Label: PLEO FORT- penicillium roqueforti solution/ drops
-
Contains inactivated NDC Code(s)
NDC Code(s): 60681-1101-1 - Packager: Sanum-Kehlbeck GmbH & Co. KG
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated September 21, 2009
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Indications
- INGREDIENTS
- Tamper Evident
- DOSAGE
- WARNING
- STORAGE AND HANDLING
- SPL UNCLASSIFIED SECTION
-
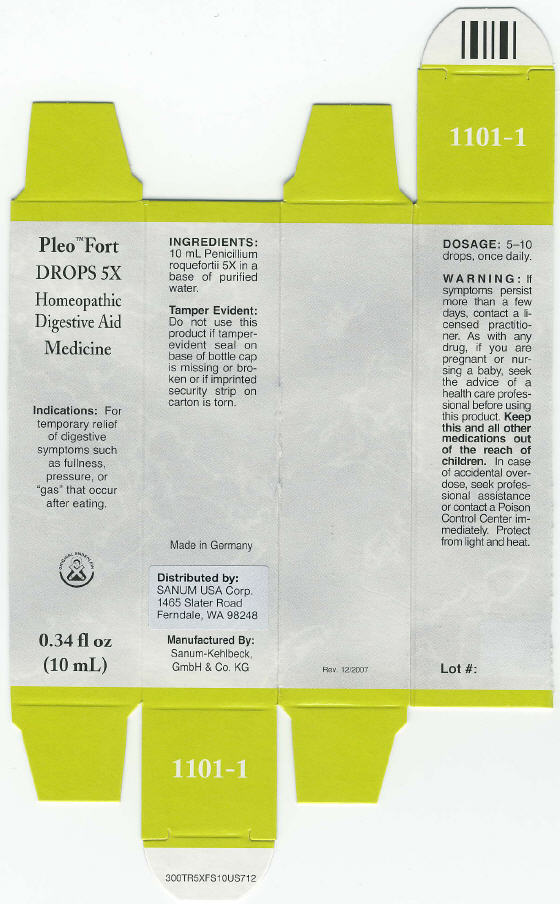
PRINCIPAL DISPLAY PANEL - 10 mL Label
Pleo™ Fort DROPS 5X
Homeopathic Digestive Aid Medicine
Indications: For temporary relief of digestive symptoms such as
fullness, pressure, or "gas" that occurs after eating.0.34 fl oz (10 mL)
DOSAGE: 5–10 drops, once daily. WARNING: If symptoms persist more than a few
days, contact a licensed practitioner. As with any drug, if you are pregnant or nursing
a baby, seek the advice of a health care professional before using this product. Keep
this and all medications out of the reach of children. In case of accidental overdose,
seek professional assistance or contact a Poison Control Center immediately. Protect
from light and heat. TAMPER EVIDENT: Do not use this product if tamper-evident
seal on base of bottle cap is missing or broken. INGREDIENTS: 10 mL Penicillium
roquefortii 5X in a base of purified water.
Rev. 06/2009
- PRINCIPAL DISPLAY PANEL - 10 mL box
-
INGREDIENTS AND APPEARANCE
PLEO FORT
penicillium roqueforti solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:60681-1101 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength penicillium roqueforti (UNII: 7ORP6R724L) (penicillium roqueforti - UNII:7ORP6R724L) penicillium roqueforti 5 [HP_X] in 10 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60681-1101-1 1 in 1 CARTON 1 10 mL in 1 BOTTLE, DROPPER Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved homeopathic 03/19/1996 Labeler - Sanum-Kehlbeck GmbH & Co. KG (318386133)