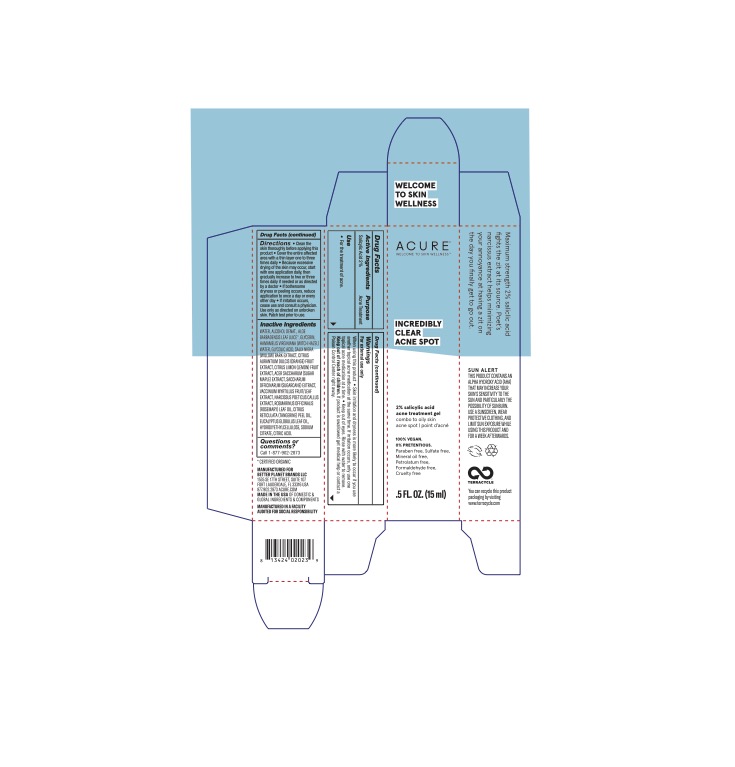
Label: INCREDIBLY CLEAR ACNE SPOT- salicylic acid gel
- NDC Code(s): 54136-118-91
- Packager: Better Planet Brands LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 21, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- WHEN USING
- KEEP OUT OF REACH OF CHILDREN
-
Directions
Directions * Clean the skin before applying this product * Cover the entire affected area with a thin layer one to three times daily * Because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor * If bothersome dryness or peeling occurs, reduce applications to once a day or every other day * If irritation occurs, cease use and consult a physician. Use only as directed on unbroken skin. Patch test prior to use.
-
Inactive Ingredients
Inactive Ingredients
WATER, ALCOHOL DENAT., ALOE BARBADENSIS LEAF JUICE*, GLYCERIN, HAMAMELIS VIRGINIANA (WITCH HAZEL) WATER, GLYCOLIC ACID, SALIX NIGRA (WILLOW) BARK EXTRACT, CITRUS AURANTIUM DULCIS (ORANGE) FRUIT EXTRACT, CITRUS LIMON (LEMON) FRUIT EXTRACT, ACER SACCHARUM (SUGAR MAPLE) EXTRACT, SACCHARUM OFFICINARUM (SUGARCANE) EXTRACT. VACCINIUM MYRTILLUS FRUIT/LEAF EXTRACT. NARCISSUS POETICS CALLUS EXTRACT. ROSMARINUS OFFICINALIS (ROSEMARY) LEAF OIL, CITRUS RETICULATA (TANGERINE) PEEL OIL EUCALYPTUS GLOBULUS LEAF OIL. HYDROXYETHYLCELLULOSE. SODIUM CITRATE, CITRIC ACID. *Organic Ingredient
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
INCREDIBLY CLEAR ACNE SPOT
salicylic acid gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54136-118 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 2 g in 100 g Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) SALIX NIGRA BARK (UNII: QU52J3A5B3) VACCINIUM MYRTILLUS FRUITING TOP (UNII: QT6362QVN8) MANDARIN OIL (UNII: NJO720F72R) WATER (UNII: 059QF0KO0R) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) GLYCERIN (UNII: PDC6A3C0OX) SUGARCANE (UNII: 81H2R5AOH3) ORANGE (UNII: 5EVU04N5QU) ALOE VERA LEAF (UNII: ZY81Z83H0X) ACER SACCHARUM BARK/SAP (UNII: Z120VL0KAC) HYDROXYETHYL CELLULOSE (4000 MPA.S AT 1%) (UNII: ZYD53NBL45) GLYCOLIC ACID (UNII: 0WT12SX38S) EUCALYPTUS OIL (UNII: 2R04ONI662) SODIUM CITRATE (UNII: 1Q73Q2JULR) LEMON (UNII: 24RS0A988O) HAMAMELIS VIRGINIANA LEAF WATER (UNII: 8FP93ED6H2) ROSEMARY OIL (UNII: 8LGU7VM393) NARCISSUS POETICUS FLOWER OIL (UNII: 449GS37I8P) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54136-118-91 1 in 1 CARTON 05/14/2020 1 15 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M006 05/14/2020 Labeler - Better Planet Brands LLC (960253719)