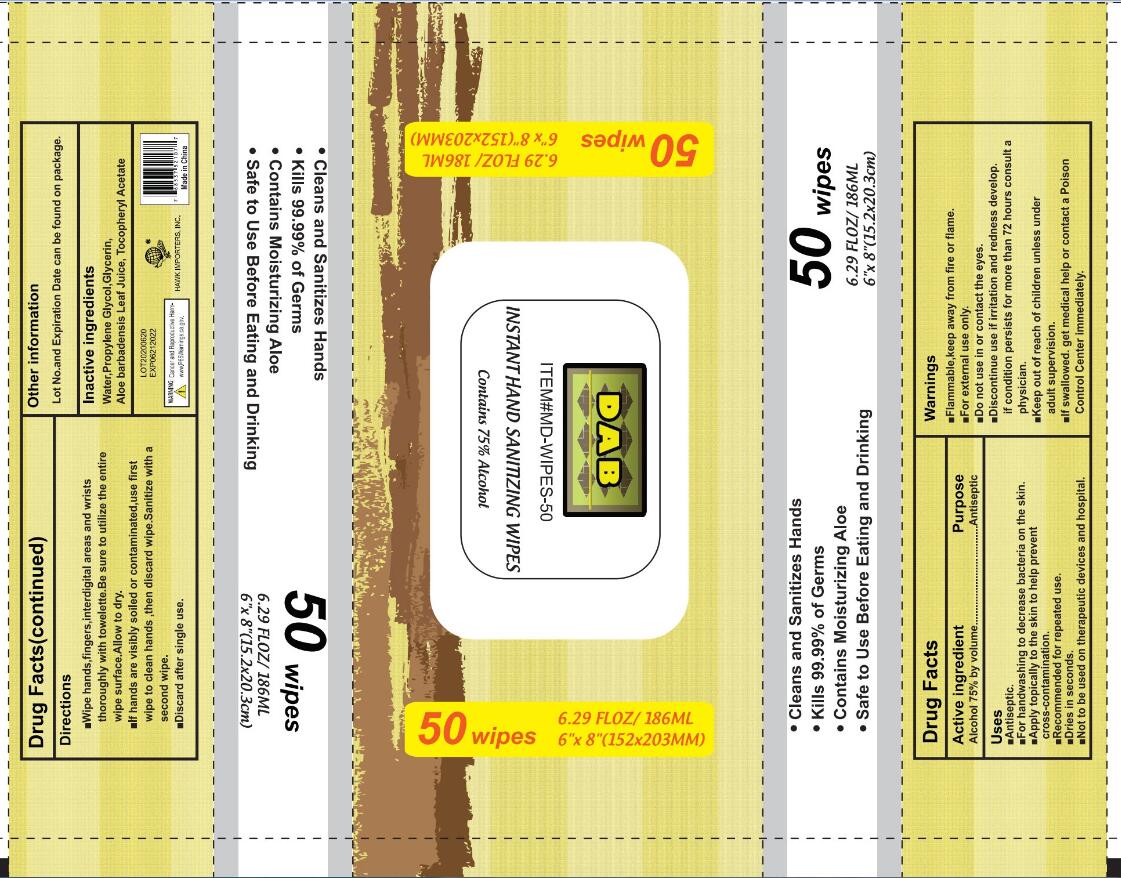
Label: INSTANT HAND SANITIZING WIPES cloth
-
Contains inactivated NDC Code(s)
NDC Code(s): 76991-010-01, 76991-010-02, 76991-010-03 - Packager: Hefei Wenqi Industrial & Trade Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 24, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient(s)
- Purpose
- Use
-
Warnings
Flammable, keep away from fire or flame
For extemal use only
Do not use in or contact the eyes
Discontinue useif irritation and redness develop. if condition persists for more than 72 hours consult a physician.
keep out of reach of children unless under adult supervision.
lf swallowed. get medical help or contact a Poison Control Center immediately. - Do not use
-
WHEN USING
Pull through about one inch. Replace cover Pull out wipes as needed and snap off at a 90° angle. Keep cap closed to prevent evaporation
Wipe hands, fingers, interdigital areas and wrists thoroughly with towelette. Be sure to utilize the entire wipe surface, Allow to dry
lf hands are visibly soiled or contaminated use first wipe to clean hands then discard wipe Sanitize with a second wipe discard after single use. - STOP USE
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Other information
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
INSTANT HAND SANITIZING WIPES
instant hand sanitizing wipes clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76991-010 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 75 mL in 100 mL Inactive Ingredients Ingredient Name Strength ALOE (UNII: V5VD430YW9) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) TOCOPHEROL (UNII: R0ZB2556P8) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76991-010-01 92.57 mL in 1 BAG; Type 0: Not a Combination Product 05/15/2020 2 NDC:76991-010-02 186 mL in 1 BAG; Type 0: Not a Combination Product 05/15/2020 3 NDC:76991-010-03 347 mL in 1 BOTTLE; Type 0: Not a Combination Product 05/15/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 03/30/2020 Labeler - Hefei Wenqi Industrial & Trade Co., Ltd (530739868) Establishment Name Address ID/FEI Business Operations Hefei Wenqi Industrial & Trade Co., Ltd 530739868 manufacture(76991-010)