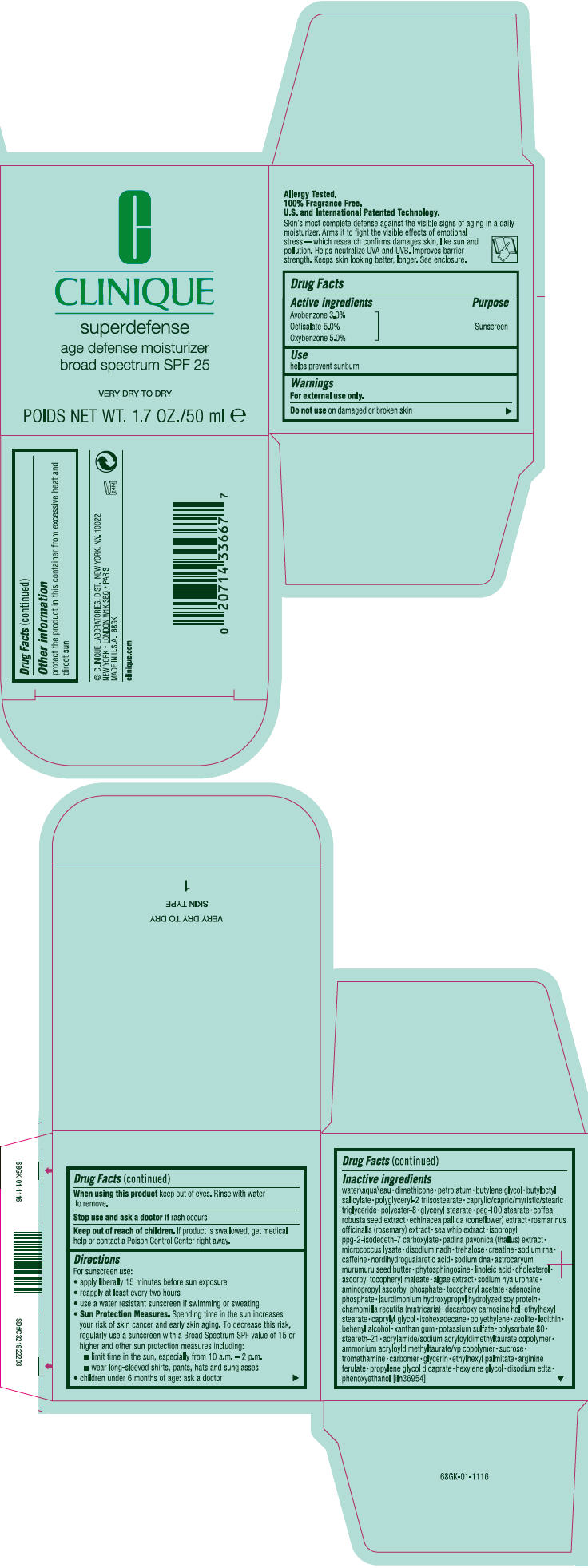
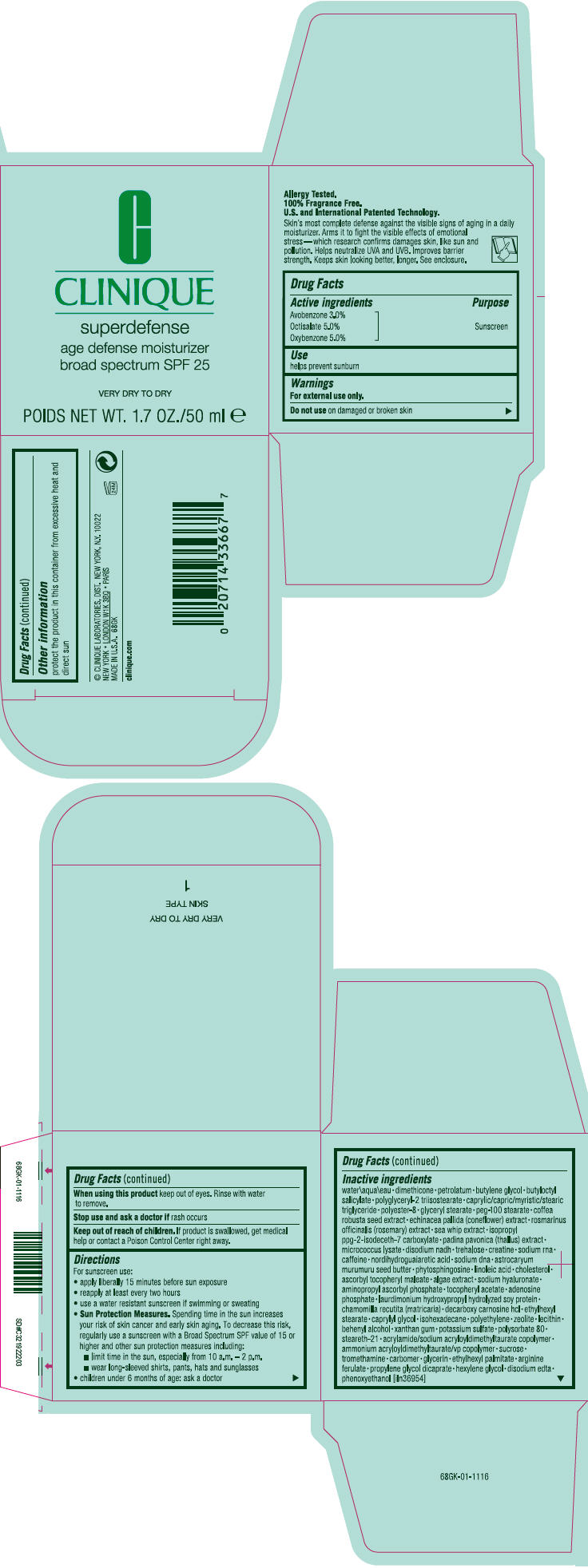
Label: SUPERDEFENSE AGE DEFENSE MOISTURIZER BROAD SPECTRUM SPF 25 VERY DRY TO DRY- octisalate, oxybenzone, and avobenzone cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 49527-034-01 - Packager: CLINIQUE LABORATORIES INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 18, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Use
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
- limit time in the sun, especially from 10 a.m. – 2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
-
Inactive ingredients
water\aqua\eau • dimethicone • petrolatum • butylene glycol • butyloctyl salicylate • polyglyceryl-2 triisostearate • caprylic/capric/myristic/stearic triglyceride • polyester-8 • glyceryl stearate • peg-100 stearate • coffea robusta seed extract • echinacea pallida (coneflower) extract • rosmarinus officinalis (rosemary) extract • sea whip extract • isopropyl ppg-2-isodeceth-7 carboxylate • padina pavonica (thallus) extract • micrococcus lysate • disodium nadh • trehalose • creatine • sodium rna • caffeine • nordihydroguaiaretic acid • sodium dna • astrocaryum murumuru seed butter • phytosphingosine • linoleic acid • cholesterol • ascorbyl tocopheryl maleate • algae extract • sodium hyaluronate • aminopropyl ascorbyl phosphate • tocopheryl acetate • adenosine phosphate • laurdimonium hydroxypropyl hydrolyzed soy protein • chamomilla recutita (matricaria) • decarboxy carnosine hcl • ethylhexyl stearate • caprylyl glycol • isohexadecane • polyethylene • zeolite • lecithin • behenyl alcohol • xanthan gum • potassium sulfate • polysorbate 80 • steareth-21 • acrylamide/sodium acryloyldimethyltaurate copolymer • ammonium acryloyldimethyltaurate/vp copolymer • sucrose • tromethamine • carbomer • glycerin • ethylhexyl palmitate • arginine ferulate • propylene glycol dicaprate • hexylene glycol • disodium edta • phenoxyethanol [iln36954]
- Other information
