Label: EIGHT HOUR CREAM NOURISHING LIP BALM SPF 20- octinoxate and avobenzone salve
-
Contains inactivated NDC Code(s)
NDC Code(s): 67938-1111-1, 67938-1111-2 - Packager: Elizabeth Arden, Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 2, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DESCRIPTION
- INDICATIONS AND USAGE
-
WARNINGS
WARNING
:
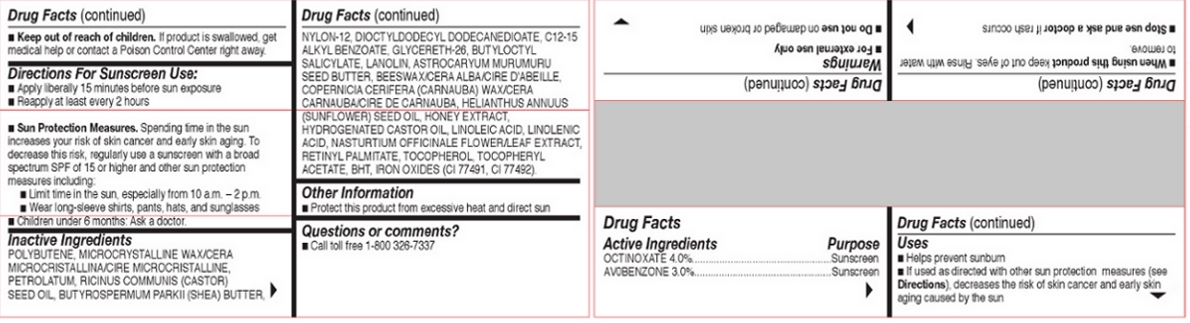
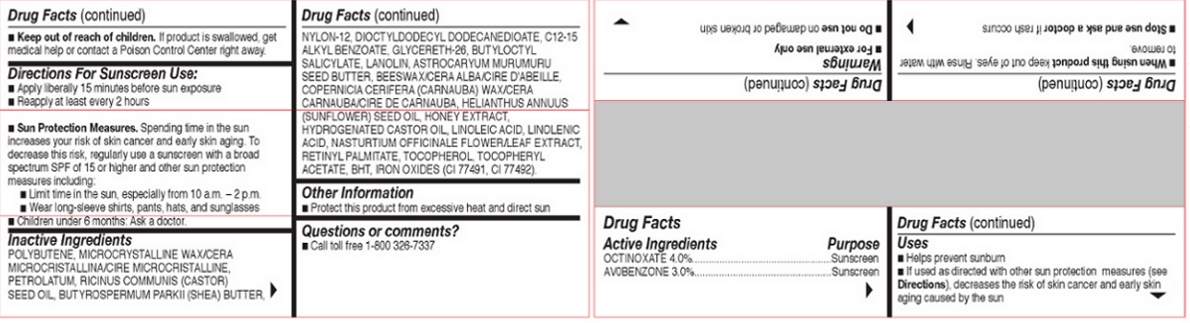
FOR EXTERNAL USE ONLY. DO NOT USEON DAMAGED OR BROKEN SKIN. WHEN USING
THIS PRODUCT KEEP OUT OF EYES. RINSE WITH
WATER TO REMOVE. STOP USE AND ASK A
DOCTOR IF RASH OCCURS. KEEP OUT OF REACH
OF CHILDREN. IF PRODUCT IS SWALLOWED, GET
MEDICAL HELP OR CONTACT A POISON CONTROL
CENTER RIGHT AWAY.
- OTC - ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Inactive Ingredients
POLYBUTENE, MICROCRYSTALLINE WAX/CERA
MICROCRISTALLINA/CIRE MICROCRISTALLINE,
PETROLATUM, RICINUS COMMUNIS (CASTOR)
SEED OIL, BUTYROSPERMUM PARKII (SHEA) BUTTER,
NYLON-12, DIOCTYLDODECYL DODECANEDIOATE, C12-15
ALKYL BENZOATE, GLYCERETH-26, BUTYLOCTYL
SALICYLATE, LANOLIN, ASTROCARYUM MURUMURU
SEED BUTTER, BEESWAX/CERA ALBA/CIRE D'ABEILLE,
COPERNICIA CERIFERA (CARNAUBA) WAX/CERA
CARNAUBA/CIRE DE CARNAUBA, HELIANTHUS ANNUUS
(SUNFLOWER) SEED OIL, HONEY EXTRACT,
HYDROGENATED CASTOR OIL, LINOLEIC ACID, LINOLENIC
ACID, NASTURTIUM OFFICINALE FLOWER/LEAF EXTRACT,
RETINYL PALMITATE, TOCOPHEROL, TOCOPHERYL
ACETATE, BHT, IRON OXIDES (CI 77491, CI 77492).
- DOSAGE & ADMINISTRATION
- OTC - KEEP OUT OF REACH OF CHILDREN
- OTC - PURPOSE
- OTC - WHEN USING
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EIGHT HOUR CREAM NOURISHING LIP BALM SPF 20
octinoxate and avobenzone salveProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:67938-1111 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 0.568 g in 14.2 g AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 0.426 g in 14.2 g Inactive Ingredients Ingredient Name Strength POLYBUTENE (1400 MW) (UNII: 1NA5AO9GH7) MICROCRYSTALLINE WAX (UNII: XOF597Q3KY) PETROLATUM (UNII: 4T6H12BN9U) NYLON-12 (UNII: 446U8J075B) DIOCTYLDODECYL DODECANEDIOATE (UNII: 7QDB5EE79Y) GLYCERETH-26 (UNII: NNE56F2N14) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) LANOLIN (UNII: 7EV65EAW6H) ASTROCARYUM MURUMURU SEED BUTTER (UNII: 12V64UPU6R) HYDROGENATED CASTOR OIL (UNII: ZF94AP8MEY) LINOLEIC ACID (UNII: 9KJL21T0QJ) LINOLENIC ACID (UNII: 0RBV727H71) TOCOPHEROL (UNII: R0ZB2556P8) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67938-1111-1 1 in 1 BOX 1 NDC:67938-1111-2 14.2 g in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part352 07/24/2012 Labeler - Elizabeth Arden, Inc (849222187)