Label: OBAGI MEDICAL BACK TO BASICS (INCLUDING OBAGI MEDICAL PROFESSIONAL-C SERUM 15)- homosalate, octisalate, and zinc oxide kit
- NDC Code(s): 62032-694-02
- Packager: Obagi Cosmeceuticals LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 6, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps prevent sunburn
- If used as directed with other sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
- Warnings
-
Directions
- apply liberally 15 minutes before sun exposure
- use a water resistant sunscreen if swimming or sweating
- reapply at least every 2 hours
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m.–2 p.m.
- wear long-sleeved shirts, pants, hats, and sunglasses
- children under 6 months: Ask a doctor
- Other information
-
Inactive ingredients
Water (Aqua), C15-19 Alkane, Octyldodecyl Neopentanoate, Polymethylsilsesquioxane, Sorbitan Olivate, Silica, Polyglyceryl-6 Polyricinoleate, Sodium Chloride, Xanthan Gum, Glycerin, Hydroxyacetophenone, Disodium EDTA, 1,2-Hexanediol, Caprylyl Glycol, Sodium Hydroxide, Triethoxycaprylylsilane, Polyhydroxystearic Acid, Disteardimonium Hectorite, Polyglyceryl-2 Isostearate, Euphorbia Cerifera (Candelilla) Wax, Beeswax (Cera Alba), Dimethicone
- Questions or comments?
- SPL UNCLASSIFIED SECTION
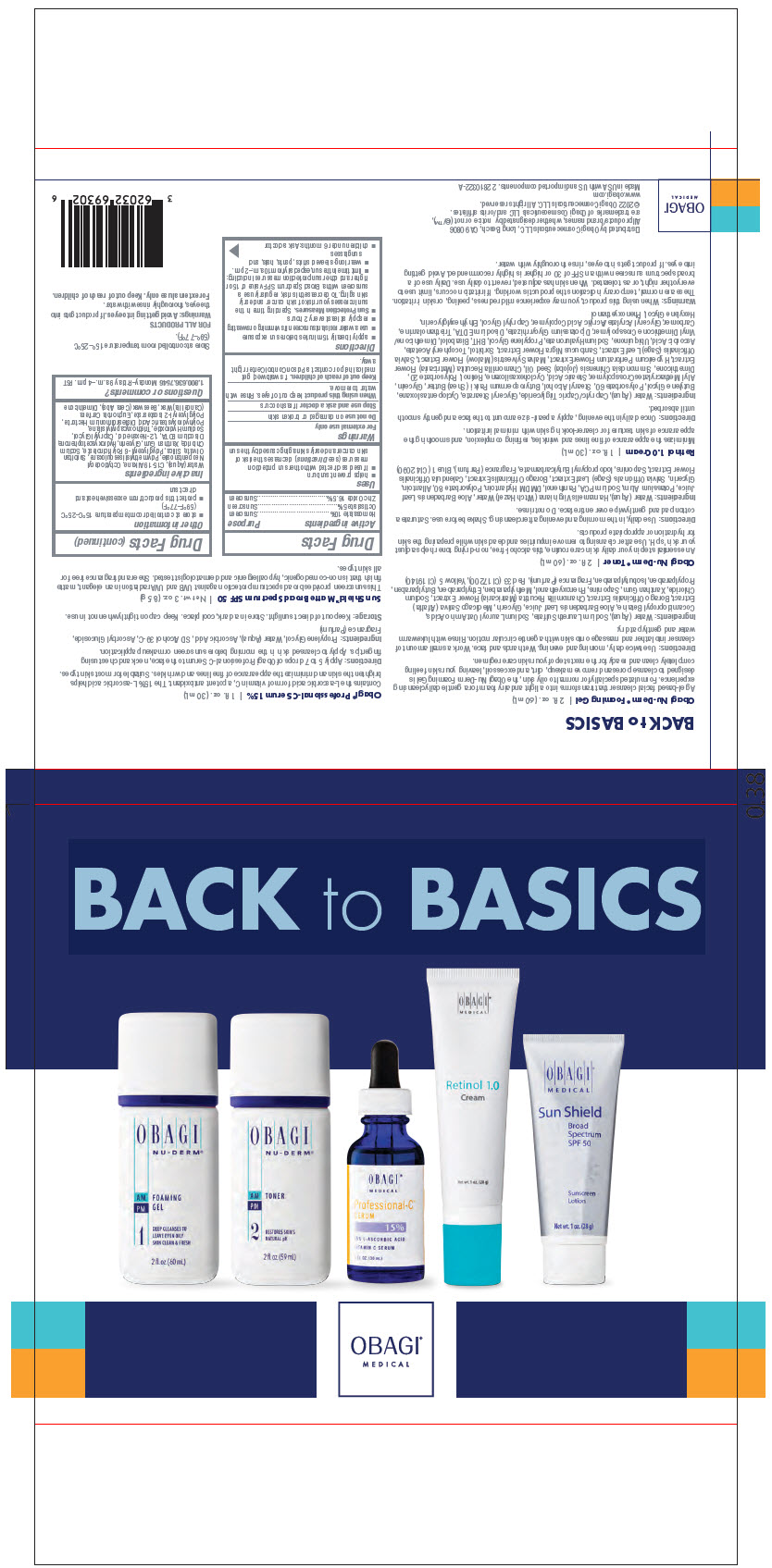
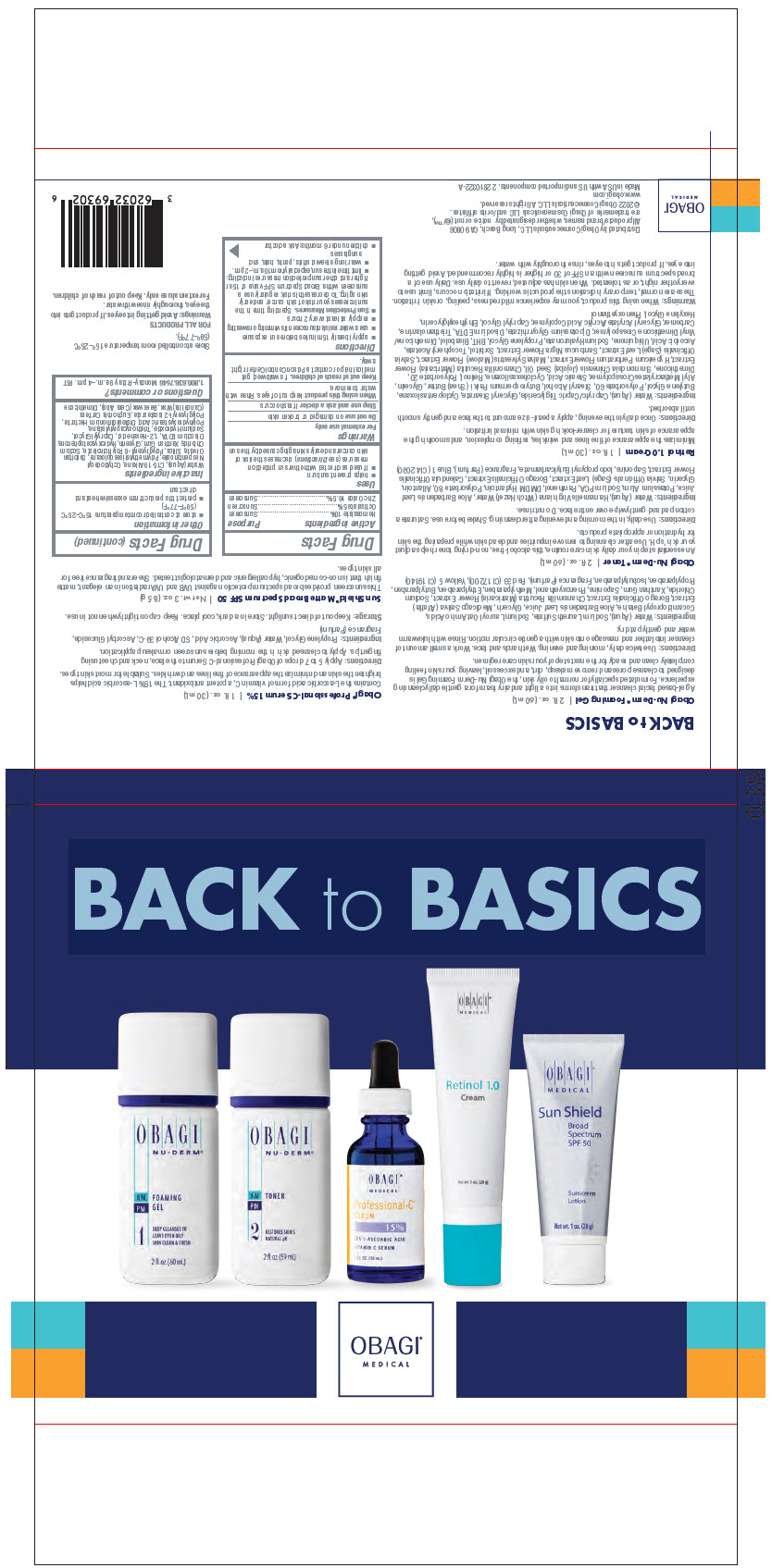
- PRINCIPAL DISPLAY PANEL - Kit Carton
-
INGREDIENTS AND APPEARANCE
OBAGI MEDICAL BACK TO BASICS (INCLUDING OBAGI MEDICAL PROFESSIONAL-C SERUM 15)
homosalate, octisalate, and zinc oxide kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:62032-694 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:62032-694-02 1 in 1 CARTON 04/01/2022 1 1 in 1 BAG Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE, PLASTIC 60 mL Part 2 1 BOTTLE, PLASTIC 59 mL Part 3 1 BOTTLE, DROPPER 30 mL Part 4 1 TUBE 30 mL Part 5 1 TUBE 28 g Part 1 of 5 OBAGI NU-DERM FOAMING
