Label: SKIN PHARMACY ADVANCED ACNE THERAPY KIT- salicylic acid, benzoyl peroxide kit
- NDC Code(s): 68634-067-01
- Packager: AMCOL Health & Beauty Solutions, Inc. DBA
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 11, 2016
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Use
-
Warnings
For external use only
Ask a doctor or pharmacist before use if you are
Using other topical acne medications at the same time or immediately following use of this product. This may increase dryness or irritation of the skin. If this occurs, only one medication should be used unless directed by a doctor.
When using this product
- Skin irritation and dryness are more likely to occur if you use another topical acne medication at the same time. If irritation occurs, only use one topical acne medication at a time.
- When using this product avoid contact with eyes. If this occurs, immediately flush with water.
- Directions
-
Inactive ingredients
Water, Sodium C14-16 Olefin Sulfonate, Peg-8, Cocamidopropyl Betaine, Xanthan Gum, Lauryl Methacrylate/Glycol Dimethacrylate Crosspolymer, DMDM Hydantoin, Disodium EDTA, Tocopheryl Acetate, Hamamelis Virginiana (Witch Hazel) Water, Butylene Glycol, Sodium Benzotriazolyl Butylphenol Sulfonate, Calendula Officinalis Flower Extract, Glycyrrhiza Glabra (Licorice) Root Extract, Phenoxyethanol, Yellow 5, Red 40, Fragrance.
M050775
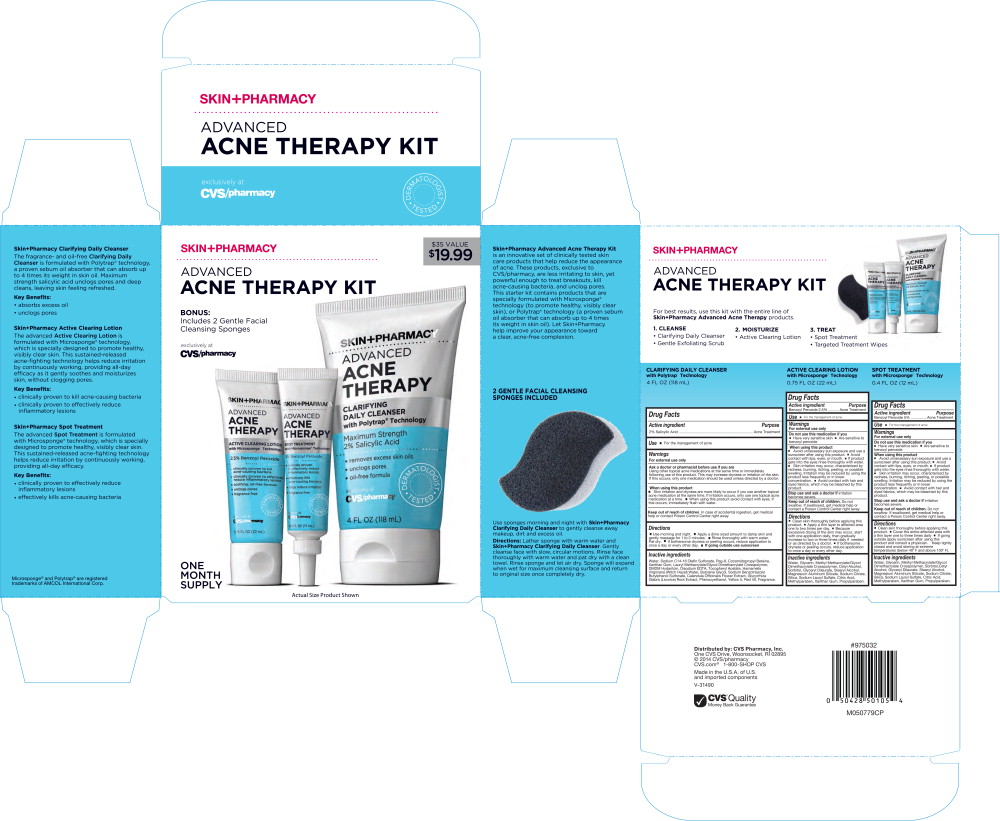
- Principal Display Panel - Skin+Pharmacy Advanced Acne Therapy Kit Carton Label
- Principal Display Panel - Skin+Pharmacy Advanced Acne Therapy Kit Label
-
Principal Display Panel - Skin+Pharmacy Advanced Acne Therapy Kit Label
SKIN+PHARMACY
ADVANCED
ACNE
THERAPYACTIVE CLEARING LOTION
with Microsponge® Technology2.5% Benzoyl Peroxide
- clinically proven to kill acne-causing bacteria
- clinically proven to effectively reduce inflammatory lesions
- soothing, oil-free formula
- unclogs pores
- fragrance free
DERMATOLOGIST TESTED
exclusively at
CVS/pharmacy0.75 FL OZ (22 mL)
-
Principal Display Panel - Skin+Pharmacy Advanced Acne Therapy Kit Label
SKIN+PHARMACY
ADVANCED
ACNE
THERAPYSPOT TREATMENT
with Microsponge® Technology5% Benzoyl Peroxide
- clinically proven to effectively reduce inflammatory lesions
- effectively kills acne-causing bacteria
- helps reduce irritation
- fragrance free
DERMATOLOGIST TESTED
exclusively at
CVS/pharmacy0.4 FL OZ (12 mL)
-
INGREDIENTS AND APPEARANCE
SKIN PHARMACY ADVANCED ACNE THERAPY KIT
salicylic acid, benzoyl peroxide kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68634-067 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68634-067-01 1 in 1 CARTON; Type 0: Not a Combination Product 10/01/2014 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 118 mL Part 2 1 TUBE 22 mL Part 3 1 TUBE 12 mL Part 1 of 3 SKIN PHARMACY ADVANCED ACNE THERAPY CLARIFYING DAILY CLEANSER
