Label: LYVISPAH- baclofen granule
-
Contains inactivated NDC Code(s)
NDC Code(s): 70257-412-87, 70257-414-87, 70257-416-87 - Packager: Saol Therapeutics Inc
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated November 26, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Medication Guide: HTML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LYVISPAH safely and effectively. See full prescribing information for LYVISPAH.
LYVISPAH (baclofen) oral granules
Initial U.S. Approval: 1977INDICATIONS AND USAGE
- LYVISPAH is a gamma-aminobutyric acid (GABA-ergic) agonist indicated for the treatment of spasticity resulting from multiple sclerosis, particularly for the relief of flexor spasms and concomitant pain, clonus, and muscular rigidity. (1)
- LYVISPAH may also be of some value in patients with spinal cord injuries and other spinal cord diseases. (1)
Limitations of Use
LYVISPAH is not indicated in the treatment of skeletal muscle spasm resulting from rheumatic disorders. (1)DOSAGE AND ADMINISTRATION
- Initiate LYVISPAH with a low dosage, preferably in divided doses, administered orally. Increase gradually based on clinical response and tolerability. (2.1))
- The maximum dosage is 80 mg daily (20 mg four times a day). (2.1)
- LYVISPAH can be taken with or without water. (2.2)
- LYVISPAH oral granules can be mixed with soft food for administration within 2 hours. (2.2)
- LYVISPAH oral granules can be administered via enteral feeding tubes. (2.2)
- When discontinuing, reduce the dose slowly. (2.2)
DOSAGE FORMS AND STRENGTHS
Oral granules: 5 mg, 10 mg, or 20 mg baclofen in a packet (3)
CONTRAINDICATIONS
- Hypersensitivity to baclofen (4)
WARNINGS AND PRECAUTIONS
- Abrupt discontinuation of baclofen has resulted in serious adverse reactions including death; therefore, reduce the dosage slowly when LYVISPAH is discontinued. (5.1)
- Neonatal withdrawal symptoms can occur; gradually reduce the dosage and discontinue LYVISPAH before delivery. (5.2)
- LYVISPAH can cause drowsiness and sedation. Patients should avoid the operation of automobiles or other dangerous machinery until they know how the drug affects them. Advise patients that the central nervous system effects of LYVISPAH may be additive to those of alcohol and other CNS depressants. (5.3)
- LYVISPAH can cause exacerbation of the following: psychotic disorders, schizophrenia, or confusional states; autonomic dysreflexia; epilepsy. Use with caution in patients with these conditions. (5.5, 5.6, 5.7)
ADVERSE REACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 11/2021
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
2.2 Administration Instructions
2.3 Discontinuation of LYVISPAH
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Adverse Reactions from Abrupt Withdrawal of LYVISPAH
5.2 Neonatal Withdrawal Symptoms
5.3 Drowsiness and Sedation
5.4 Poor Tolerability in Stroke Patients
5.5 Exacerbation of Psychotic Disorders, Schizophrenia, or Confusional States
5.6 Exacerbation of Autonomic Dysreflexia
5.7 Exacerbation of Epilepsy
5.8 Posture and Balance Effects
5.9 Ovarian Cysts
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 CNS Depressants and Alcohol
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
10 OVERDOSAGE
10.1 Symptoms of Baclofen Overdose
10.2 Treatment for Overdose
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
LYVISPAH is indicated for the treatment of spasticity resulting from multiple sclerosis, particularly for the relief of flexor spasms and concomitant pain, clonus, and muscular rigidity.
LYVISPAH may also be of some value in patients with spinal cord injuries and other spinal cord diseases.
Limitations of Use
LYVISPAH is not indicated in the treatment of skeletal muscle spasm resulting from rheumatic disorders.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage
Initiate LYVISPAH with a low dosage, preferably in divided doses, administered orally. The following gradually increasing dosage regimen is suggested, but should be adjusted based on clinical response and tolerability:
5 mg three times a day for three days
10 mg three times a day for three days
15 mg three times a day for three days
20 mg three times a day for three daysAdditional increases may be necessary up to the maximum recommended dosage of 80 mg daily (20 mg four times a day). Multiple packets or multiple strengths can be used to achieve the prescribed dosage.
2.2 Administration Instructions
The entire contents of the packet should be emptied into the mouth. The granules will dissolve in the mouth or can be swallowed. LYVISPAH can be taken with liquids or soft foods if needed.
Administration with Liquids or Soft Foods
LYVISPAH can be administered orally as a mixture with liquids or soft foods, such as apple sauce, yogurt, or pudding. The contents of one packet can be emptied and mixed with up to 15 mL of liquid or soft food. The mixture should be administered no more than 2 hours after mixing. If multiple packets are to be administered, each packet must be mixed with a separate volume of liquid or soft food.dministration via Feeding Tube
LYVISPAH can also be administered via enteral feeding tubes such as nasogastric (NG) at sizes 8 FR or higher, gastrostomy (G) at sizes 12 FR or higher, percutaneous endoscopic gastrostomy (PEG) at sizes 14 FR or higher, and gastrojejunostomy (GJ) tubes at sizes 16 FR or higher.- Flush the feeding tube with up to 15 mL of water using a catheter tip syringe.
- Open and empty the full contents of one packet of LYVISPAH in 15 mL of liquid, such as apple juice or milk, in a clean container. Mix the suspension to ensure all granules are wetted.
- Draw up the suspension of granules into a dosing syringe immediately after mixing and administer the dose via the feeding tube. Administration should be no longer than two hours after mixing. If the syringe is allowed to stand for 15 minutes before administration, invert the syringe three times.
- Refill the dosing syringe with 15 mL of water and flush the feeding tube.
- If multiple packets are to be administered, each packet must be mixed with a separate volume of liquid.
2.3 Discontinuation of LYVISPAH
When discontinuing LYVISPAH, reduce the dosage slowly and avoid abrupt withdrawal from the drug to help minimize the risk of adverse reactions [see Warnings and Precautions (5.1)].
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Adverse Reactions from Abrupt Withdrawal of LYVISPAH
Abrupt discontinuation of baclofen, regardless of the cause, has resulted in adverse reactions that include hallucinations, seizures, high fever, altered mental status, exaggerated rebound spasticity, and muscle rigidity, that in rare cases has advanced to rhabdomyolysis, multiple organ-system failure, and death. Therefore, reduce the dosage slowly when LYVISPAH is discontinued, unless the clinical situation justifies a rapid withdrawal.
5.2 Neonatal Withdrawal Symptoms
Withdrawal symptoms in neonates whose mothers were treated with oral baclofen throughout pregnancy have been reported starting hours to days after delivery. The symptoms of withdrawal in these infants have included increased muscle tone, tremor, jitteriness, and seizure. If the potential benefit justifies the potential risk to the fetus and LYVISPAH is continued during pregnancy, gradually reduce the dosage, and discontinue LYVISPAH before delivery. If slow withdrawal is not feasible, advise the parents or caregivers of the exposed neonate of the potential for neonatal withdrawal.
