Label: WORKFORCE PERSONAL CARE HAND SANITIZER LEMON TEA TREE- ethyl alcohol hand sanitizer gel
- NDC Code(s): 71824-7900-1, 71824-7902-1, 71824-7908-1, 71824-7916-1
- Packager: Prima Fleur Botanicals, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated February 1, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
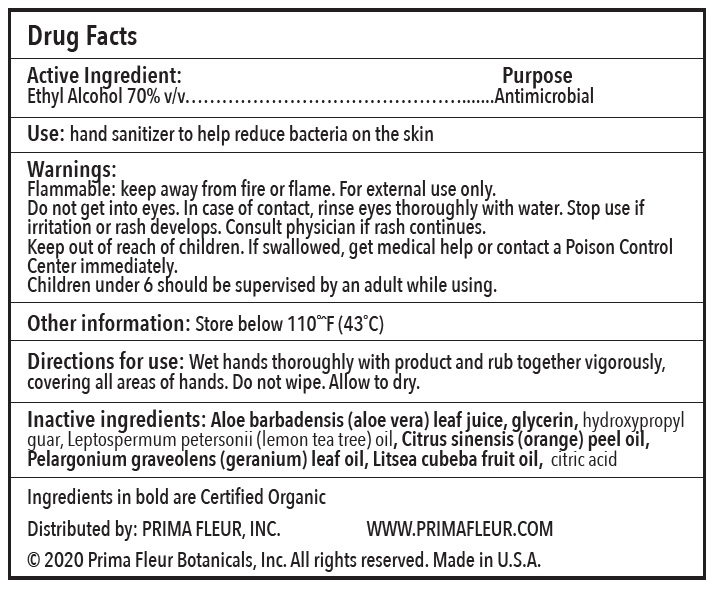
- Official Label (Printer Friendly)
- Directions for Use
- Warnings
- Inactive Ingredients
- Use
- Warnings:
- Use
- ACTIVE INGREDIENT
- Workforce Personal Care Hand Sanitizer Lemon Tea Tree
- 8oz Lemon Tea Tree Label
- Lemon Tea Tree 16oz front label
- Lemon Tea Tree 128oz (gallon) front label
-
INGREDIENTS AND APPEARANCE
WORKFORCE PERSONAL CARE HAND SANITIZER LEMON TEA TREE
ethyl alcohol hand sanitizer gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71824-7916 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 331.2232 mL in 473.176 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) PELARGONIUM GRAVEOLENS FLOWER OIL (UNII: 3K0J1S7QGC) HYDROXYPROPYL GUAR (2500-4500 MPA.S AT 1%) (UNII: 3A1I7376TC) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) LITSEA CUBEBA FRUIT (UNII: R3617MQA5Z) ORANGE OIL, DISTILLED (UNII: H4QNH2ZN7A) LEPTOSPERMUM PETERSONII LEAF OIL (UNII: N37UWG52T3) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71824-7916-1 1 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/21/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 05/21/2020 WORKFORCE PERSONAL CARE HAND SANITIZER LEMON TEA TREE
ethyl alcohol hand sanitizer gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71824-7902 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 41.40297 mL in 59.1471 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) PELARGONIUM GRAVEOLENS FLOWER OIL (UNII: 3K0J1S7QGC) HYDROXYPROPYL GUAR (2500-4500 MPA.S AT 1%) (UNII: 3A1I7376TC) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) LITSEA CUBEBA FRUIT (UNII: R3617MQA5Z) ORANGE OIL, DISTILLED (UNII: H4QNH2ZN7A) LEPTOSPERMUM PETERSONII LEAF OIL (UNII: N37UWG52T3) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71824-7902-1 1 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/21/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 05/21/2020 WORKFORCE PERSONAL CARE HAND SANITIZER LEMON TEA TREE
ethyl alcohol hand sanitizer gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71824-7900 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 2649.787 mL in 3785.41 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) PELARGONIUM GRAVEOLENS FLOWER OIL (UNII: 3K0J1S7QGC) HYDROXYPROPYL GUAR (2500-4500 MPA.S AT 1%) (UNII: 3A1I7376TC) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) LITSEA CUBEBA FRUIT (UNII: R3617MQA5Z) ORANGE OIL, DISTILLED (UNII: H4QNH2ZN7A) LEPTOSPERMUM PETERSONII LEAF OIL (UNII: N37UWG52T3) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71824-7900-1 1 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/21/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 05/21/2020 WORKFORCE PERSONAL CARE HAND SANITIZER LEMON TEA TREE
ethyl alcohol hand sanitizer gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71824-7908 Route of Administration CUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 165.6116 mL in 236.588 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) PELARGONIUM GRAVEOLENS FLOWER OIL (UNII: 3K0J1S7QGC) HYDROXYPROPYL GUAR (2500-4500 MPA.S AT 1%) (UNII: 3A1I7376TC) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) LITSEA CUBEBA FRUIT (UNII: R3617MQA5Z) ORANGE OIL, DISTILLED (UNII: H4QNH2ZN7A) LEPTOSPERMUM PETERSONII LEAF OIL (UNII: N37UWG52T3) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71824-7908-1 1 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 05/21/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 05/21/2020 Labeler - Prima Fleur Botanicals, Inc. (825058308) Registrant - Prima Fleur Botanicals, Inc. (825058308) Establishment Name Address ID/FEI Business Operations Prima Fleur Botanicals, Inc. 825058308 manufacture(71824-7908, 71824-7916, 71824-7902, 71824-7900)