Label: FOLKS ALCOHOL FREE HAND SANITIZER FOAM- benzalkonium chloride liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 74538-0008-1 - Packager: WORLDCHEM CO LTD
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 30, 2020
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
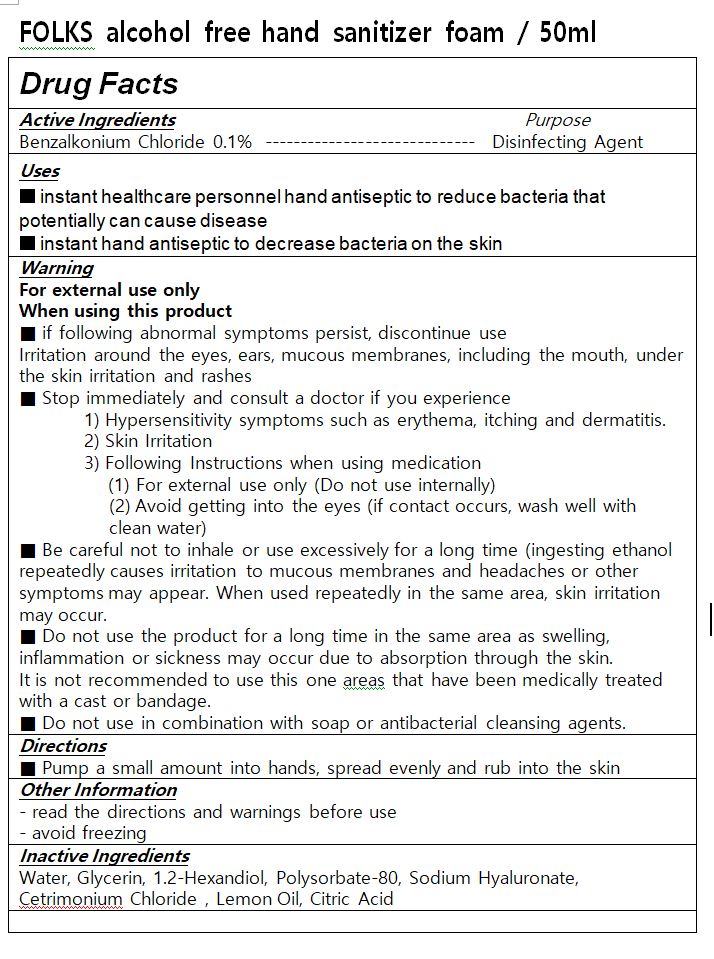
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
-
WARNINGS
■ if following abnormal symptoms persist, discontinue use
Irritation around the eyes, ears, mucous membranes, including the mouth, under the skin irritation and rashes
■ Stop immediately and consult a doctor if you experience
1) Hypersensitivity symptoms such as erythema, itching and dermatitis.
2) Skin Irritation
3) Following Instructions when using medication
(1) For external use only (Do not use internally)
(2) Avoid getting into the eyes (if contact occurs, wash well with clean water)
■ Be careful not to inhale or use excessively for a long time (ingesting ethanol repeatedly causes irritation to mucous membranes and headaches or other symptoms may appear. When used repeatedly in the same area, skin irritation may occur.
■ Do not use the product for a long time in the same area as swelling, inflammation or sickness may occur due to absorption through the skin.
It is not recommended to use this one areas that have been medically treated with a cast or bandage.
■ Do not use in combination with soap or antibacterial cleansing agents.
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
FOLKS ALCOHOL FREE HAND SANITIZER FOAM
benzalkonium chloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:74538-0008 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.1 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) LEVOMENTHOL (UNII: BZ1R15MTK7) GLYCERIN (UNII: PDC6A3C0OX) CETRIMONIUM CHLORIDE (UNII: UC9PE95IBP) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) POLYSORBATE 80 (UNII: 6OZP39ZG8H) HYALURONATE SODIUM (UNII: YSE9PPT4TH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:74538-0008-1 50 mL in 1 BOTTLE; Type 0: Not a Combination Product 04/04/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 04/04/2020 Labeler - WORLDCHEM CO LTD (631058864) Registrant - WORLDCHEM CO LTD (631058864) Establishment Name Address ID/FEI Business Operations WORLDCHEM CO LTD 631058864 manufacture(74538-0008)