5.3 Drowsiness and Sedation
Drowsiness and sedation have been reported in up to 63% of patients taking baclofen, the active ingredient in LYVISPAH [see Adverse Reactions (6.1)]. Patients should avoid operation of automobiles or other dangerous machinery and activities made hazardous by decreased alertness when starting LYVISPAH or increasing the dosage until they know how the drug affects them. Advise patients that the central nervous system depressant effects of LYVISPAH may be an additive to those of alcohol and other CNS depressants.
5.4 Poor Tolerability in Stroke Patients
LYVISPAH should be used with caution in patients who have had a stroke. Baclofen has not significantly benefited patients with stroke. These patients have also shown poor tolerability to the drug.
5.5 Exacerbation of Psychotic Disorders, Schizophrenia, or Confusional States
LYVISPAH should be used with caution in patients suffering from psychotic disorders, schizophrenia, or confusional states. If treated with LYVISPAH, these patients should be kept under careful surveillance because exacerbations of these conditions have been observed with oral baclofen administration.
5.6 Exacerbation of Autonomic Dysreflexia
LYVISPAH should be used with caution in patients with a history of autonomic dysreflexia. The presence of nociceptive stimuli or abrupt withdrawal of LYVISPAH may cause an autonomic dysreflexic episode.
5.7 Exacerbation of Epilepsy
LYVISPAH should be used with caution in patients with epilepsy. Deterioration in seizure control has been reported in patients taking baclofen.
5.8 Posture and Balance Effects
LYVISPAH should be used with caution in patients where spasticity is utilized to sustain upright posture and balance in locomotion or whenever spasticity is utilized to obtain increased function.
5.9 Ovarian Cysts
A dose-related increase in incidence of ovarian cysts was observed in female rats treated chronically with oral baclofen. Ovarian cysts have been found by palpation in about 4% of the multiple sclerosis patients who were treated with oral baclofen for up to one year. In most cases, these cysts disappeared spontaneously while patients continue to receive the drug. Ovarian cysts are estimated to occur spontaneously in approximately 1% to 5% of the normal female population.
-
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Adverse Reactions from Abrupt Withdrawal of LYVISPAH [see Warnings and Precautions (5.1)]
- Neonatal Withdrawal Symptoms [see Warnings and Precautions (5.2)]
- Drowsiness and Sedation [see Warnings and Precautions (5.3)]
- Poor Tolerability in Stroke Patients [see Warnings and Precautions (5.4)]
- Exacerbation of Psychotic Disorders, Schizophrenia, or Confusional States [see Warnings and Precautions (5.5)]
- Exacerbation of Autonomic Dysreflexia [see Warnings and Precautions (5.6)]
- Exacerbation of Epilepsy [see Warnings and Precautions (5.7)]
- Posture and Balance Effects [see Warnings and Precautions (5.8)]
- Ovarian Cysts [see Warnings and Precautions (5.9)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reaction is transient drowsiness. In one controlled study of 175 patients, transient drowsiness was observed in 63% of those receiving baclofen compared to 36% of those in the placebo group. Other common adverse reactions (up to 15%) are dizziness and weakness. Adverse reactions with a frequency of ≥1% are listed in Table 1.
Table 1. Common (≥1%) Adverse Reactions in Patients Treated with Baclofen for Spasticity ADVERSE REACTION PERCENT Drowsiness 10-63% Dizziness 5-15% Weakness 5-15% Nausea 4-12% Confusion 1-11% Hypotension 0-9% Headache 4-8% Insomnia 2-7% Constipation 2-6% Urinary Frequency 2-6% Fatigue 2-4% The following adverse reactions not included in Table 1, classified by body system, were also reported:
Neuropsychiatric: euphoria, excitement, depression, hallucinations, paresthesia, muscle pain, tinnitus, slurred speech, coordination disorder, tremor, rigidity, dystonia, ataxia, blurred vision, nystagmus, strabismus, miosis, mydriasis, diplopia, dysarthria, epileptic seizure
Cardiovascular: dyspnea, palpitation, chest pain, syncope
Gastrointestinal: dry mouth, anorexia, taste disorder, abdominal pain, vomiting, diarrhea, and positive test for occult blood in stool
Genitourinary: enuresis, urinary retention, dysuria, impotence, inability to ejaculate, nocturia, hematuria
Other: rash, pruritus, ankle edema, excessive perspiration, weight gain, nasal congestionThe following laboratory tests have been found to be abnormal in patients receiving baclofen: increased SGOT, elevated alkaline phosphatase, and elevation of blood sugar.
-
7 DRUG INTERACTIONS
7.1 CNS Depressants and Alcohol
LYVISPAH can cause CNS depression, including drowsiness and sedation, which may be an additive when used concomitantly with other CNS depressants or alcohol [see Warnings and Precautions (5.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate data on the risk of major birth defects, miscarriages, or other maternal adverse outcomes associated with the use of LYVISPAH in pregnant women. There are adverse effects on fetal outcomes associated with withdrawal from baclofen after delivery (see Clinical Considerations). Oral administration of baclofen to pregnant rats resulted in an increased incidence of fetal structural abnormalities at a dose which was also associated with maternal toxicity. The background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.Clinical Considerations
Fetal/Neonatal Adverse Reactions
LYVISPAH may increase the risk of late-onset neonatal withdrawal symptoms [see Warnings and Precautions (5.2)].Data
Animal Data
Baclofen given orally has been shown to increase the incidence of omphaloceles (ventral hernias) in fetuses of rats given approximately 13 times on a mg/kg basis, or 3 times on a mg/m2 basis, the maximum oral dose recommended for human use; this dose also caused reductions in food intake and weight gain in the dams. This abnormality was not seen in mice or rabbits.8.2 Lactation
Risk Summary
At recommended oral doses, baclofen is present in human milk. There are no human data on effects of baclofen on milk production. Withdrawal symptoms can occur in breastfed infants when maternal administration of LYVISPAH is stopped, or when breastfeeding is stopped [see Warnings and Precautions (5.2)]. There are no adequate data on other effects of baclofen on the breastfed infant.The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for LYVISPAH and any potential adverse effects on the breastfed infant from LYVISPAH or from the underlying maternal condition.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients below the age of 12 have not been established.
8.5 Geriatric Use
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. This drug is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Use in Specific Populations (8.6)].
- 10 OVERDOSAGE
-
11 DESCRIPTION
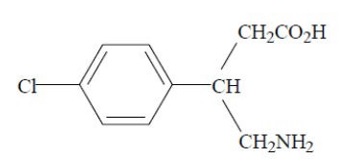
LYVISPAH (baclofen) oral granules is a gamma-aminobutyric acid (GABA-ergic) agonist available as 5 mg, 10 mg, or 20 mg of baclofen oral granules in a packet. Its chemical name is 4-amino-3-(4- chlorophenyl)-butanoic acid and its structural formula is:
Molecular formula is C10H12ClNO2
Molecular Weight is 213.66.
Baclofen USP is a white to off-white, odorless or practically odorless crystalline powder. It is slightly soluble in water, very slightly soluble in methanol, and insoluble in chloroform.