cleansing (cold creams, cleansing lotions, liquids, and pads) gelProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) INGR ACETYL HEXAMETHYL TETRALIN (UNII: 360Q8Z01IP) INGR MEDICAGO SATIVA WHOLE (UNII: DJO934BRBD) INGR ALOE VERA LEAF (UNII: ZY81Z83H0X) INGR BORAGO OFFICINALIS SEED (UNII: 2GXJ790US0) INGR BUTYLPARABEN (UNII: 3QPI1U3FV8) INGR BUTYLPHENYL METHYLPROPIONAL (UNII: T7540GJV69) INGR CHAMOMILE (UNII: FGL3685T2X) INGR COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) INGR D&C RED NO. 33 (UNII: 9DBA0SBB0L) INGR DIHYDROMYRCENOL (UNII: 46L1B02ND9) INGR DIOSCOREA VILLOSA TUBER (UNII: IWY3IWX2G8) INGR ETHYLPARABEN (UNII: 14255EXE39) INGR FD&C YELLOW NO. 5 (UNII: I753WB2F1M) INGR GERANYL ACETATE (UNII: 3W81YG7P9R) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR HEXYL SALICYLATE (UNII: 8F78EY72YL) INGR HYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE (UNII: QUE43B9Z2Q) INGR ISOBUTYLPARABEN (UNII: 0QQJ25X58G) INGR ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) INGR LIMONENE, (+)- (UNII: GFD7C86Q1W) INGR LINALOOL, (+/-)- (UNII: D81QY6I88E) INGR METHYLPARABEN (UNII: A2I8C7HI9T) INGR PHENOXYETHANOL (UNII: HIE492ZZ3T) INGR PHENYLETHYL ALCOHOL (UNII: ML9LGA7468) INGR PROPYLPARABEN (UNII: Z8IX2SC1OH) INGR QUILLAJA SAPONARIA BARK (UNII: 8N0K3807ZW) INGR SMILAX ARISTOLOCHIIFOLIA ROOT (UNII: NR100Y25G0) INGR SODIUM CHLORIDE (UNII: 451W47IQ8X) INGR SODIUM LAURETH-3 SULFATE (UNII: BPV390UAP0) INGR SODIUM LAUROYL OAT AMINO ACIDS (UNII: FSW2K9B9N5) INGR WATER (UNII: 059QF0KO0R) INGR XANTHAN GUM (UNII: TTV12P4NEE) INGR YUCCA SCHIDIGERA ROOT (UNII: E2H9ET15AT) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 60 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 04/01/2022 Part 2 of 5 OBAGI NU-DERM TONER
