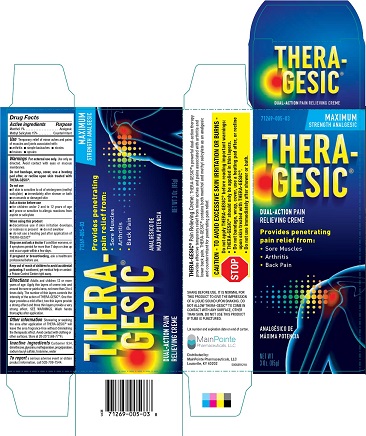
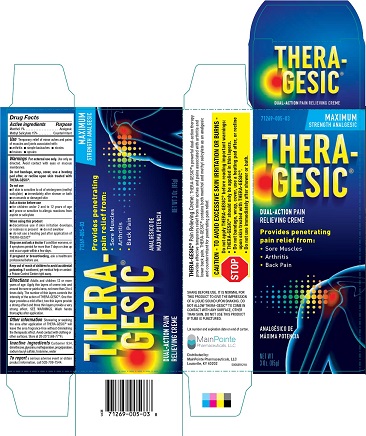
Label: THERAGESIC CREME- methyl salicylate cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 71269-005-03 - Packager: Mainpointe Pharmaceuticals, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 20, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Drug Facts
Active Ingredients Purpose
Menthol 1%...................................................................................................................... Analgesic
Methyl Salicylate 15%............................................................................................... Counterirritant - PURPOSE
- WARNINGS
- DO NOT USE
- ASK DOCTOR
- WHEN USING
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
-
PRECAUTIONS
SHARE BEFORE USE: IT IS NORMAL FOR THIS PRODUCT TO GIVE THE IMPRESSION OF A LIQUID SOUND UON SHAKING. DO NOT ALLOW THERA-GESIC ® TO COMEIN CONTACT WITH ANY SURFACE, OTHER THAN SKIN. Do not use this product if tube is punctured. Lot number and expiration date on end of carton. MainPointe Pharmaceuticals, LLC Louisville, KY 40202 Rev 50563R1218
- DOSAGE & ADMINISTRATION
- WARNINGS AND PRECAUTIONS
-
INDICATIONS & USAGE
Directions Adults and children 12 or more years of age. Apply thin layers of crème into and around the sore or painful area, not more than 3 to 4 times daily. The number of thin layers controls the intensity of the action of THERA-GESIC ®. One this layer provides a mild effect, two thin layers provide a strong effect and three thin layers provide a very strong effect. SEE WARNINGS. Wash hands thoroughly after application.
- THERA-GESIC 3-oz
-
INGREDIENTS AND APPEARANCE
THERAGESIC CREME
methyl salicylate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71269-005 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYL SALICYLATE (UNII: LAV5U5022Y) (SALICYLIC ACID - UNII:O414PZ4LPZ) METHYL SALICYLATE 150 mg in 1 g MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 10 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCERIN (UNII: PDC6A3C0OX) CARBOMER 934 (UNII: Z135WT9208) TROLAMINE (UNII: 9O3K93S3TK) METHYLPARABEN (UNII: A2I8C7HI9T) SODIUM LAURYL SULFATE (UNII: 368GB5141J) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71269-005-03 1 in 1 CARTON 12/01/2018 1 85 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 01/01/1981 Labeler - Mainpointe Pharmaceuticals, LLC (080544378) Registrant - Mission Pharmacal Company (927726893)