LYVISPAH (baclofen) oral granules inactive ingredients include amino methacrylate copolymer, calcium stearate, colloidal silicon dioxide, crospovidone, hypromellose, mannitol, saccharin sodium, strawberry flavor, talc, and xylitol.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The precise mechanism of action of baclofen is not fully understood. Baclofen inhibits both monosynaptic and polysynaptic reflexes at the spinal level, possibly by decreasing excitatory neurotransmitter release from afferent terminals, although actions at supraspinal sites may also occur and contribute to its clinical effect. Baclofen is a structural analog of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA), and may exert its effects by stimulation of the GABA-β receptor subtype.
12.2 Pharmacodynamics
Baclofen has been shown to have general CNS depressant properties, as indicated by the production of sedation with tolerance, somnolence, ataxia, and respiratory and cardiovascular depression [see Warnings and Precautions (5.3), Adverse Reactions (6.1), and Overdosage (10.1)].
12.3 Pharmacokinetics
Pharmacokinetic studies in heathy adult subjects under fasting conditions at 20 mg dose level demonstrated similar bioavailability for baclofen oral granules and oral tablets.
Absorption
The peak plasma concentrations of baclofen oral granule formulation were achieved in about one hour and the apparent elimination half-life is about 5.5 hours. The exposure of baclofen was dose proportional across the dose range of 5 mg, 10 mg, and 20 mg.Effect of Food
Administering LYVISPAH with or without water, or with applesauce did not affect the bioavailability of baclofen oral granules. Administration with a high fat meal resulted in 10% decrease in AUC and 29% decrease in Cmax compared to the fasted state.Elimination
Baclofen is excreted primarily by the kidney in unchanged form, and there is relatively large intersubject variation in absorption and/or elimination. -
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
No increase in tumors was seen in rats receiving baclofen orally for two years at approximately 30 to 60 times on a mg/kg basis, or 10 to 20 times on a mg/m2 basis, the maximum oral dose recommended for human use.Mutagenesis
