Label: ANTIBACTERIAL WIPES- benzalkonium chloride cloth
-
NDC Code(s):
76635-001-01,
76635-001-02,
76635-001-03,
76635-001-04, view more76635-001-05, 76635-001-06, 76635-001-07, 76635-001-08, 76635-001-09, 76635-001-10, 76635-001-11, 76635-001-12
- Packager: Hangzhou Glamcos Biotech CO.,LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 23, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient(s)
- Purpose
- Use
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- Directions
- Inactive ingredients
- 100 Package Label - Principal Display Panel
- 80 Package Label - Principal Display Panel
- 40 Package Label - Principal Display Panel
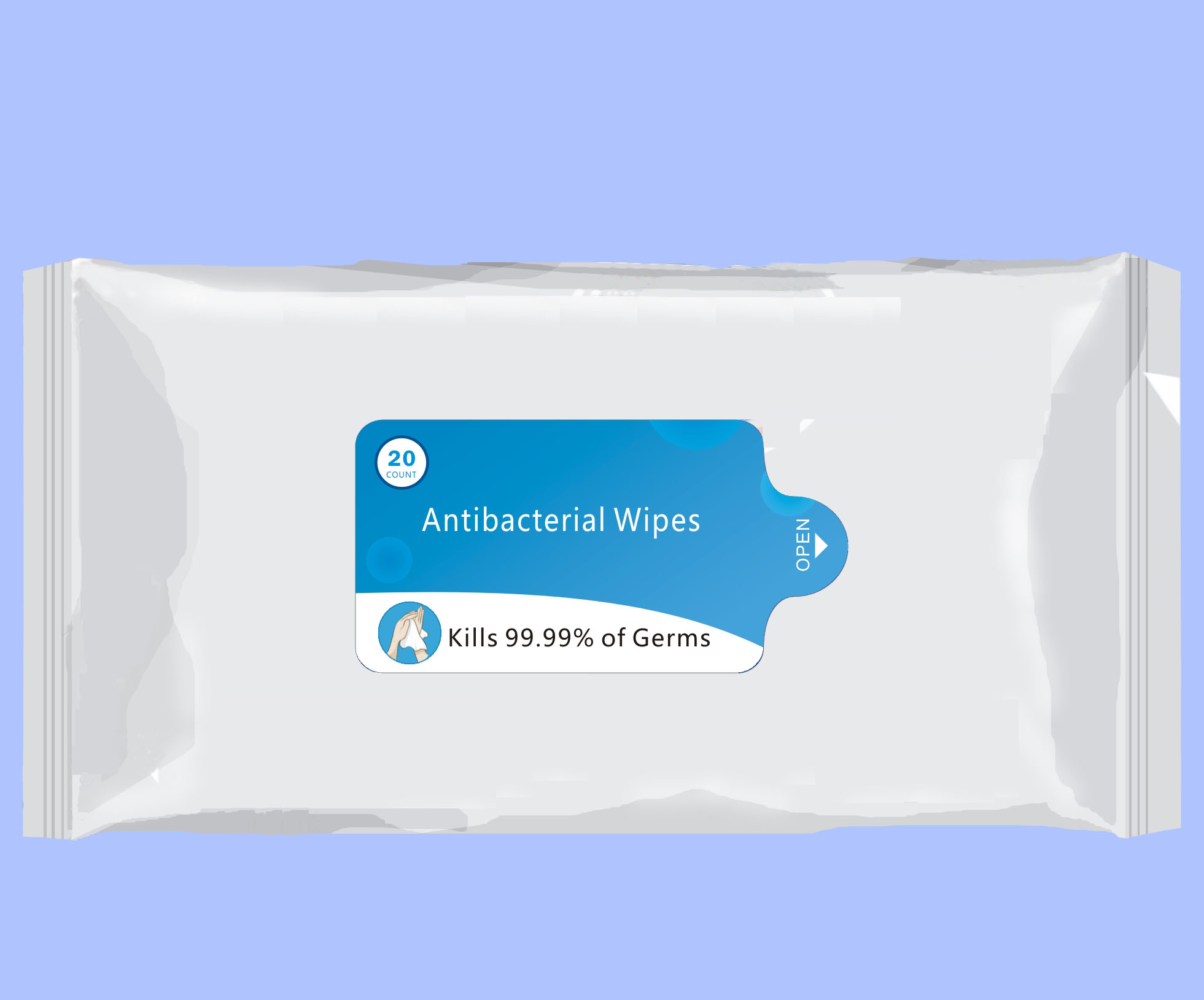
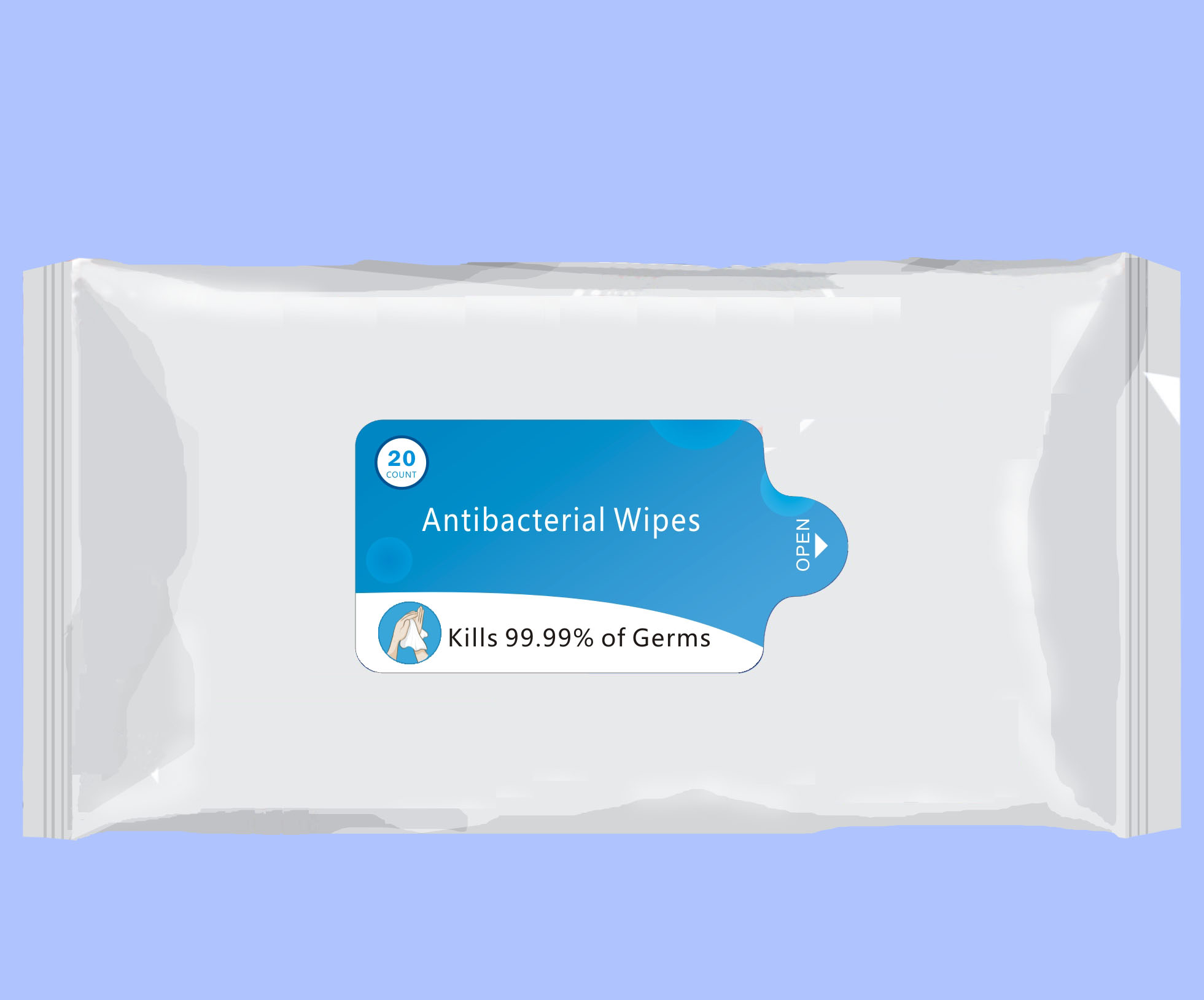