salicylic acid liquidProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength salicylic acid (UNII: O414PZ4LPZ) (salicylic acid - UNII:O414PZ4LPZ) salicylic acid 20 g in 2 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) sodium C14-16 olefin sulfonate (UNII: O9W3D3YF5U) polyethylene glycol 4000 (UNII: 4R4HFI6D95) cocamidopropyl betaine (UNII: 5OCF3O11KX) Xanthan gum (UNII: TTV12P4NEE) DMDM Hydantoin (UNII: BYR0546TOW) edetate disodium (UNII: 7FLD91C86K) .alpha.-tocopherol acetate, DL- (UNII: WR1WPI7EW8) Hamamelis virginiana flower water (UNII: 222MYC9QUV) Butylene glycol (UNII: 3XUS85K0RA) Calendula Officinalis flower (UNII: P0M7O4Y7YD) licorice (UNII: 61ZBX54883) Phenoxyethanol (UNII: HIE492ZZ3T) FD&C Yellow No. 5 (UNII: I753WB2F1M) FD&C red no. 40 (UNII: WZB9127XOA) Lauryl Methacrylate/Glycol Dimethacrylate Crosspolymer (UNII: EX0F4CZ66H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 118 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 10/01/2014 Part 2 of 3 SKIN PHARMACY ADVANCED ACNE THERAPY ACTIVE CLEARING
benzoyl peroxide lotionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength benzoyl peroxide (UNII: W9WZN9A0GM) (benzoyl peroxide - UNII:W9WZN9A0GM) benzoyl peroxide 25 g in 2 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) glycerin (UNII: PDC6A3C0OX) methyl methacrylate/glycol dimethacrylate crosspolymer (UNII: EG97988M5Q) cetyl alcohol (UNII: 936JST6JCN) sorbitol (UNII: 506T60A25R) glyceryl dilaurate (UNII: MFL3ZIE8SK) stearyl alcohol (UNII: 2KR89I4H1Y) magnesium aluminum silicate (UNII: 6M3P64V0NC) sodium citrate (UNII: 1Q73Q2JULR) silicon dioxide (UNII: ETJ7Z6XBU4) sodium lauryl sulfate (UNII: 368GB5141J) citric acid monohydrate (UNII: 2968PHW8QP) methylparaben (UNII: A2I8C7HI9T) xanthan gum (UNII: TTV12P4NEE) propylparaben (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 22 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 10/01/2014 Part 3 of 3 SKIN PHARMACY ADVANCED ACNE THERAPY SPOT TREATMENT
benzoyl peroxide lotionProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength benzoyl peroxide (UNII: W9WZN9A0GM) (benzoyl peroxide - UNII:W9WZN9A0GM) benzoyl peroxide 50 g in 2 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) glycerin (UNII: PDC6A3C0OX) methyl methacrylate/glycol dimethacrylate crosspolymer (UNII: EG97988M5Q) cetyl alcohol (UNII: 936JST6JCN) sorbitol (UNII: 506T60A25R) glyceryl dilaurate (UNII: MFL3ZIE8SK) stearyl alcohol (UNII: 2KR89I4H1Y) magnesium aluminum silicate (UNII: 6M3P64V0NC) sodium citrate (UNII: 1Q73Q2JULR) silicon dioxide (UNII: ETJ7Z6XBU4) sodium lauryl sulfate (UNII: 368GB5141J) citric acid monohydrate (UNII: 2968PHW8QP) methylparaben (UNII: A2I8C7HI9T) xanthan gum (UNII: TTV12P4NEE) propylparaben (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 12 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 10/01/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333D 10/01/2014 Labeler - AMCOL Health & Beauty Solutions, Inc. DBA (872684803) Establishment Name Address ID/FEI Business Operations AMCOL Household & Personal Care 872684803 MANUFACTURE(68634-067) , PACK(68634-067) , LABEL(68634-067) , ANALYSIS(68634-067)