Label: SOOTHING BATH TREATMENT- colloidal oatmeal powder
-
Contains inactivated NDC Code(s)
NDC Code(s): 37205-178-51 - Packager: Cardinal Health
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated December 23, 2009
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT (in each packet)
- PURPOSE
- USE
- WARNINGS
-
DIRECTIONS
- Turn warm water faucet on to full force.
- Slowly sprinkle 1 packet of colloidal oatmeal directly under the faucet into the tub or container.
- Stir any colloidal oatmeal that may have settled to the bottom.
- Soak affected area for 15-30 minutes as needed or as directed by a doctor.
- Pat dry (do not rub) to keep a thin layer on the skin.
- SPL UNCLASSIFIED SECTION
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- QUESTIONS
-
PACKAGE INFORMATION
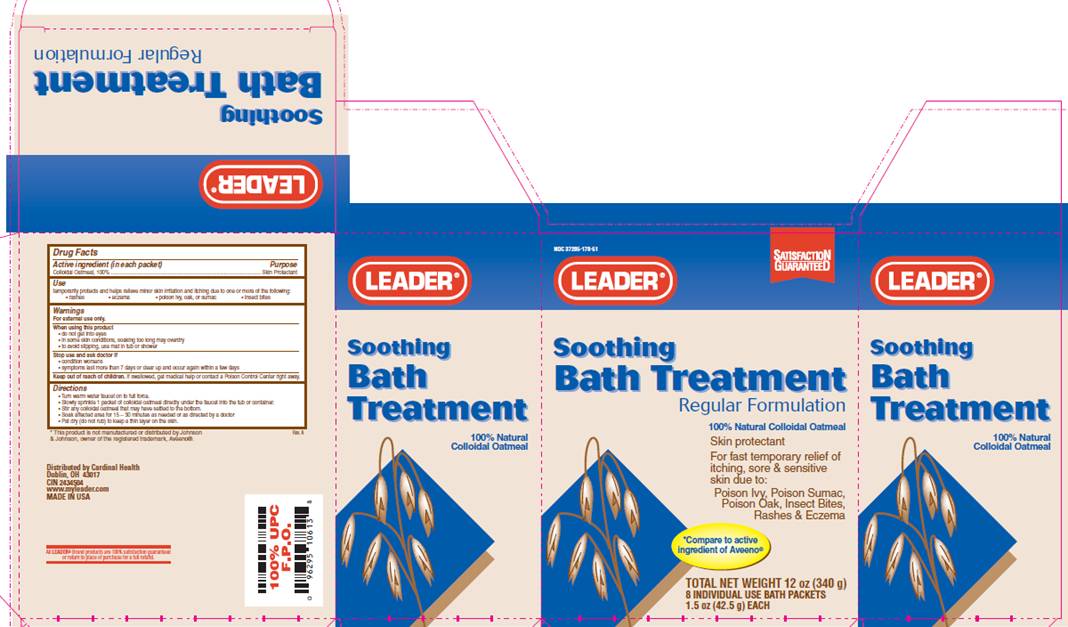
NDC 37205-178-51
LEADER SATISFACTION GUARANTEED
Soothing
Bath Treatment
Regular Formulation
100% Natural Colloidal Oatmeal
Skin Protectant
For fast temporary relief of itching , sore and sensitive skin due to:
Poison Ivy, Poison Sumac, Poison Oak, Insect Bites, Rashes and Eczema
*Compare to active ingredient of Aveeno®
TOTAL NET WEIGHT 12 oz (340 g)
8 INDIVIDUAL USE BATH PACKETS
1.5 oz (42.5 g) EACH
*This product is not manufactured or distributed by Johnson and Johnson, owner of the registered trademark, Aveeno®.
Distributed by Cardinal Health
Dublin, OH 43017
CIN 2434504
www.myleader.com
MADE IN USA
All Leader Brand products are 100% satisfaction guaranteed or return to place of purchase for a full refund.
-
INGREDIENTS AND APPEARANCE
SOOTHING BATH TREATMENT
colloidal oatmeal powderProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:37205-178 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OATMEAL (UNII: 8PI54V663Y) (OATMEAL - UNII:8PI54V663Y) OATMEAL 1 g in 1 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:37205-178-51 8 in 1 CARTON 1 42.5 g in 1 PACKET Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 07/21/2005 Labeler - Cardinal Health (097537435)