- 20 Package Label - Principal Display Panel
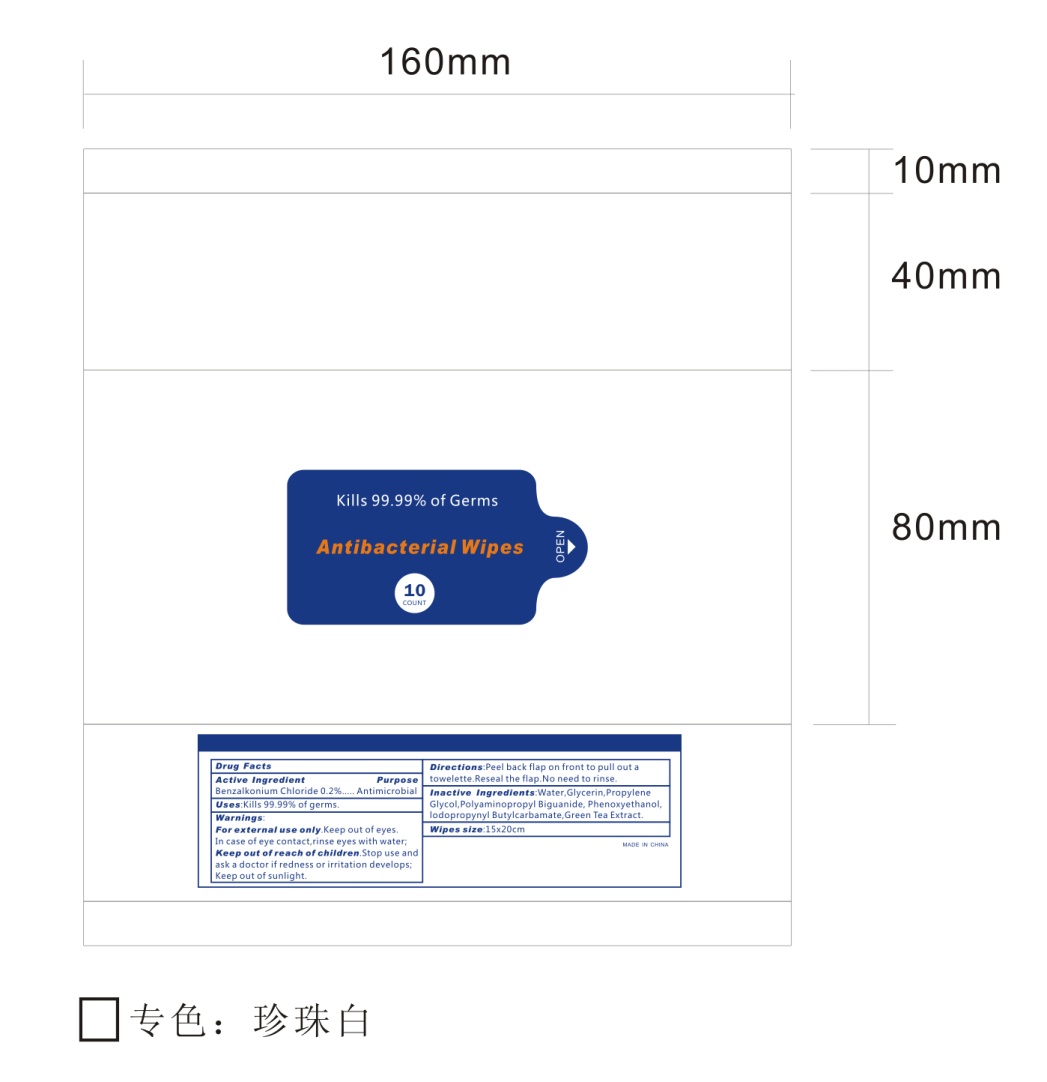
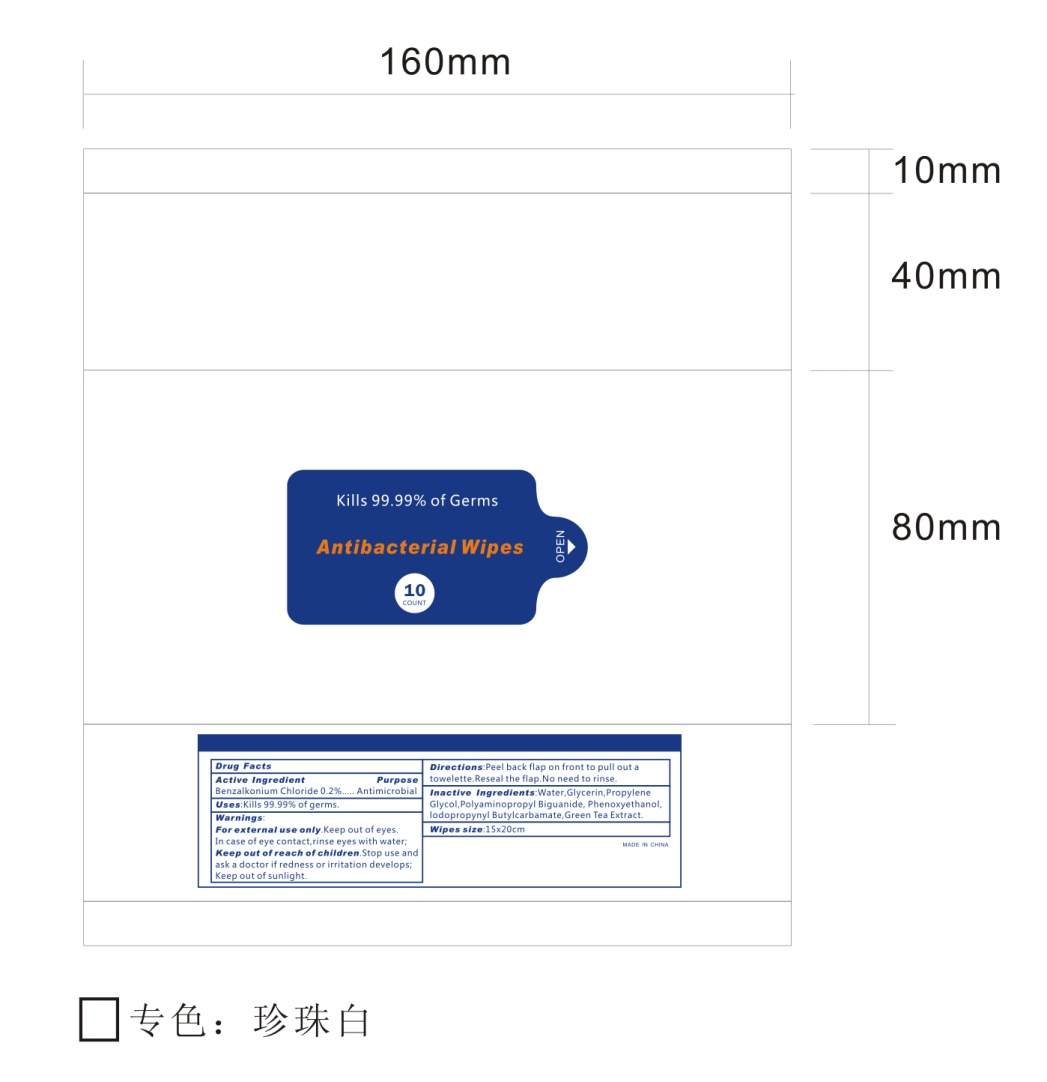
- 10 Package Label - Principal Display Panel
- 400 Pail Label - Principal Display Panel
- 1 Package Label - Principal Display Panel
- 70 Package Label - Principal Display Panel
- 250 Pail Package Label - Principal Display Panel
- 100 Pail Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ANTIBACTERIAL WIPES
benzalkonium chloride clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76635-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.2 g in 100 g Inactive Ingredients Ingredient Name Strength POLYAMINOPROPYL BIGUANIDE (UNII: DT9D8Z79ET) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) GREEN