Genetic toxicology assays have not been conducted for baclofen.Impairment of Fertility
Studies to evaluate the effects of baclofen on fertility have not been conducted. -
14 CLINICAL STUDIES
The efficacy of LYVISPAH is based upon a bioavailability study in healthy adults comparing baclofen oral tablets to LYVISPAH [see Clinical Pharmacology (12.3)].
-
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
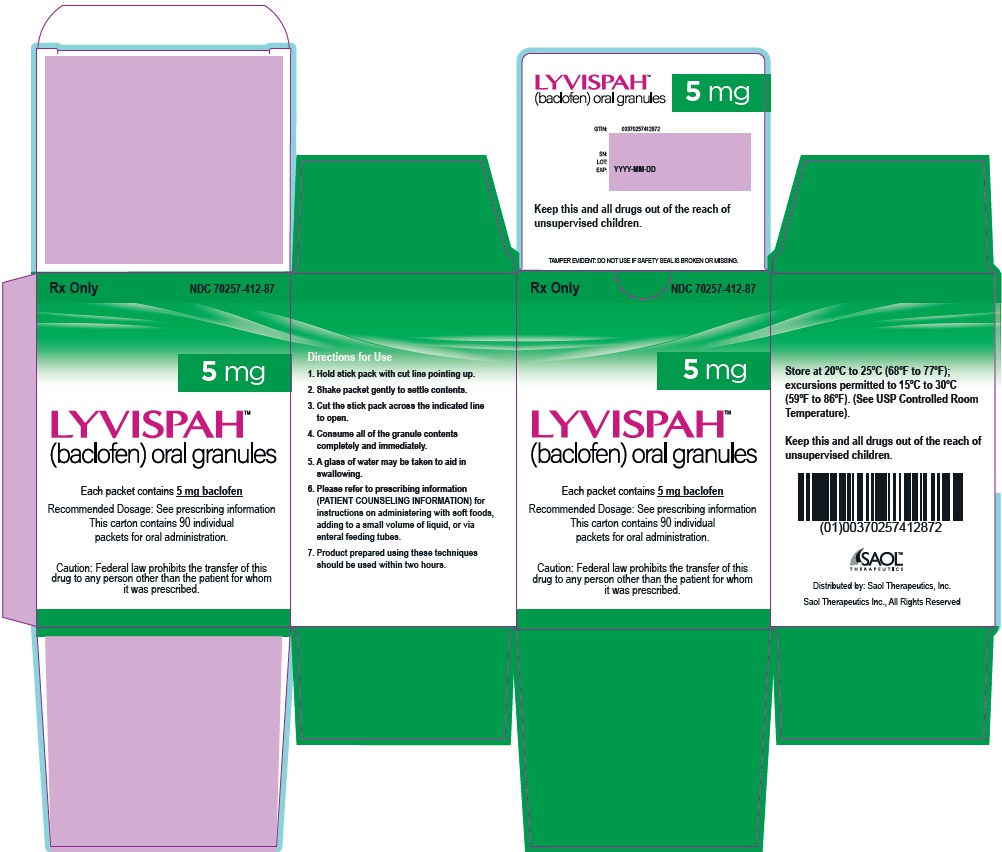
LYVISPAH (baclofen) oral granules is supplied as follows:
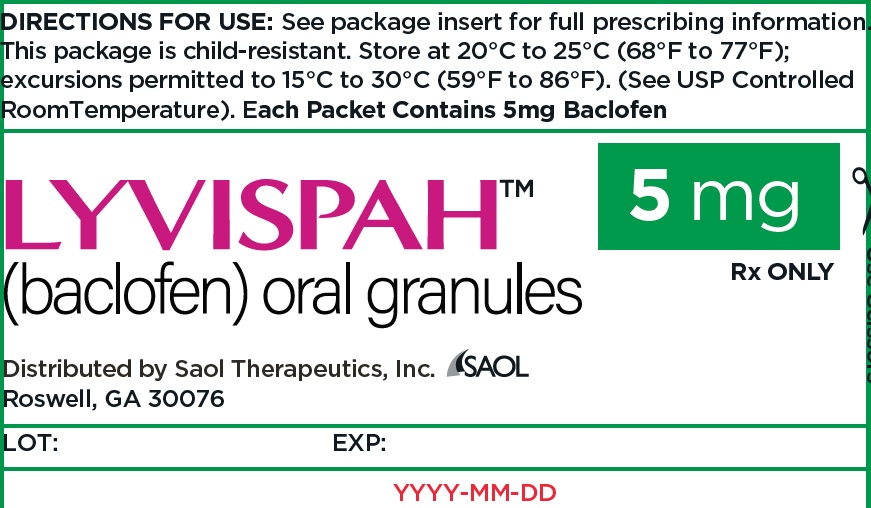
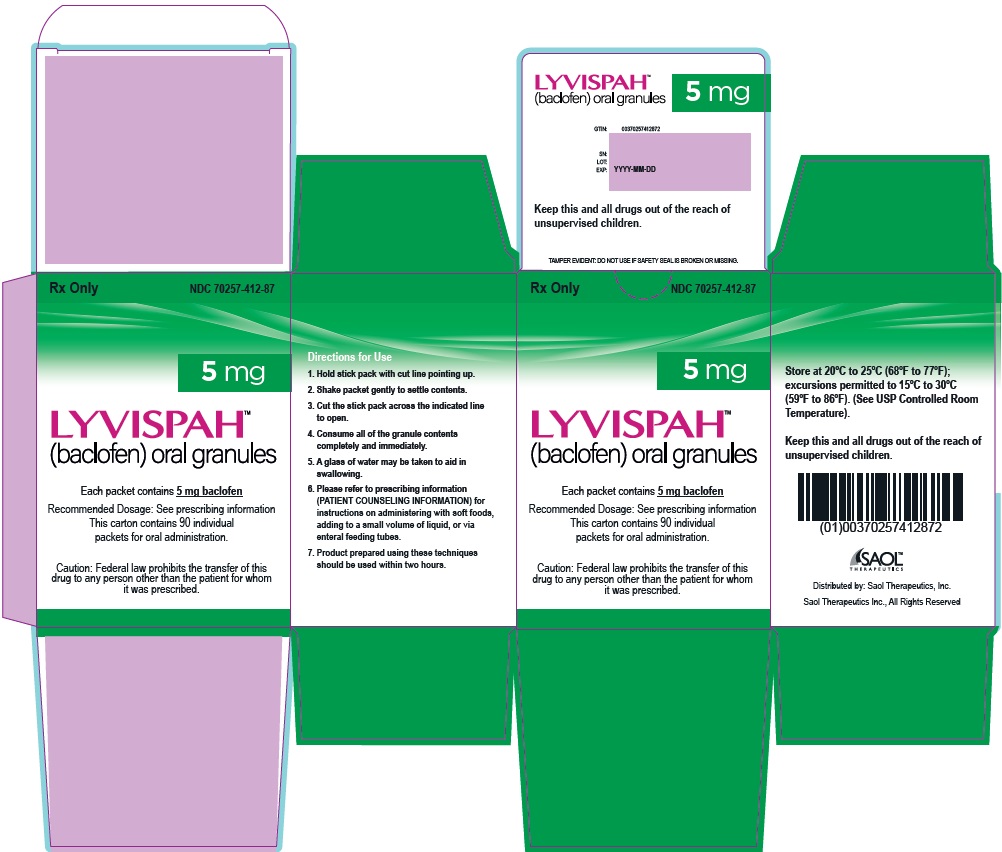
- 5 mg white to off-white, strawberry flavored oral granules in a child-resistant single dose packet
- NDC 70257-412-87: carton of 90 containing packets
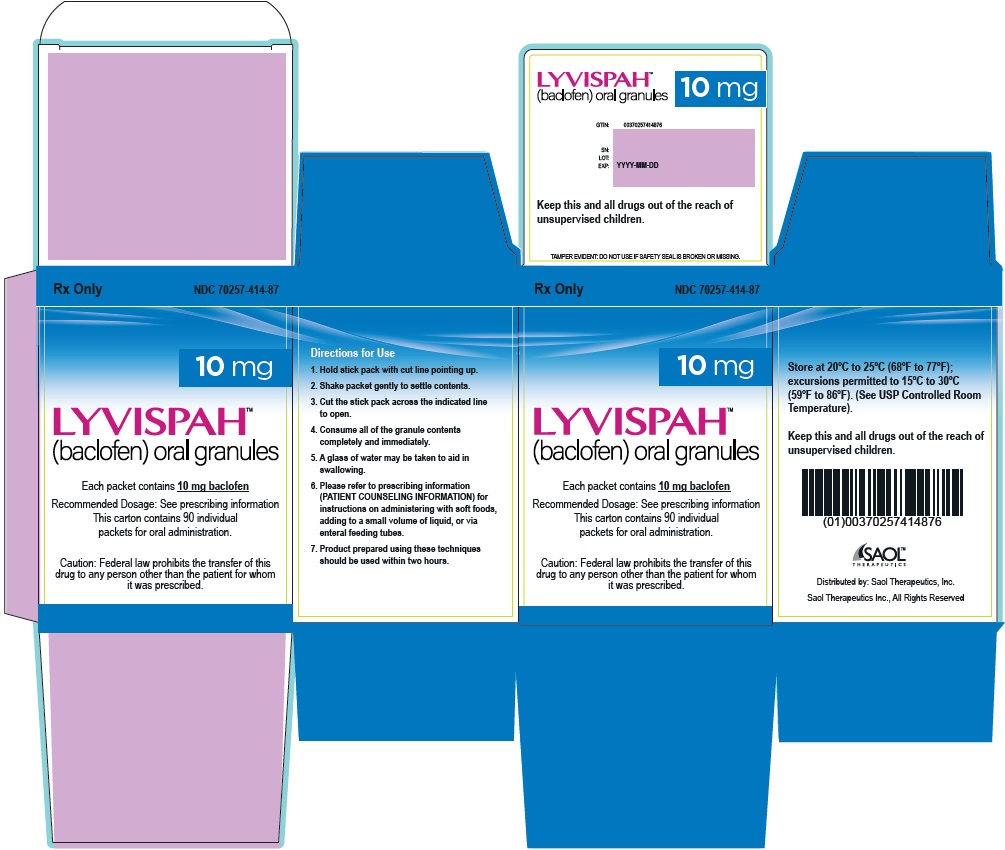
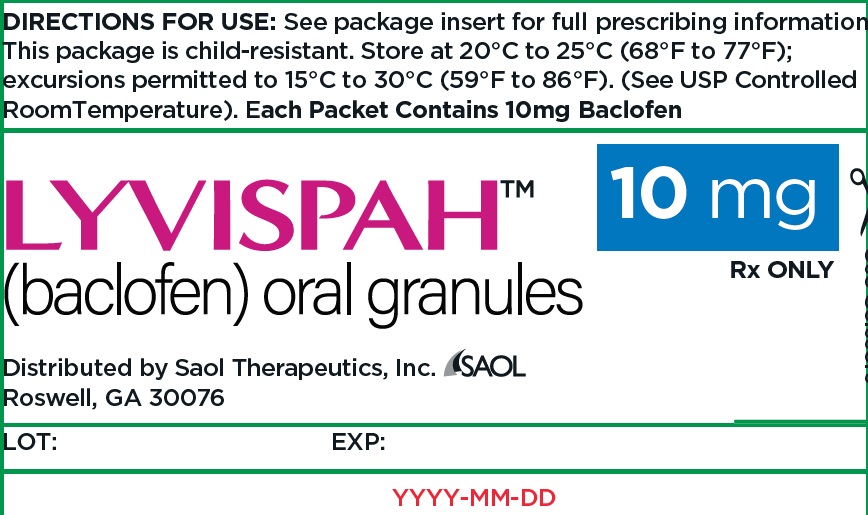
- 10 mg white to off-white, strawberry flavored oral granules in a child-resistant single dose packet
- NDC 70257-414-87: carton of 90 containing packets
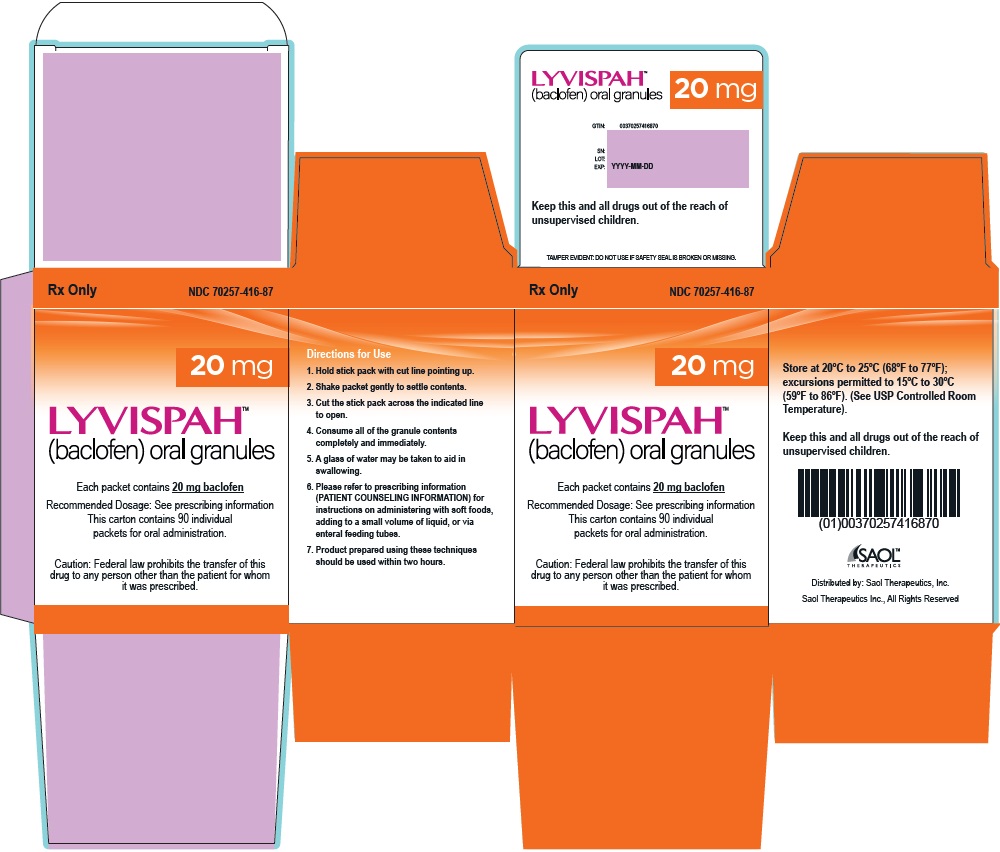
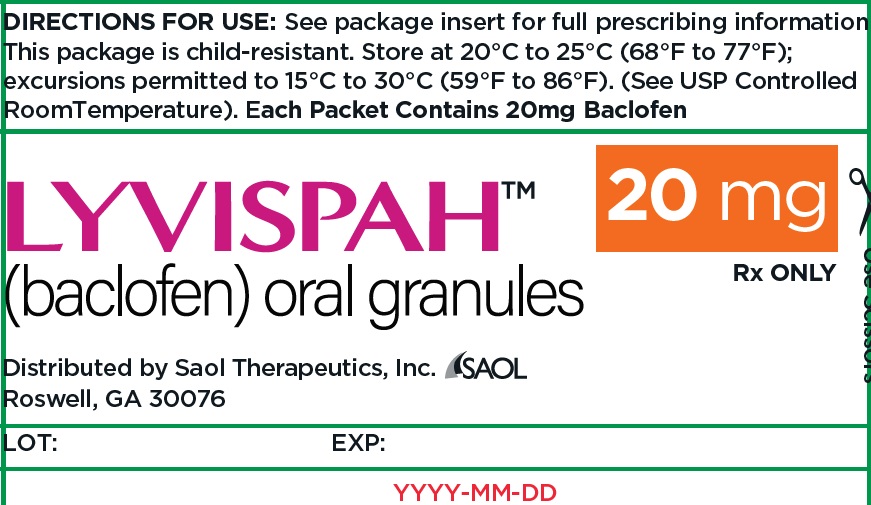
- 20 mg white to off-white, strawberry flavored oral granules in a child-resistant single dose packet
- NDC 70257-416-87: carton of 90 containing packets
- 5 mg white to off-white, strawberry flavored oral granules in a child-resistant single dose packet
-
17 PATIENT COUNSELING INFORMATION
Advise the patient or caregiver to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
General Administration Instructions
Instruct patient or caregiver to carefully open the LYVISPAH packet and empty the entire contents, to obtain the prescribed amount of medication. Packet contents can be administered directly into the mouth. The contents of the packet can be swallowed or will dissolve in the mouth. LYVISPAH may be taken with liquids or can also be administered in soft foods if needed [see Dosage and Administration (2.2)].Administration Instructions with Soft Foods
LYVISPAH can also be administered by mouth as a mixture with liquids or soft foods, such as apple sauce, yogurt, or pudding. One packet can be mixed with up to 15 mL (one tablespoonful) of soft food. The mixture should be administered no more than 2 hours after mixing [see Dosage and Administration (2.2)].Administration Instructions via Feeding Tubes