face and neck (excluding shaving preparations) liquidProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR .ALPHA.-AMYLCINNAMALDEHYDE (UNII: WC51CA3418) INGR ACETYL HEXAMETHYL TETRALIN (UNII: 360Q8Z01IP) INGR ALLANTOIN (UNII: 344S277G0Z) INGR ALOE VERA LEAF (UNII: ZY81Z83H0X) INGR BORAGO OFFICINALIS SEED (UNII: 2GXJ790US0) INGR BUTYLPHENYL METHYLPROPIONAL (UNII: T7540GJV69) INGR CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) INGR DIHYDROMYRCENOL (UNII: 46L1B02ND9) INGR DIOSCOREA VILLOSA TUBER (UNII: IWY3IWX2G8) INGR DMDM HYDANTOIN (UNII: BYR0546TOW) INGR FD&C BLUE NO. 1 (UNII: H3R47K3TBD) INGR GERANYL ACETATE (UNII: 3W81YG7P9R) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR HAMAMELIS VIRGINIANA TOP WATER (UNII: NT00Y05A2V) INGR HEXYL SALICYLATE (UNII: 8F78EY72YL) INGR HYDROXYISOHEXYL 3-CYCLOHEXENE CARBOXALDEHYDE (UNII: QUE43B9Z2Q) INGR IODOPROPYNYL BUTYLCARBAMATE (UNII: 603P14DHEB) INGR ISOMETHYL-.ALPHA.-IONONE (UNII: 9XP4LC555B) INGR LIMONENE, (+)- (UNII: GFD7C86Q1W) INGR LINALOOL, (+/-)- (UNII: D81QY6I88E) INGR PANTHENOL (UNII: WV9CM0O67Z) INGR PHENYLETHYL ALCOHOL (UNII: ML9LGA7468) INGR POLYSORBATE 80 (UNII: 6OZP39ZG8H) INGR POTASSIUM ALUM (UNII: 1L24V9R23S) INGR QUILLAJA SAPONARIA BARK (UNII: 8N0K3807ZW) INGR SAGE (UNII: 065C5D077J) INGR SMILAX ARISTOLOCHIIFOLIA ROOT (UNII: NR100Y25G0) INGR SODIUM PYRROLIDONE CARBOXYLATE (UNII: 469OTG57A2) INGR WATER (UNII: 059QF0KO0R) INGR YUCCA SCHIDIGERA ROOT (UNII: E2H9ET15AT) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 59 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 04/01/2022 Part 3 of 5 OBAGI MEDICAL PROFESSIONAL-C SERUM 15
face and neck (excluding shaving preparations) liquidProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR ALCOHOL (UNII: 3K9958V90M) INGR ASCORBIC ACID (UNII: PQ6CK8PD0R) INGR ASCORBYL GLUCOSIDE (UNII: 2V52R0NHXW) INGR EVERNIA PRUNASTRI (UNII: O3034Q5AHK) INGR GRAPEFRUIT PEEL (UNII: 3582N05Q44) INGR OCTOXYNOL-9 (UNII: 7JPC6Y25QS) INGR PROPYLENE GLYCOL (UNII: 6DC9Q167V3) INGR PSEUDEVERNIA FURFURACEA (UNII: 49ZMN09Q0K) INGR WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 CARTON 1 30 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 04/01/2022 Part 4 of 5 OBAGI MEDICAL RETINOL
