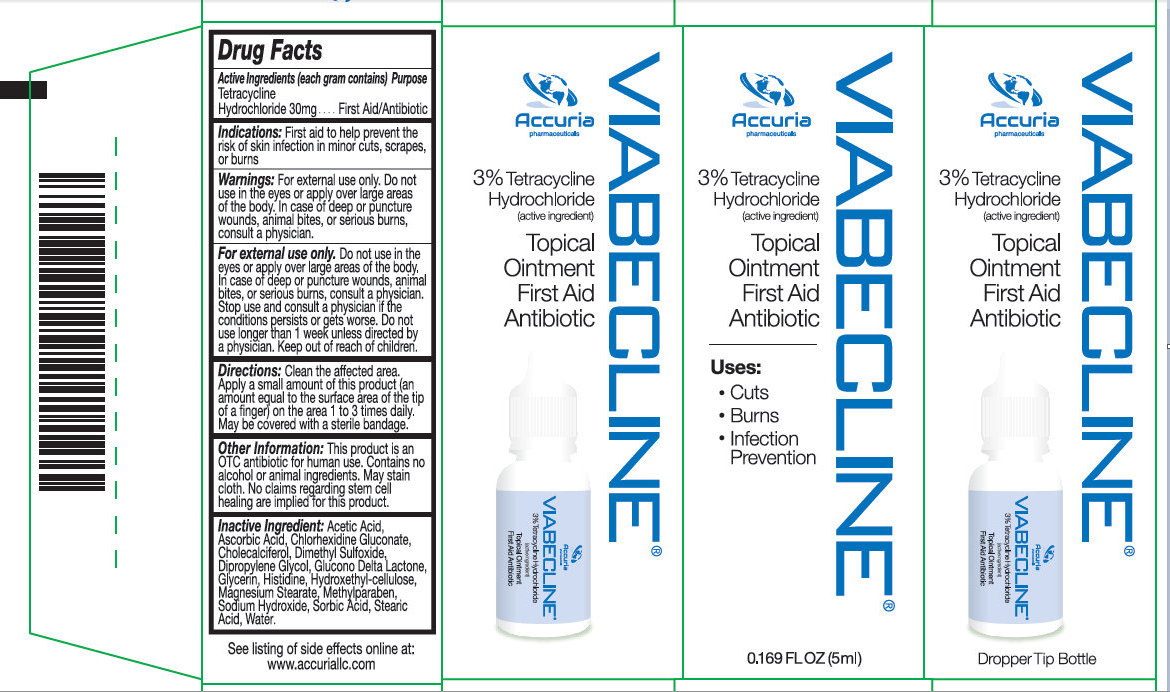
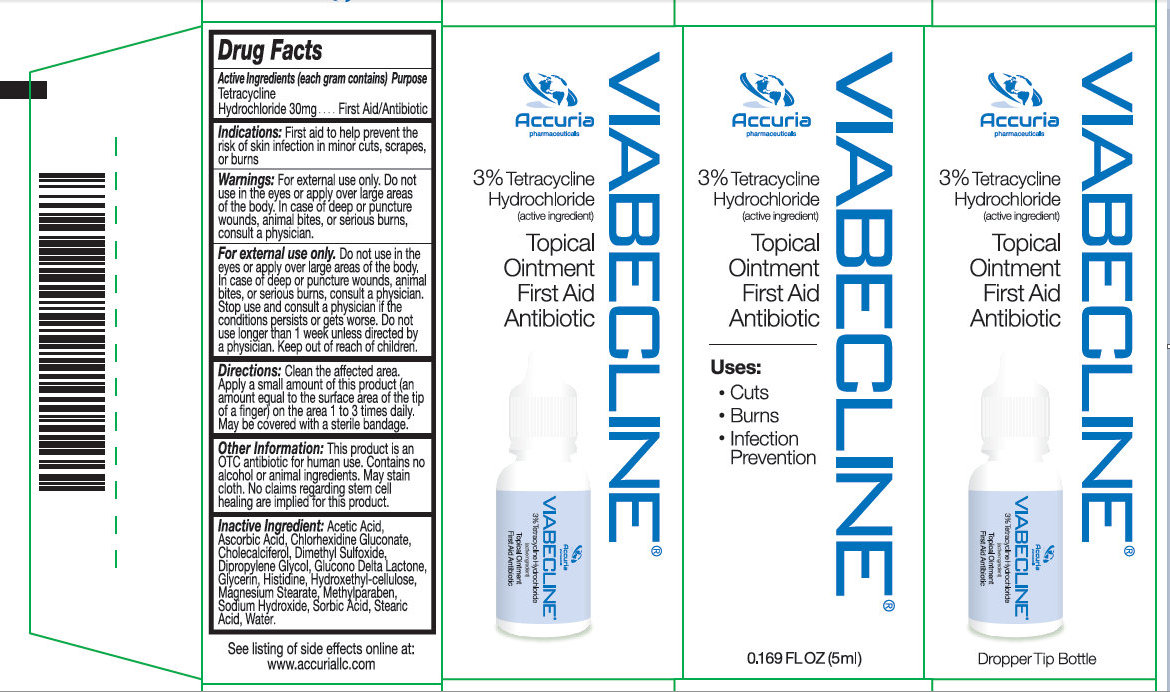
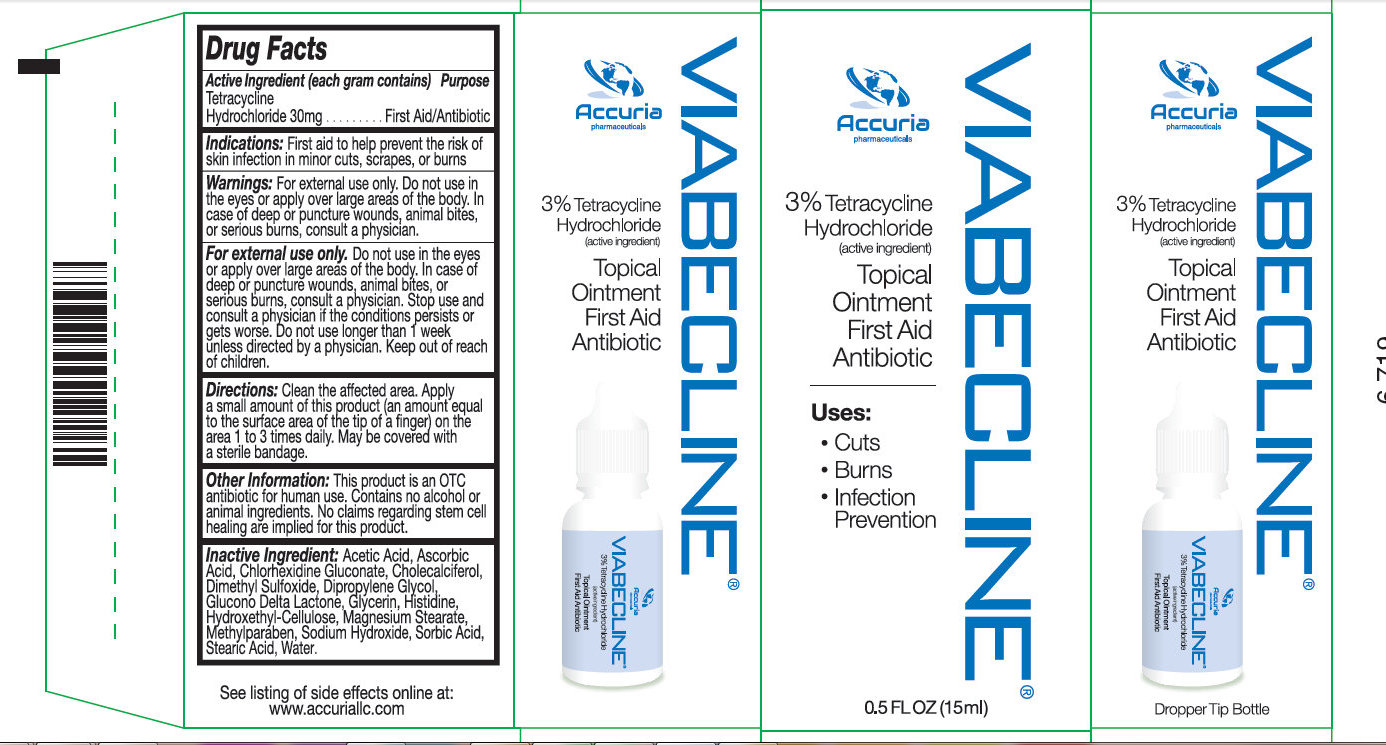
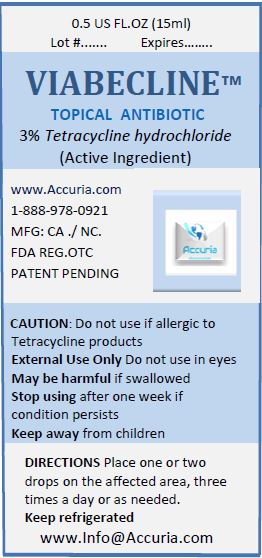
Label: VIABECLINE- tetracycline hydrochloride ointment
- NDC Code(s): 71491-100-00, 71491-100-01
- Packager: Accuria, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 1, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Activer Ingredient (in each gram)
- Purpose
- Use
-
Warnings
May be harmful if swallowed. For external use only.