LYVISPAH can be administered via enteral feeding tubes, such a nasogastric (NG), gastrostomy (G), percutaneous endoscopic gastrostomy (PEG), and gastrojejunostomy (GJ) tubes.- Flush the feeding tube with up to 15 mL (one tablespoonful) of water.
- Open and empty the full contents of one packet of LYVISPAH in 15 mL (one tablespoonful) of the preferred liquid. Stir the suspension to ensure all granules are wetted.
- Draw up the suspension of granules into a dosing syringe immediately after stirring and administer the dose via the feeding tube. Administer no longer than two hours after mixing.
- Refill the dosing syringe with 15 mL (one tablespoonful) of water and flush the feeding tube with the remaining contents.
- Any unused suspensions should be discarded.
Risks Related to Sudden Withdrawal of LYVISPAH
Advise patients and caregivers not to discontinue use of LYVISPAH without consulting with their healthcare provider because sudden withdrawal of LYVISPAH can result in serious complications that include hallucinations, seizures, high fever, confusion, muscle stiffness, multiple organ-system failure, and death [see Warnings and Precautions (5.1)]. Inform patients that early symptoms of LYVISPAH withdrawal may include increased spasticity, itching, and tingling of extremities.Neonatal Withdrawal Symptoms
Advise patients to notify their healthcare provider if they are pregnant, plan to become pregnant, or plan to breastfeed [see Warnings and Precautions (5.2) and Use in Specific Populations (8.2)].Increased Risk of Drowsiness with Alcohol and Other CNS Depressants
Advise patients that LYVISPAH may cause drowsiness, and that they should avoid the operation of automobiles or other dangerous machinery, or activities made hazardous by decreased alertness when starting LYVISPAH or increasing the dose of LYVISPAH until they know how the drug affects them [see Warnings and Precautions (5.3)]. Inform patients and their caregivers that the drowsiness associated with LYVISPAH use can be worsened by alcohol and other CNS depressants. Advise patients to read all medicine labels carefully, and to tell their healthcare provider about all prescription and nonprescription drugs they may use.
