Label: VISIONBLUE- trypan blue injection, solution
- NDC Code(s): 68803-612-10
- Packager: D.O.R.C. Dutch Ophthalmic Research Center (International) B.V.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated October 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
VisionBlue® 0.06% Trypan blue Ophthalmic Solution. These highlights do not include all the information needed to use VISIONBLUE® 0.06% safely and effectively. see full prescribing information for VISIONBLUE® 0.06%. VisionBlue® (trypan blue ophthalmic solution) 0.06% Initial U.S. Approval: 2004
INDICATIONS AND USAGE
HIGHLIGHTS OF PRESCRIBING INFORMATION (1)
These highlights do not include all the information needed to use VISIONBLUE® 0.06% safely and effectively. see full prescribing information for VISIONBLUE® 0.06%. VisionBlue® (trypan blue ophthalmic solution) 0.06% Initial U.S. Approval: 2004 (1)
(1)
VisionBlue® 0.06% is an acid di-azo group dye indicated for use as an aid in ophthalmic surgery by staining the anterior capsule of the lens. (1)
DOSAGE AND ADMINISTRATION
- Inject an air bubble into the anterior chamber;
- Carefully apply VisionBlue® 0.06% onto the lens capsule;
- Remove all excess dye from the anterior chamber.
DOSAGE FORMS AND STRENGTHS
VisionBlue® (trypan blue ophthalmic solution) 0.06% in a volume of 0.5 mL. (3)
CONTRAINDICATIONS
Insertion of a non-hydrated (dry-state), hydrophilic acrylic intraocular lens (IOL). (4)
WARNINGS AND PRECAUTIONS
Excessive staining: Excess VisionBlue® 0.06% should be removed from the eye immediately after staining. (5)
ADVERSE REACTIONS
USE IN SPECIFIC POPULATIONS
Trypan blue should not be given to pregnant women. (7)
Revised: 12/2012
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
VisionBlue 0.06% - Indications & Usage Section
VisionBlue 0.06% - Dosage & Administration Section
VisionBlue 0.06% - Dosage forms & Strengths section
VisionBlue 0.06% - Contraindications section
VisionBlue 0.06% - Warnings and Precautions section
VisionBlue 0.06% - Adverse Reactions section
VisionBlue 0.06% - Use in specific populations section
VisionBlue 0.06% - Pregnancy section
VisionBlue 0.06% - Nursing Mothers section
VisionBlue 0.06% - Pediatric use section
VisionBlue 0.06% - Geriatric use section
VisionBlue 0.06% - Description section
VisionBlue 0.06% - Clinical Pharmacology section
VisionBlue 0.06% - Mechanism of action section
VisionBlue 0.06% - Nonclinical toxicology section
VisionBlue 0.06% - Carcinogenesis & Mutagenesis & Impairment of fertility section
VisionBlue 0.06% - How supplied section
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- VisionBlue 0.06% - Indications & Usage Section
-
VisionBlue 0.06% - Dosage & Administration Section
Cataract surgery.
VisionBlue® 0.06% is packaged in a 2.25 mL syringe to which a blunt cannula has to be attached.
After opening the eye, an air bubble is injected into the anterior chamber of the eye in order to minimize dilution of VisionBlue® 0.06% by the aqeous. VisionBlue® 0.06% is carefully applied onto the anterior lens capsule using a blunt cannula. Sufficient staining is achieved as soon as the dye has contacted the capsule. The anterior chamber is then irrigated with balanced salt solution to remove all excess dye. An anterior capsulotomy can then be performed.
- VisionBlue 0.06% - Dosage forms & Strengths section
- VisionBlue 0.06% - Contraindications section
- VisionBlue 0.06% - Warnings and Precautions section
-
VisionBlue 0.06% - Adverse Reactions section
Adverse reactions reported following use of VisionBlue® 0.06% include discoloration of high water content hydrogen intraocular lenses (see contraindications) and inadvertent staining of the posterior lens capsule and vitreous face. Staining of the posterior lens capsule or staining of the vitreous face is generally self limited, lasting up to one week.
-
VisionBlue 0.06% - Use in specific populations section
VisionBlue 0.06% - Pregnancy section
Teratogenic Effects: Pregnancy Category C. Trypan blue is teratogenic in rats, mice, rabbits, hamsters, dogs, guinea pigs, pigs, and chickens. The majority of teratogenicity studies performed involve intravenous, intraperitoneal, or subcutaneous administration in the rat. The teratogenic dose is 50 mg/kg as a single dose or 25 mg/kg/day during embryogenesis in the rat. These doses are approximately 10,000- and 5,000-fold the maximum recommended human dose of 0.3 mg per injection based in a 60 kg person, assuming that the whole dose is completely absorbed. Characteristic anomalies included neural tube, cardiovascular, vertebral, tail, and eye defects. Trypan blue also caused an increase in post-implantation mortality, and decreased fetal weight. In the monkey, trypan blue caused abortions with single or two daily doses of 50 mg/kg between 20th to 25th days of pregnancy, but no apparent increase in birth defects (approximately 10,000-fold the maximum recommended human daily dose of 0.3 mg per injection, assuming total absorption). There are no adequate and well-controlled studies in pregnant women. Trypan blue should be given to a pregnant woman only if the potential benefit justifies the potential risk to the fetus.