TEA LEAF (UNII: W2ZU1RY8B0) PHENOXYETHANOL (UNII: HIE492ZZ3T) IODOPROPYNYL BUTYLCARBAMATE (UNII: 603P14DHEB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76635-001-01 432 g in 1 BAG; Type 0: Not a Combination Product 05/01/2020 2 NDC:76635-001-02 346 g in 1 BAG; Type 0: Not a Combination Product 05/01/2020 3 NDC:76635-001-03 173 g in 1 BAG; Type 0: Not a Combination Product 05/01/2020 4 NDC:76635-001-04 87 g in 1 BAG; Type 0: Not a Combination Product 05/01/2020 5 NDC:76635-001-05 47 g in 1 BAG; Type 0: Not a Combination Product 05/10/2020 6 NDC:76635-001-06 1700 g in 1 PAIL; Type 0: Not a Combination Product 05/10/2020 7 NDC:76635-001-07 1700 g in 1 PAIL; Type 0: Not a Combination Product 05/28/2020 8 NDC:76635-001-08 4.3 g in 1 BAG; Type 0: Not a Combination Product 07/25/2020 9 NDC:76635-001-09 1700 g in 1 PAIL; Type 0: Not a Combination Product 08/10/2020 10 NDC:76635-001-10 303 g in 1 BAG; Type 0: Not a Combination Product 08/29/2020 11 NDC:76635-001-11 1215 g in 1 PAIL; Type 0: Not a Combination Product 10/08/2020 12 NDC:76635-001-12 480 g in 1 PAIL; Type 0: Not a Combination Product 10/08/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 05/01/2020 Labeler - Hangzhou Glamcos Biotech CO.,LTD (554476017) Registrant - Hangzhou Glamcos Biotech CO.,LTD (554476017) Establishment Name Address ID/FEI Business Operations Hangzhou Glamcos Biotech CO.,LTD 554476017 manufacture(76635-001)