- PRINCIPAL DISPLAY PANEL - 50 ml Jar Carton
-
INGREDIENTS AND APPEARANCE
SUPERDEFENSE AGE DEFENSE MOISTURIZER BROAD SPECTRUM SPF 25 VERY DRY TO DRY
octisalate, oxybenzone, and avobenzone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49527-034 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 0.0508 g in 1 mL OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 0.0508 g in 1 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 0.0305 g in 1 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) dimethicone (UNII: 92RU3N3Y1O) petrolatum (UNII: 4T6H12BN9U) butylene glycol (UNII: 3XUS85K0RA) butyloctyl salicylate (UNII: 2EH13UN8D3) glyceryl monostearate (UNII: 230OU9XXE4) peg-100 stearate (UNII: YD01N1999R) robusta coffee bean (UNII: V5032728L7) echinacea pallida (UNII: 904CK3270L) rosemary (UNII: IJ67X351P9) pseudopterogorgia elisabethae (UNII: UDY3H1OUX5) padina pavonica (UNII: 177U06NHZI) trehalose (UNII: B8WCK70T7I) creatine (UNII: MU72812GK0) caffeine (UNII: 3G6A5W338E) masoprocol (UNII: 7BO8G1BYQU) astrocaryum murumuru seed butter (UNII: 12V64UPU6R) phytosphingosine (UNII: GIN46U9Q2Q) linoleic acid (UNII: 9KJL21T0QJ) cholesterol (UNII: 97C5T2UQ7J) ascorbyl tocopheryl maleate (UNII: D2G6259XR5) hyaluronate sodium (UNII: YSE9PPT4TH) aminopropyl ascorbyl phosphate (UNII: 290O2PQ83R) .alpha.-tocopherol acetate (UNII: 9E8X80D2L0) adenosine phosphate (UNII: 415SHH325A) decarboxy carnosine Hydrochloride (UNII: 6X7K9I5QR7) ethylhexyl stearate (UNII: EG3PA2K3K5) caprylyl glycol (UNII: 00YIU5438U) isohexadecane (UNII: 918X1OUF1E) high density polyethylene (UNII: UG00KM4WR7) docosanol (UNII: 9G1OE216XY) xanthan gum (UNII: TTV12P4NEE) potassium sulfate (UNII: 1K573LC5TV) polysorbate 80 (UNII: 6OZP39ZG8H) steareth-21 (UNII: 53J3F32P58) sucrose (UNII: C151H8M554) ammonium acryloyldimethyltaurate/vp copolymer (UNII: W59H9296ZG) tromethamine (UNII: 023C2WHX2V) glycerin (UNII: PDC6A3C0OX) ethylhexyl palmitate (UNII: 2865993309) propylene glycol