Label: PAIN RELIEF BALM- menthol 4% cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 73672-988-40 - Packager: HEMPFUSION, INC.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 17, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
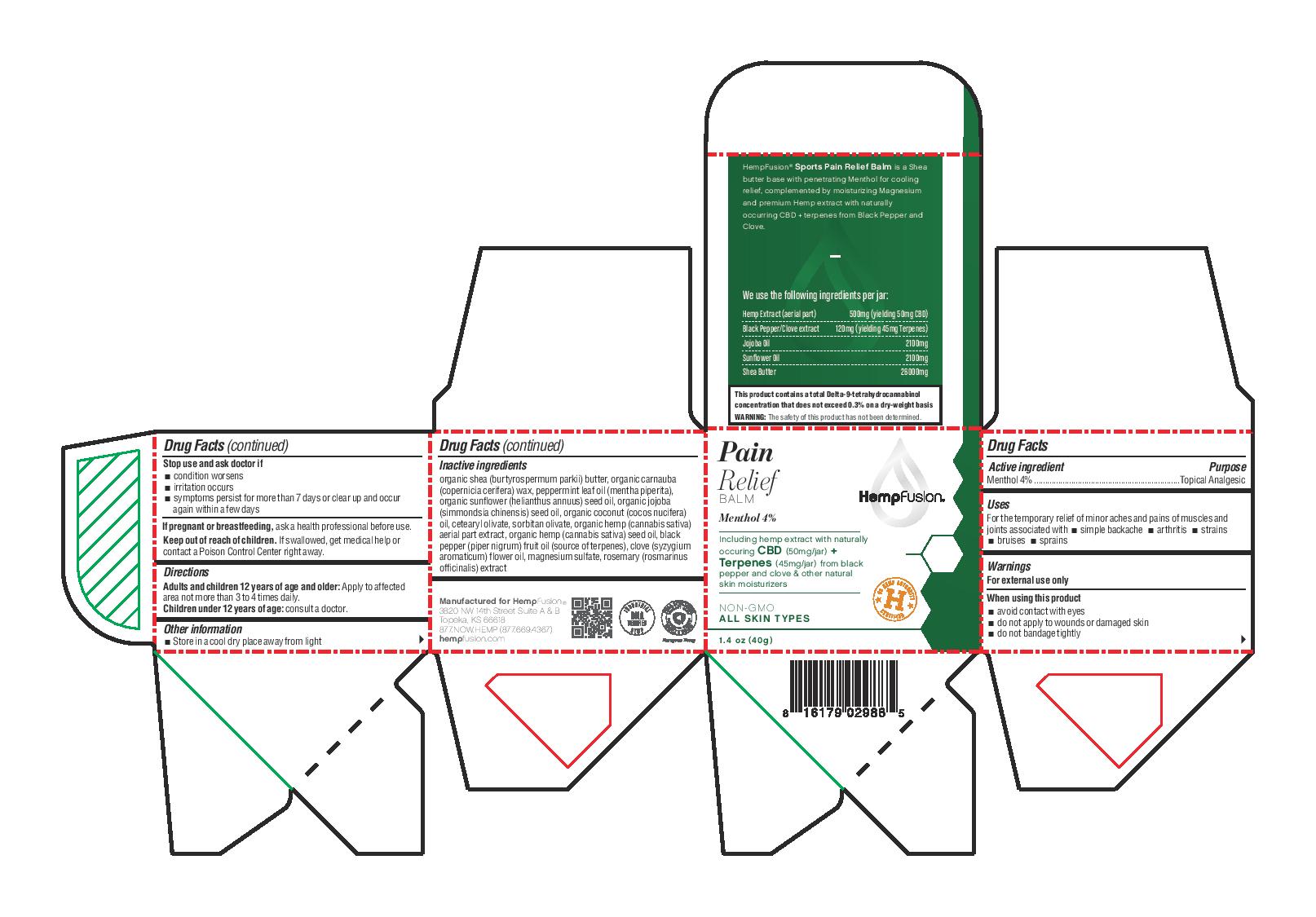
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other information
-
Inactive ingredients
organic shea (burtyrospermum parkii) butter, organic carnauba (copernicia cerifera) wax, peppermint leaf oil (mentha piperita), organic sunflower (helianthus annuus) seed oil, organic jojoba (simmondsia chinensis) seed oil, organic coconut (cocos nucifera) oil, cetearyl olivate, sorbitan olivate, organic hemp (cannabis sativa) aerial part extract, organic hemp (cannabis sativa) seed oil, black pepper (piper nigrum) fruit oil (source of terpenes), clove (syzygium aromaticum) flower oil, magnesium sulfate, rosemary (rosmarinus officinalis) extract
- QUESTIONS
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
PAIN RELIEF BALM
menthol 4% creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73672-988 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 40 mg in 1 g Inactive Ingredients Ingredient Name Strength SUNFLOWER OIL (UNII: 3W1JG795YI) PEPPERMINT OIL (UNII: AV092KU4JH) MAGNESIUM SULFATE, UNSPECIFIED FORM (UNII: DE08037SAB) ROSEMARY (UNII: IJ67X351P9) CARNAUBA WAX (UNII: R12CBM0EIZ) HEMP (UNII: TD1MUT01Q7) BLACK PEPPER OIL (UNII: U17J84S19Z) SHEA BUTTER (UNII: K49155WL9Y) JOJOBA OIL (UNII: 724GKU717M) COCONUT OIL (UNII: Q9L0O73W7L) CETEARYL OLIVATE (UNII: 58B69Q84JO) SORBITAN OLIVATE (UNII: MDL271E3GR) CLOVE OIL (UNII: 578389D6D0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73672-988-40 1 in 1 CARTON 02/10/2020 1 40 g in 1 CONTAINER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 02/10/2020 Labeler - HEMPFUSION, INC. (117152485) Establishment Name Address ID/FEI Business Operations Universal Packaging Systems, Inc. (DBA Paklab) 177711082 manufacture(73672-988)