Do not use if allergic to any ingredient listed on this label. Allergy Alert:
Do not use
Ask doctor before use if you have
Stop use and ask a doctor if the condition persists or gets worse.
- in the eyes
- over large areas of the body
- longer than 1 week unless directed by a doctor
- deep or puncture wounds
- animal bites
- serious burns
- KEEP OUT OF REACH OF CHILDREN
- Directions
-
Other information
- Keep product refrigerated to preserve its effectiveness and color
- Stop use if the product is misused: If the bottle is left open and or not refrigerated, the liquid will tend to turn black over time. Discard the product if the liquid turns black due to misuse.
- This product is an OTC antibiotic for human use.
- Contains no alcohol, no animal ingredients.
- Blended for typical skin color.
- May stain cloth.
- No claims regarding stem cell healing are implied for this product.
- Inactive Ingredients
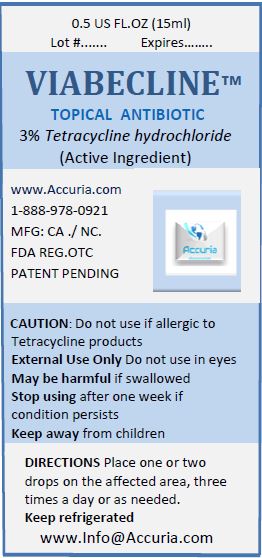
- Package Label: 71491-100-00 VIABECLICNE 5 ML Package Label: 71491-100-01 VIABECLINE 15ML
-
INGREDIENTS AND APPEARANCE
VIABECLINE
tetracycline hydrochloride ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71491-100 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TETRACYCLINE HYDROCHLORIDE (UNII: P6R62377KV) (TETRACYCLINE - UNII:F8VB5M810T) TETRACYCLINE HYDROCHLORIDE 30 mg in 1 mL Inactive Ingredients Ingredient Name Strength ACETIC ACID (UNII: Q40Q9N063P) ASCORBIC ACID (UNII: PQ6CK8PD0R) CHLORHEXIDINE GLUCONATE (UNII: MOR84MUD8E) CHOLECALCIFEROL (UNII: 1C6V77QF41) DIMETHYL SULFOXIDE (UNII: YOW8V9698H) DIPROPYLENE GLYCOL (UNII: E107L85C40) GLUCONOLACTONE (UNII: WQ29KQ9POT) GLYCERIN (UNII: PDC6A3C0OX) HISTIDINE (UNII: 4QD397987E) HYDROXYMETHYL CELLULOSE (UNII: 273FM27VK1) MAGNESIUM STEARATE (UNII: 70097M6I30) METHYLPARABEN (UNII: A2I8C7HI9T) SODIUM HYDROXIDE (UNII: 55X04QC32I) SORBIC ACID (UNII: X045WJ989B) STEARIC ACID (UNII: 4ELV7Z65AP) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71491-100-01 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 06/15/2017 2 NDC:71491-100-00 5 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 06/15/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M004 06/14/2017 Labeler - Accuria, LLC (080520669) Establishment Name Address ID/FEI Business Operations Filltech USA, LLC 926433855 manufacture(71491-100)