VisionBlue 0.06% - Nursing Mothers section
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when trypan blue is administered to a nursing woman.
-
VisionBlue 0.06% - Description section
VisionBlue® (trypan blue ophthalmic solution) 0.06% is a sterile solution of trypan blue (an acid di-azo group dye). VisionBlue® 0.06% is a selective tissue staining agent for use as a medical aid in ophthalmic surgery.
Each mL of VisionBlue® 0.06% contains: 0.6 mg trypan blue; 1.9 mg sodium mono-hydrogen orthophosphate (Na 2HPO 4·2H 2O); 0.3 mg sodium di-hydrogen orthophosphate (NaH 2PO 4·2H 2O); 8.2 mg sodium chloride (NaCl); and water for injection. The pH is 7.3 – 7.6. the osmolality is 257-314 mOsm/kg.
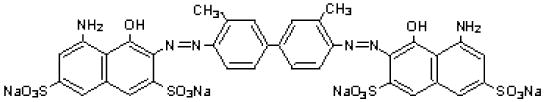
The drug substance trypan blue has the chemical name 3,3’-[(3,3’-dimethyl-4,4’-biphenylylene) bis (azo)] bis (5-amino-4-hydroxy-2,7-naphthalenedisulfonic acid) tetra sodium salt, a molecular weight of 960.8, a molecular formula of C 34H 24N 6Na 4O 14S 4, and has the following chemical structure:
-
VisionBlue 0.06% - Clinical Pharmacology section
VisionBlue 0.06% - Mechanism of action section
VisionBlue® 0.06% selectively stains connective tissue structures in the human eye such as the anterior lens capsule of the human crystalline lens.
VisionBlue® 0.06% is intended to be applied directly on the anterior lens capsule, staining any portion of the capsule which comes in contact with the dye. Excess dye is washed out of the anterior chamber. The dye does not penetrate the capsule, permitting visualization of the anterior capsule in contrast to the non-stained lens cortex and inner lens material.
-
VisionBlue 0.06% - Nonclinical toxicology section
VisionBlue 0.06% - Carcinogenesis & Mutagenesis & Impairment of fertility section
Trypan blue is carcinogenic in rats. Wistar/Lewis rats developed lymphomas after receiving subcutaneous injections of 1% trypan blue dosed at 50 mg/kg every other week for 52 weeks (total does approximately 250,000-fold the maximum recommended human dose of 0.3 mg per injection in a 60 kg person, assuming total absorption).
Trypan blue was mutagenic in the Ames test and caused DNA strand breaks in vitro.
- VisionBlue 0.06% - How supplied section
- VisionBlue 0.06% - Storage and Handling section
- VisionBlue 0.06% - Patient counseling information
-
SPL UNCLASSIFIED SECTION
Manufactured by
D.O.R.C. International b.v.
Scheijdelveweg 2
3214 VN Zuidland
The Netherlands
Distributed in the United States by
Dutch Ophthalmic, USA
10 Continental Drive, Bldg 1
Exeter, NH 03833, USA
Phone: 800-75-DUTCH of 603-778-6929
US Patents 6,367,480 and 6,720,314
Copyright ©, 2012 Dutch Ophthalmic Research Center
- VisionBlue 0.06% - Package label.Principal display panel
-
INGREDIENTS AND APPEARANCE
VISIONBLUE
trypan blue injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:68803-612 Route of Administration OPHTHALMIC, INTRAOCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRYPAN BLUE (UNII: I2ZWO3LS3M) (TRYPAN BLUE FREE ACID - UNII:768N7QO4KH) TRYPAN BLUE 0.3 mg in 0.5 mL Inactive Ingredients Ingredient Name Strength SODIUM PHOSPHATE, MONOBASIC, DIHYDRATE (UNII: 5QWK665956) 0.15 mg in 0.5 mL SODIUM PHOSPHATE, DIBASIC, DIHYDRATE (UNII: 94255I6E2T) 0.85 mg in 0.5 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 4.1 mg in 0.5 mL WATER (UNII: 059QF0KO0R) 0.5 mL in 0.5 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68803-612-10 10 in 1 CARTON 12/16/2004 1 1 in 1 POUCH 1 0.5 mL in 1 SYRINGE, GLASS; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021670 12/16/2004 Labeler - D.O.R.C. Dutch Ophthalmic Research Center (International) B.V. (407522184) Registrant - D.O.R.C. Dutch Ophthalmic Research Center (International) B.V. (407522184) Establishment Name Address ID/FEI Business Operations Pharmpur GmbH 340805167 manufacture(68803-612)






