Label: AMPITREXYL- andrographis paniculata whole capsule
- NHRIC Code(s): 58988-1140-1
- Packager: ProMex LLC
- Category: DIETARY SUPPLEMENT
- DEA Schedule: None
- Marketing Status: Dietary Supplement
Drug Label Information
Updated January 24, 2018
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
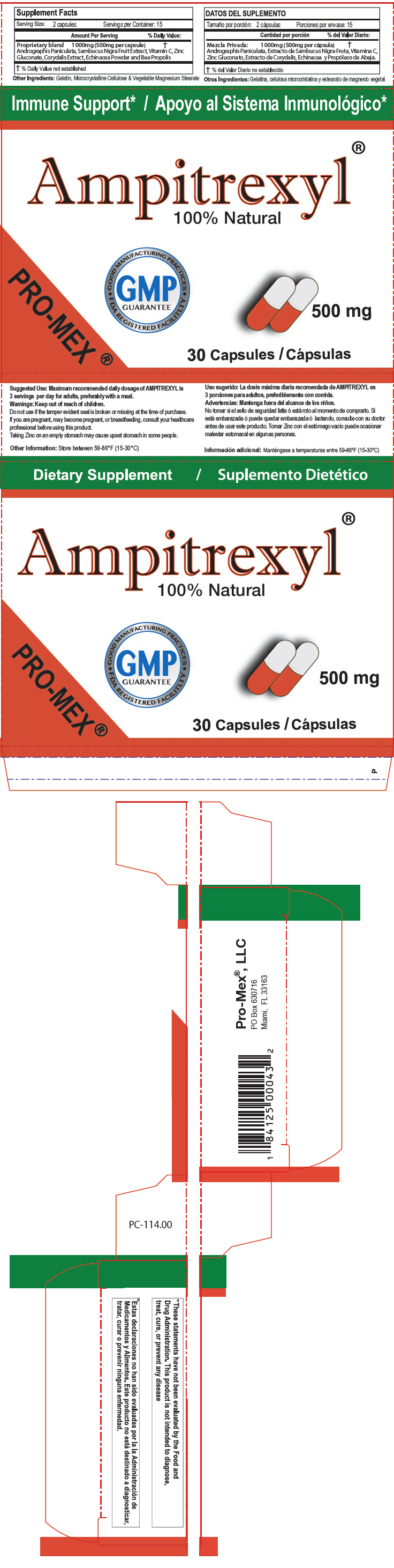
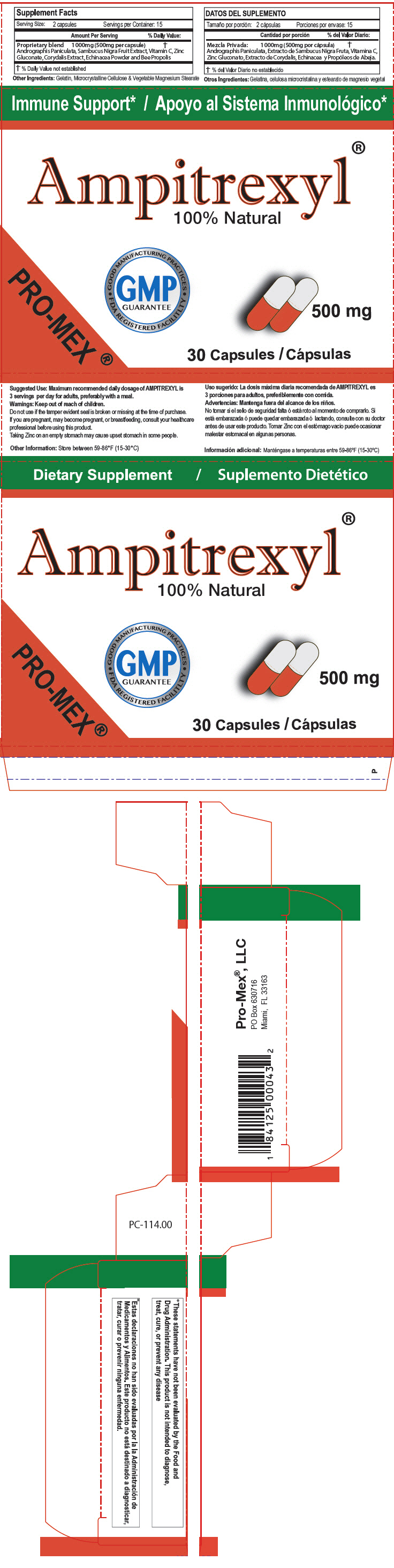
STATEMENT OF IDENTITY
Supplements Facts Serving Size: 2 capsules Servings per Container: 15 Amount Per Serving % Daily Value Proprietary blend 1000mg (500mg per capsule) Andrographis Paniculata, Sambucus Nigra Fruit Extract, Vitamin C, Zinc Gluconate, Corydalis Extract, Echinacea Powder and Bee Propolis % Daily value not established Other ingredients:Gelatin, Microcrystalline Cellulose & Vegetable Magnesium Stearate - Suggested Use
-
Warnings
Keep out of reach of children.
Do not use if the tamper evident seal is broken or missing at the time of purchase.
If you are pregnant, may become pregnant, or breastfeeding, consult your healthcare professional before using this product.
Taking Zinc on an empty stomach may cause upset stomach in some people.
- Other Information
- HEALTH CLAIM
- PRINCIPAL DISPLAY PANEL - 500 mg Capsule Blister Pack Carton
-
INGREDIENTS AND APPEARANCE
AMPITREXYL
andrographis paniculata whole capsuleProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:58988-1140 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ANDROGRAPHIS PANICULATA WHOLE (UNII: 0P49L952WZ) (ANDROGRAPHIS PANICULATA WHOLE - UNII:0P49L952WZ) ANDROGRAPHIS PANICULATA WHOLE 500 mg Inactive Ingredients Ingredient Name Strength EUROPEAN ELDERBERRY (UNII: BQY1UBX046) ASCORBIC ACID (UNII: PQ6CK8PD0R) ZINC GLUCONATE (UNII: U6WSN5SQ1Z) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MAGNESIUM STEARATE (UNII: 70097M6I30) DICENTRA CANADENSIS ROOT (UNII: Y5JS7YM46F) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) PROPOLIS WAX (UNII: 6Y8XYV2NOF) ECHINACEA, UNSPECIFIED (UNII: 4N9P6CC1DX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:58988-1140-1 2 in 1 CARTON 1 15 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 05/31/2006 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color color scoring 1 shape size (solid drugs) 20 mm Labeler - ProMex LLC (789974388) Establishment Name Address ID/FEI Business Operations ProMex LLC 789974388 MANUFACTURE(58988-1140)