Label: JISBERRY ANTIBACTERIAL HANDLOTION- triclosan lotion
-
Contains inactivated NDC Code(s)
NDC Code(s): 52738-005-01 - Packager: Hangzhou Xizi Sanitary Disinfection Medical Equipment Co., Ltd
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated April 26, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
-
INDICATIONS & USAGE
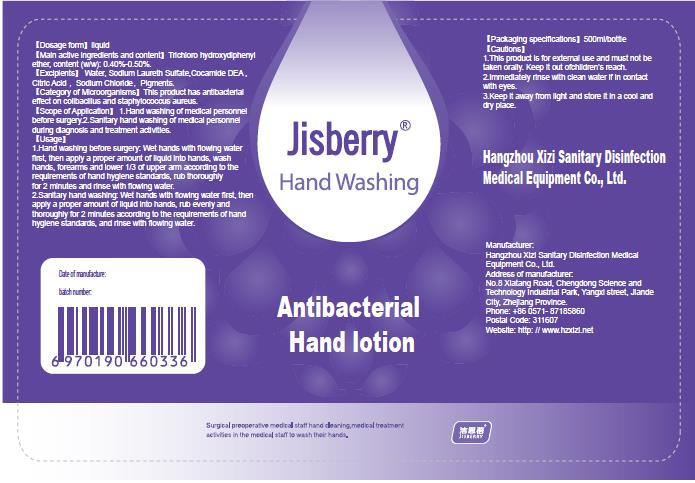
1.Hand washing before surgery: Wet hands with flowing water first, then apply a proper amount of liquid into hands, wash hands, forearms and lower 1/3 of upper arm according to the requirements of hand hygiene standards, rub thoroughly for 2 minutes and rinse with flowing water.
2.Sanitary hand washing: Wet hands with flowing water first, then apply a proper amount of liquid into hands, rub evenly and thoroughly for 2 minutes according to the requirements of hand hygiene standards, and rinse with flowing water. - ACTIVE INGREDIENT
- KEEP OUT OF REACH OF CHILDREN
- PURPOSE
- WARNINGS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
JISBERRY ANTIBACTERIAL HANDLOTION
triclosan lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52738-005 Route of Administration EXTRACORPOREAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TRICLOSAN (UNII: 4NM5039Y5X) (TRICLOSAN - UNII:4NM5039Y5X) TRICLOSAN 2 g in 500 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM LAURETH SULFATE (UNII: BPV390UAP0) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM CHLORIDE (UNII: 451W47IQ8X) CHLOROPHYLL (UNII: 00WNZ48OR9) COCO DIETHANOLAMIDE (UNII: 92005F972D) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52738-005-01 500 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/12/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 04/12/2020 Labeler - Hangzhou Xizi Sanitary Disinfection Medical Equipment Co., Ltd (527389022) Establishment Name Address ID/FEI Business Operations Hangzhou Xizi Sanitary Disinfection Medical Equipment Co., Ltd 527389022 manufacture(52738-005)