Distributed by:
Saol Therapeutics, Inc.
Roswell, GA 30076
-
MEDICATION GUIDE
This Patient Information has been approved by the U.S. Food and Drug Administration Issued: 11/2021 PATIENT INFORMATION
LYVISPAH (lye vis' pah)
(baclofen)
oral granulesWhat is LYVISPAH?
- LYVISPAH is a prescription medicine used to treat muscle stiffness, spasms, and pain from multiple sclerosis.
- LYVISPAH may be used to treat people with spinal cord injuries and other spinal cord diseases.
- LYVISPAH should not be used to treat muscle spasms from inflammatory (rheumatic) disorders.
- It is not known if LYVISPAH is safe and effective in children under 12 years of age.
Do not take LYVISPAH if you:
- are allergic to baclofen. See the end of this Patient Information leaflet for a complete list of ingredients in LYVISPAH.
Before taking LYVISPAH, tell your healthcare provider about all of your medical conditions, including if you:
- take certain medicines that cause drowsiness.
- have had a stroke.
- are being treated for mental illness, especially a psychotic disorder, schizophrenia, or other illness that may cause you to become confused.
- have or have had high blood pressure.
- have or have had seizures.
- have kidney problems.
- have problems with posture and balance.
- have a history of ovarian cysts
- drink alcohol.
- are pregnant or plan to become pregnant. It is not known if LYVISPAH will harm your unborn baby.
- are breastfeeding or plan to breastfeed. LYVISPAH can pass into your breast milk and may harm your baby. Talk to your healthcare provider about the best way to feed your baby if you take LYVISPAH.
Tell your healthcare provider about all the medicines you take, including prescription and over-the- counter medicines, vitamins, and herbal supplements.
How should I take LYVISPAH?
See the “Instructions for Use” at the end of this Patient Information leaflet for instructions on how to take or give LYVISPAH.- Take LYVISPAH exactly as your healthcare provider tells you to.
- Your healthcare provider will start you on a low dose of LYVISPAH and slowly increase your dose as needed.
- LYVISPAH can be taken the following different ways:
- LYVISPAH granules can be emptied into the mouth. You can let the granules dissolve in your mouth or you can swallow them. LYVISPAH can be taken with or without liquids or with soft foods.
- LYVISPAH granules from a packet can be mixed with a tablespoonful of liquid or soft foods like apple sauce, yogurt, or pudding. Take LYVISPAH within 2 hours of mixing.
- LYVISPAH can be given through feeding tubes.
See the Instructions for Use for more information.
- Do not stop taking LYVISPAH without talking to your healthcare provider first. Stopping LYVISPAH suddenly can cause serious problems such as severe muscle spasms, organ failure, and death. See “What are the possible side effects of LYVISPAH?”.
What should I avoid while taking LYVISPAH?
- Do not drive, operate dangerous machinery, or do any other dangerous activities until you know how LYVISPAH affects you. LYVISPAH can make you drowsy and less alert when you first start taking LYVISPAH and when your LYVISPAH dose is increased.
What are the possible side effects of LYVISPAH?
LYVISPAH can cause serious side effects, including:
- risk of symptoms from stopping LYVISPAH. Stopping LYVISPAH suddenly can cause severe muscle problems, organ failure, and death. Early symptoms include increased muscle stiffness, itching, and tingling in hands and feet. Other symptoms include seeing or hearing things that are not there (hallucinations), seizures, high fever, and confusion (altered mental status). Your healthcare provider will slowly reduce your dose of LYVISPAH when stopping treatment.
- withdrawal symptoms in infants (Neonatal Withdrawal Symptoms). LYVISPAH can cause withdrawal symptoms in infants after delivery. Symptoms of withdrawal of LYVISPAH in infants include stiffness in arms or legs, tremor, jitteriness, and seizure.
- drowsiness and sedation. If you take certain medicines or drink alcohol while taking LYVISPAH, your drowsiness may get worse. See “What should I avoid while taking LYVISPAH?” and “Before taking LYVISPAH, tell your healthcare provider about all of your medical conditions, including if you:”.
- decreased effectiveness. People who have had a stroke may not respond to or tolerate baclofen, the active ingredient in LYVISPAH.
- increase in symptoms of mental illness especially in people with psychotic disorders, schizophrenia, or other illness that may cause you to become confused.
- sudden increase in high blood pressure (autonomic dysreflexia).
- decrease in seizure control. People who have seizures (epilepsy) may have seizures more often than normal.
- changes in posture and balance. People who take LYVISPAH may have more problems standing or sitting up straight (posture) and keeping their balance.
- fluid filled sac on ovary (ovarian cysts). People may be at increased risk of developing ovarian cysts while taking LYVISPAH.
The most common side effects of LYVISPAH include drowsiness, dizziness, and weakness.
These are not all the side effects of LYVISPAH. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088 or Saol Therapeutics at 1-833-644-4216.
How should I store LYVISPAH?
Store LYVISPAH at room temperature between 68°F to 77°F (20°C to 25°C).General information about the safe and effective use of LYVISPAH.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use LYVISPAH for a condition for which it was not prescribed. Do not give LYVISPAH to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about LYVISPAH that is written for health professionals.
What are the ingredients in LYVISPAH?
Active ingredient: baclofen
Inactive ingredients: amino methacrylate copolymer, calcium stearate, colloidal silicon dioxide, crospovidone, hypromellose, mannitol, saccharin sodium, strawberry flavor, talc, and xylitol.
For more information visit www.lyvispah.com or contact Saol Therapeutics at 1-833-644-4216
-
INSTRUCTIONS FOR USE
INSTRUCTIONS FOR USE
LYVISPAH (lye vis' pah)
(baclofen)
oral granulesImportant information to know before taking LYVISPAH
LYVISPAH can be taken without liquids.
If needed, LYVISPAH can be mixed with liquids or soft foods. LYVISPAH should be taken within 2 hours of mixing into liquids or soft foods.
How to take LYVISPAH?
Opening LYVISPAH
Shake the packet to distribute settled granules to the bottom of the packet. Carefully open the LYVISPAH packet by cutting the dotted lines across the top of the packet (see the Figure).
Taking LYVISPAH by emptying the granules into the mouth
Empty all the granules in the LYVISPAH packet directly into your mouth. The granules will dissolve in your mouth or can be swallowed. After taking LYVISPAH, you can drink water if needed to swallow any granules left in your mouth.Taking LYVISPAH with liquids or soft foods
LYVISPAH can be taken or given by mouth as a mixture with liquids such as milk or apple juice. LYVISPAH can also be taken or given by mouth as a mixture with soft foods such as apple sauce, yogurt, or pudding.
