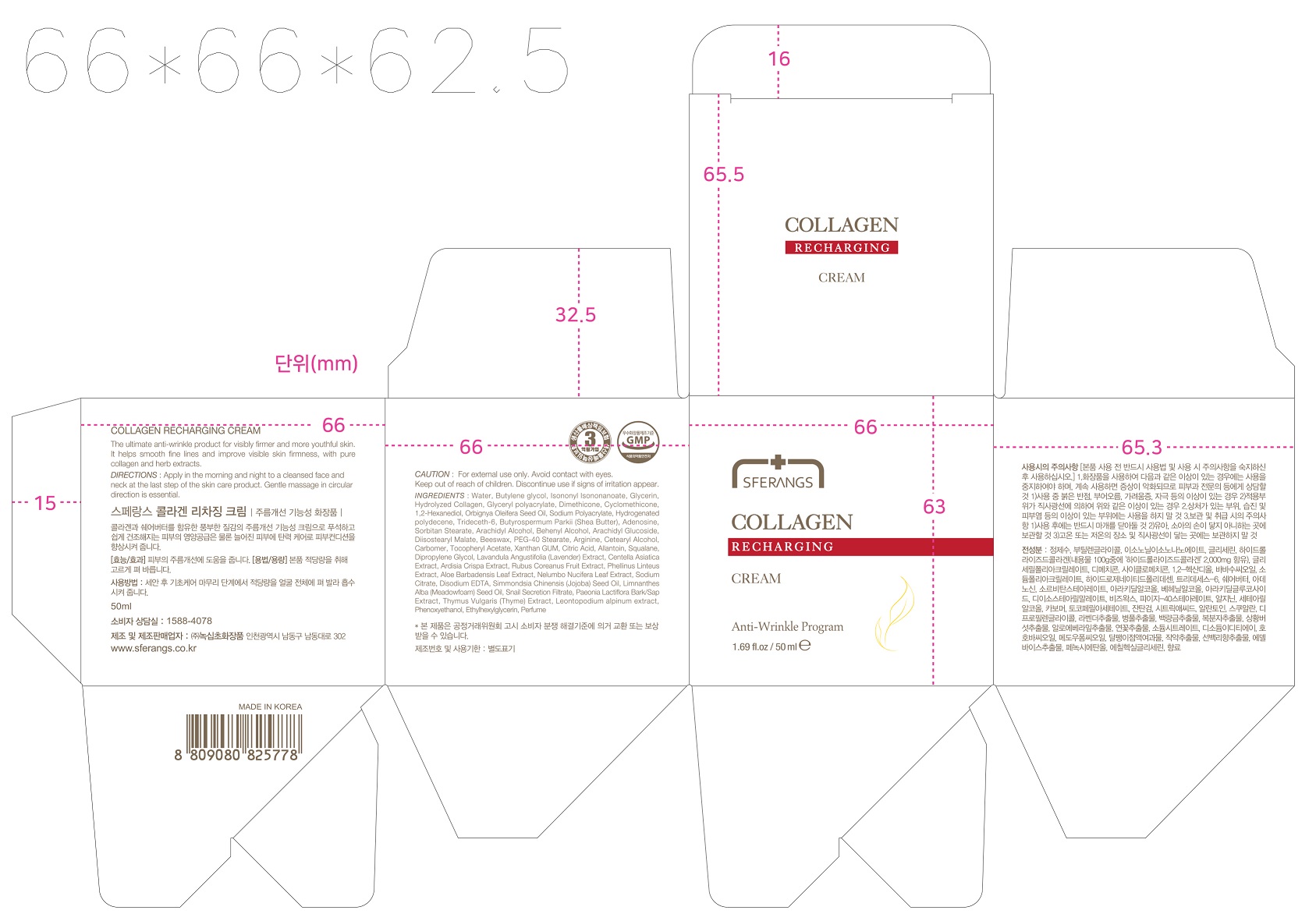
Label: SFERANGS COLLAGEN RECHARGINGCREAM- adenosine cream
- NDC Code(s): 73590-0051-1, 73590-0051-2
- Packager: NOKSIBCHO cosmetic Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated April 30, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Water
Butylene glycol
Isononyl Isononanoate
Glycerin
Hydrolyzed Collagen
Glyceryl polyacrylate
1,2-Hexanediol
Orbignya Oleifera Seed Oil
Sodium Polyacrylate
Hydrogenated polydecene
Trideceth-6
Butyrospermum Parkii (Shea Butter)
Sorbitan Stearate
Arachidyl Alcohol
Behenyl Alcohol
Arachidyl Glucoside
Glucose
Diisostearyl Malate
Beeswax
PEG-40 Stearate
Arginine
Cetearyl Alcohol
Carbomer
Tocopheryl Acetate
Xanthan GUM
Citric Acid
Allantoin
Dimethicone
Squalane
Dipropylene Glycol
Lavandula Angustifolia (Lavender) Extract
Propylene Glycol
Centella Asiatica Extract
Phellinus Linteus Extract
Aloe Barbadensis Leaf Extract
Nelumbo Nucifera Leaf Extract
Sodium Citrate
Disodium EDTA
Simmondsia Chinensis (Jojoba) Seed Oil
Limnanthes Alba (Meadowfoam) Seed Oil
Snail Secretion Filtrate
Paeonia Lactiflora Root Extract
Thymus Vulgaris (Thyme) Extract
Leontopodium alpinum extract
Phenoxyethanol
Ethylhexylglycerin
Parfum - PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
For external use only
When using this product■ if the following symptoms occurs after use, stop use and consult with a skin specialist
red specks, swelling, itching
■ don’t use on the part where there is injury, eczema, or dermatitis
Keep out of reach of children
■ if swallowed, get medical help or contact a person control center immediately
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SFERANGS COLLAGEN RECHARGINGCREAM
adenosine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73590-0051 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ADENOSINE (UNII: K72T3FS567) (ADENOSINE - UNII:K72T3FS567) ADENOSINE 0.04 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73590-0051-1 50 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/31/2020 2 NDC:73590-0051-2 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 03/08/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/08/2020 Labeler - NOKSIBCHO cosmetic Co., Ltd. (690182175) Registrant - NOKSIBCHO cosmetic Co., Ltd. (690182175) Establishment Name Address ID/FEI Business Operations NOKSIBCHO cosmetic Co., Ltd. 690182175 manufacture(73590-0051)