dicaprate (UNII: U783H9JHWY) hexylene glycol (UNII: KEH0A3F75J) edetate disodium (UNII: 7FLD91C86K) phenoxyethanol (UNII: HIE492ZZ3T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49527-034-01 1 in 1 CARTON 1 50 mL in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part352 06/01/2010 Labeler - CLINIQUE LABORATORIES INC. (173047747) Establishment Name Address ID/FEI Business Operations ELGC K.K. 712808195 RELABEL(49527-034) , REPACK(49527-034) Establishment Name Address ID/FEI Business Operations Estee Lauder Pennsylvania Distribution Center 2 (PADC 2) 828534516 MANUFACTURE(49527-034) , RELABEL(49527-034) , REPACK(49527-034) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS DISTRIBUTION CENTER 208579636 REPACK(49527-034) , RELABEL(49527-034) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD 244669714 MANUFACTURE(49527-034) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER COSMETICS, LTD. 205952385 MANUFACTURE(49527-034) Establishment Name Address ID/FEI Business Operations ESTEE LAUDER N.V. 370151326 MANUFACTURE(49527-034) Establishment Name Address ID/FEI Business Operations LEN-RON MANUFACTURING DIVISION OF ARAMIS INC 809771152 MANUFACTURE(49527-034) Establishment Name Address ID/FEI Business Operations NORTEC KEYSTONE 943871157 MANUFACTURE(49527-034) , RELABEL(49527-034) , REPACK(49527-034) Establishment Name Address ID/FEI Business Operations NORTHTEC BRISTOL 959338336 MANUFACTURE(49527-034) , RELABEL(49527-034) , REPACK(49527-034) Establishment Name Address ID/FEI Business Operations NORTHTEC KEYSTONE 949264774 MANUFACTURE(49527-034) , RELABEL(49527-034) , REPACK(49527-034) Establishment Name Address ID/FEI Business Operations WHITMAN LABORATORIES, LTD. 216866277 MANUFACTURE(49527-034) Establishment Name Address ID/FEI Business Operations Aveda Corporation 071352058 MANUFACTURE(49527-034) Establishment Name Address ID/FEI Business Operations Pennsylvania Logistics Center 078364654 REPACK(49527-034) , RELABEL(49527-034)