face and neck (excluding shaving preparations) creamProduct Information Route of Administration TOPICAL Other Ingredients Ingredient Kind Ingredient Name Quantity INGR .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) INGR ALLYL METHACRYLATE/GLYCOL DIMETHACRYLATE CROSSPOLYMER (UNII: B9J55EA6QX) INGR ASCORBIC ACID (UNII: PQ6CK8PD0R) INGR BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) INGR BUTYLENE GLYCOL (UNII: 3XUS85K0RA) INGR CAPRYLYL GLYCOL (UNII: 00YIU5438U) INGR CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) INGR CHAMOMILE (UNII: FGL3685T2X) INGR CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) INGR CYCLOMETHICONE 6 (UNII: XHK3U310BA) INGR DIMETHICONE (UNII: 92RU3N3Y1O) INGR DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (SOFT PARTICLE) (UNII: 9E4CO0W6C5) INGR EDETATE DISODIUM (UNII: 7FLD91C86K) INGR ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) INGR GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) INGR HEXYLENE GLYCOL (UNII: KEH0A3F75J) INGR HYALURONATE SODIUM (UNII: YSE9PPT4TH) INGR HYPERICUM PERFORATUM FLOWER (UNII: A6V4CUE7PV) INGR JOJOBA OIL (UNII: 724GKU717M) INGR LEVOMENOL (UNII: 24WE03BX2T) INGR MALVA SYLVESTRIS FLOWER (UNII: 12X9JI52BS) INGR MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) INGR PHENOXYETHANOL (UNII: HIE492ZZ3T) INGR POLYSORBATE 20 (UNII: 7T1F30V5YH) INGR POLYSORBATE 60 (UNII: CAL22UVI4M) INGR PROPYLENE GLYCOL (UNII: 6DC9Q167V3) INGR RETINOL (UNII: G2SH0XKK91) INGR SAGE (UNII: 065C5D077J) INGR SAMBUCUS NIGRA FLOWER (UNII: 07V4DX094T) INGR SHEA BUTTER (UNII: K49155WL9Y) INGR SORBITOL (UNII: 506T60A25R) INGR STEARIC ACID (UNII: 4ELV7Z65AP) INGR STEARYL ALCOHOL (UNII: 2KR89I4H1Y) INGR TROLAMINE (UNII: 9O3K93S3TK) INGR UBIDECARENONE (UNII: EJ27X76M46) INGR WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 CARTON 1 30 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date COSMETIC 04/01/2022 Part 5 of 5 OBAGI MEDICAL SUN SHIELD BROAD SPECTRUM SPF 50 MATTE SUNCREEN
