° - 30°C).
Protect from freezing.
Do not refrigerate or store above 86° (30°C).
Keep out of reach of children.Marketed by:
GALDERMA LABORATORIES, L.P.
Dallas, TX 75201 USA
All trademarks are the property of their respective owners.
Made in Canada.
www.clobex.com
P51015-4
-
INGREDIENTS AND APPEARANCE
CLOBEX
clobetasol propionate sprayProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:0299-3849 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLOBETASOL PROPIONATE (UNII: 779619577M) (CLOBETASOL - UNII:ADN79D536H) CLOBETASOL PROPIONATE 0.05 g in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) SODIUM LAURYL SULFATE (UNII: 368GB5141J) UNDECYLENIC ACID (UNII: K3D86KJ24N) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0299-3849-02 1 in 1 CARTON 10/27/2005 1 59 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:0299-3849-04 1 in 1 CARTON 10/27/2005 2 125 mL in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:0299-3849-01 1 in 1 CARTON 10/27/2005 3 10 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021835 10/27/2005 Labeler - Galderma Laboratories, L.P. (047350186) Establishment Name Address ID/FEI Business Operations Contract Pharmaceuticals Limited Canada 248761249 manufacture(0299-3849) Establishment Name Address ID/FEI Business Operations G Production, Inc. 251676961 manufacture(0299-3849)