- Open 1 packet of LYVISPAH (see the figure above).
- Empty the full contents of the LYVISPAH packet into 1 tablespoon (15 mL) liquid or soft food and mix it.
- Take LYVISPAH within 2 hours of mixing into liquids or soft foods
- If more than 1 packet of LYVISPAH is needed for your prescribed dose, mix each packet with a separate amount of liquid or soft food.
Giving LYVISPAH through a feeding tube
LYVISPAH can be given through enteral feeding tubes such as nasogastric (NG), gastrostomy (G), percutaneous endoscopic gastrostomy (PEG), and gastrojejunostomy (GJ) tubes.
- Flush the feeding tube with up to 1 tablespoon (15 mL) of water using a catheter tip syringe.
- Open and empty the full contents of 1 packet of LYVISPAH into a clean container and mix with 1 tablespoon (15 mL) of liquid (apple juice or milk).
- Stir the mixture to make sure all the granules are wet.
- Draw up the mixture of granules into a dosing syringe right away after stirring.
- Give the dose of LYVISPAH through the feeding tube within 2 hours after mixing. If the mixture is in the dosing syringe for 15 minutes and not given, turn the dosing syringe upside down 3 times before you give the dose.
- Fill the dosing syringe with 1 tablespoon (15 mL) of water and flush the feeding tube.
- If more than 1 packet of LYVISPAH is needed for the prescribed dose, mix each packet with a separate amount of liquid.
- Throw away (dispose of) any unused mixture.
Important Information
Take the entire packet of LYVISPAH to get the prescribed dose. Do not take only part of the medicine. Do not save a portion for later.
Keep LYVISPAH and all medicines out of the reach of children.
Distributed by: Saol Therapeutics, Inc. Roswell, GA
Part Code DI41601This Patient Information and Instructions for Use have been approved by the US Food and Drug Administration. Approved 11/2021
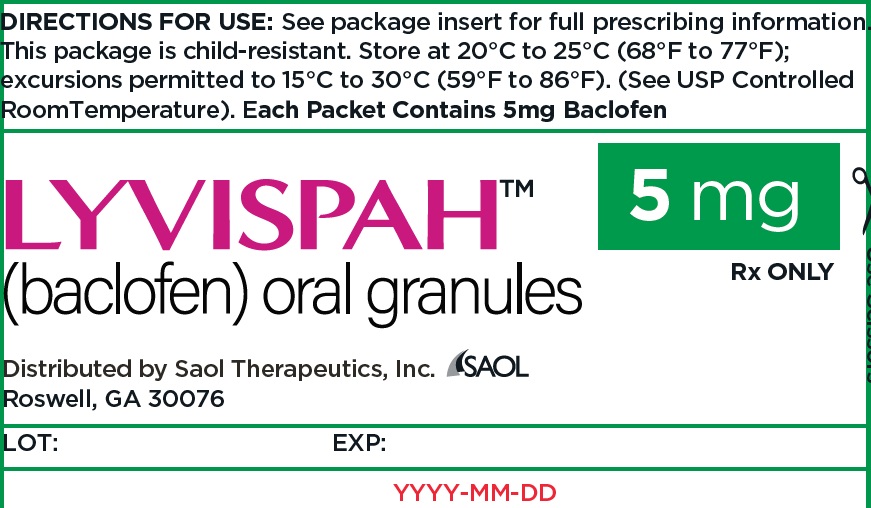
- PRINCIPAL DISPLAY PANEL - NDC: 70257-412-87 - 5mg Carton
- PRINCIPAL DISPLAY PANEL - NDC: 70257-412-87 - 5mg Container