homosalate, octisalate, and zinc oxide lotionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 10 g in 100 g OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 g ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 16.5 g in 100 g Inactive Ingredients Ingredient Name Strength 1,2-HEXANEDIOL (UNII: TR046Y3K1G) C15-19 ALKANE (UNII: CI87N1IM01) CANDELILLA WAX (UNII: WL0328HX19) CAPRYLYL GLYCOL (UNII: 00YIU5438U) DIMETHICONE (UNII: 92RU3N3Y1O) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) GLYCERIN (UNII: PDC6A3C0OX) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) OCTYLDODECYL NEOPENTANOATE (UNII: X8725R883T) POLYGLYCERYL-2 ISOSTEARATE (UNII: 7B8OE71MQC) POLYGLYCERYL-6 POLYRICINOLEATE (UNII: YPM0ZOC2HR) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) POLYMETHYLSILSESQUIOXANE (4.5 MICRONS) (UNII: 59Z907ZB69) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBITAN OLIVATE (UNII: MDL271E3GR) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) WATER (UNII: 059QF0KO0R) XANTHAN GUM (UNII: TTV12P4NEE) YELLOW WAX (UNII: 2ZA36H0S2V) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 07/15/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part352 04/01/2022 Labeler - Obagi Cosmeceuticals LLC (790553353) Establishment Name Address ID/FEI Business Operations G.S.COSMECEUTICAL USA, INC. 017014734 MANUFACTURE(62032-694) Establishment Name Address ID/FEI Business Operations Denison Pharmaceuticals, LLC 001207208 MANUFACTURE(62032-694) Establishment Name Address ID/FEI Business Operations Swiss-American CDMO, LLC 080170933 MANUFACTURE(62032-694) Establishment Name Address ID/FEI Business Operations TENCO ASSEMBLIES, INC 075535609 PACK(62032-694)