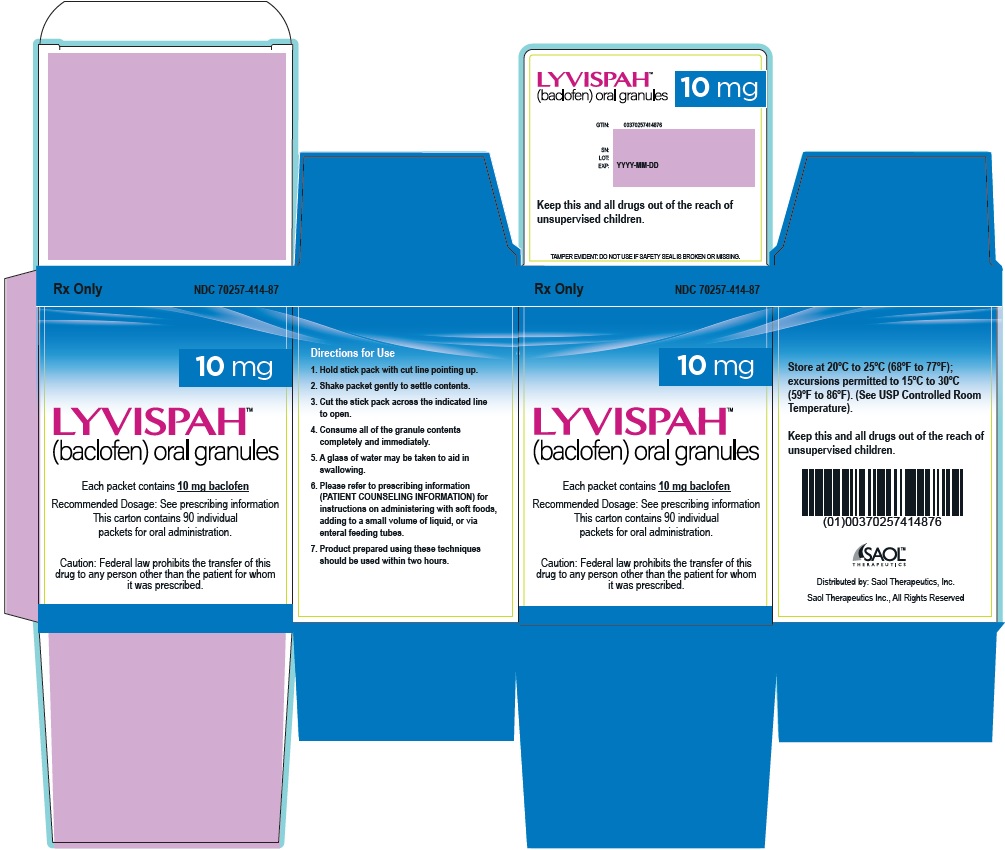
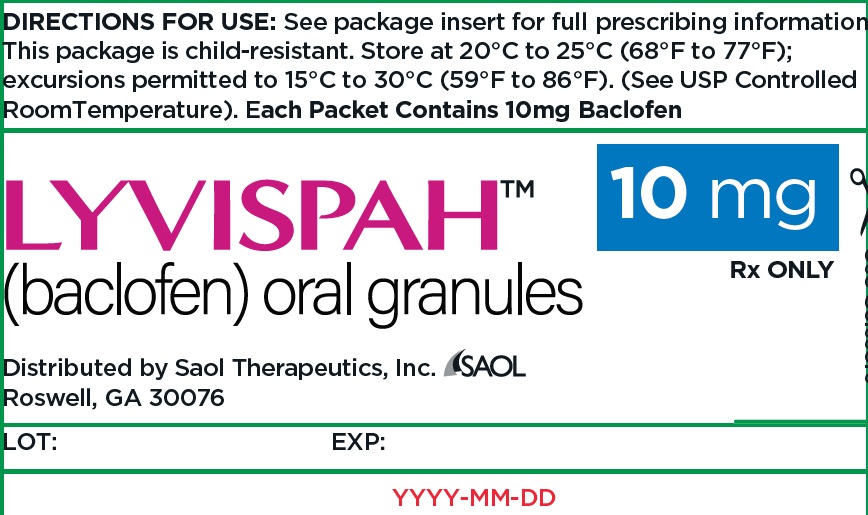
- PRINCIPAL DISPLAY PANEL - NDC: 70257-414-87 - 10mg Container
- PRINCIPAL DISPLAY PANEL - NDC: 70257-414-87 - 10mg Container
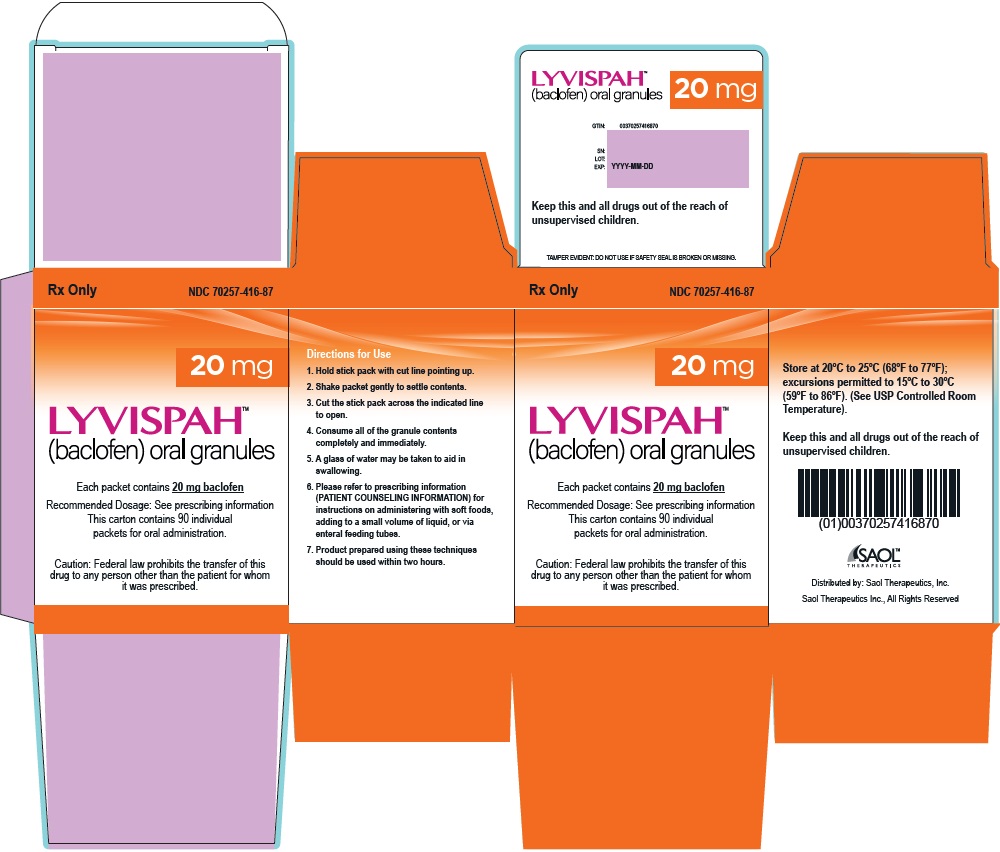
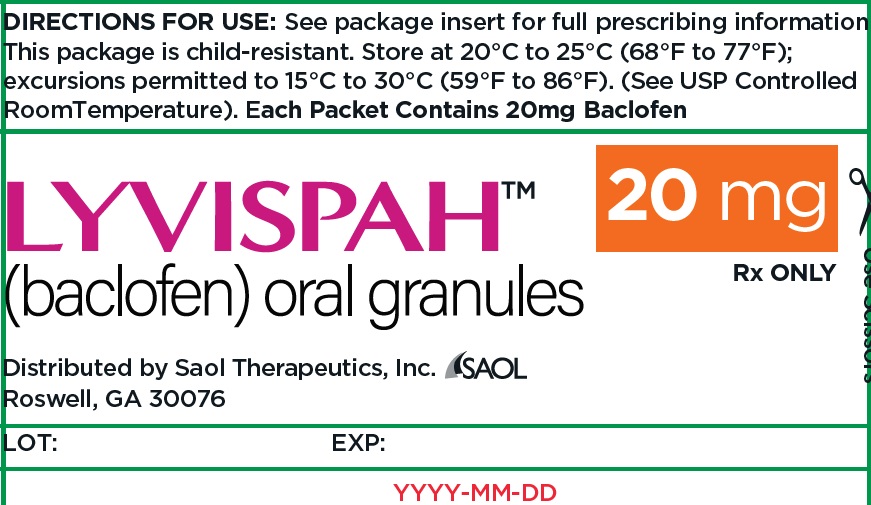
- PRINCIPAL DISPLAY PANEL - NDC: 70257-416-87 - 20mg Container
- PRINCIPAL DISPLAY PANEL - NDC: 70257-416-87 - 20mg Container
-
INGREDIENTS AND APPEARANCE
LYVISPAH
baclofen granuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70257-412 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACLOFEN (UNII: H789N3FKE8) (BACLOFEN - UNII:H789N3FKE8) BACLOFEN 5 mg Product Characteristics Color white (off-white) Score no score Shape FREEFORM (FREEFORM) Size Flavor STRAWBERRY (STRAWBERRY) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70257-412-87 90 in 1 BOTTLE, UNIT-DOSE 11/22/2021 1 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA215422 11/22/2021 LYVISPAH
baclofen granuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70257-414 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACLOFEN (UNII: H789N3FKE8) (BACLOFEN - UNII:H789N3FKE8) BACLOFEN 10 mg Product Characteristics Color white (off-white) Score no score Shape FREEFORM (FREEFORM) Size Flavor STRAWBERRY (STRAWBERRY) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70257-414-87 90 in 1 BOTTLE, UNIT-DOSE 11/22/2021 1 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA215422 11/22/2021 LYVISPAH
baclofen granuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70257-416 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACLOFEN (UNII: H789N3FKE8) (BACLOFEN - UNII:H789N3FKE8) BACLOFEN 20 mg Product Characteristics Color white (off-white) Score no score Shape FREEFORM (FREEFORM) Size Flavor STRAWBERRY (STRAWBERRY) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70257-416-87 90 in 1 BOTTLE, UNIT-DOSE 11/22/2021 1 1 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA215422 11/22/2021 Labeler - Saol Therapeutics Inc (080040201)