Label: APO-VARENICLINE- varenicline kit
APO-VARENICLINE- varenicline tablet, film coated
-
NDC Code(s):
60505-3283-6,
60505-4765-5,
60505-4766-5,
60505-4766-6, view more60505-4767-0, 60505-8282-3
- Packager: Apotex Corp
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Unapproved drug for use in drug shortage
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated January 26, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HEALTH CARE PROVIDER LETTER
IMPORTANT PRESCRIBING INFORMATION
July 2, 2021
Apotex Corp.
2400 N Commerce Parkway
Suite 400
Weston, FL 33326
Phone: 1-800-706-5575
Subject: Importation of Apotex Inc.’s Canadian approved Apo-Varenicline (varenicline tartrate) 0.5 mg and 1 mg (based on free base equivalents) Tablets to Address Drug Shortage Dear Healthcare Professional:
In order to alleviate a critical shortage of CHANTIX® (varenicline tartrate) tablets 0.5 mg and 1 mg in the United States (U.S.) market for the indicated use as an aid to smoking cessation, Apotex Corp. is coordinating with the U.S. Food and Drug Administration (FDA) to temporarily import into the U.S. the non-FDA approved drug, Apo-Varenicline (varenicline tartrate) 0.5 mg and 1 mg (free base equivalent) tablets to increase the availability of the drug. The varenicline tartrate tablets marketed in Canada under the name Apo-Varenicline by Apotex Inc. are approved by Health Canada as a generic equivalent to CHAMPIX® tablets manufactured by Pfizer Canada, Inc., and are manufactured in an FDA-inspected facility that complies with current Good Manufacturing Practice (cGMP) requirements.
At this time, no other entity except Apotex Corp. is authorized by the FDA to import or distribute Apotex Inc.’s Apo-Varenicline (varenicline tartrate) 0.5 mg and 1 mg (free base equivalent) tablets in the U.S. However, this does not represent a formal FDA approval of Apotex Inc.’s Apo-Varenicline (varenicline tartrate) tablets in the United States.
Effective immediately, Apotex Corp. will distribute the following presentations of Apotex Inc.’s Apo-Varenicline (varenicline tartrate) tablets to address the critical shortage:
Table 1
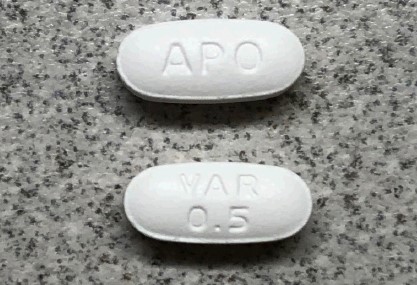
Product Name Quantity Tablet Description U.S. NDC Apo-Varenicline (varenicline tartrate) tablet, 0.5 mg (free base equivalent) Bottle of 56 tablets White colored, modified capsule-shaped, biconvex, film coated tablets, engraved with “APO” on one side and “VAR” over “0.5” on the other side. 60505-4765-5

Apo-Varenicline (varenicline tartrate) tablet, 1 mg (free base equivalent) Bottle of 56 tablets Blue colored, modified capsule-shaped, biconvex, film coated tablets, engraved with “APO” on one side and “VAR” over “1” on the other side. 60505-4766-6

Apo-Varenicline (varenicline tartrate) tablet, 0.5 mg and 1 mg (free base equivalent) Starter Blister Pack of 53 tablets 0.5 mg: White colored, modified capsule-shaped, biconvex, film coated tablets, engraved with “APO” on one side and “VAR” over “0.5” on the other side. 1 mg: Blue colored, modified capsule-shaped, biconvex, film coated tablets, engraved with “APO” on one side and “VAR” over “1” on the other side. 60505-4767-0

Apo-Varenicline (varenicline tartrate) tablet, 1 mg (free base equivalent) Continuation Blister Pack of 56 tablets Blue colored, modified capsule-shaped, biconvex, film coated tablets, engraved with “APO” on one side and “VAR” over “1” on the other side. 60505-4766-5

1 CHAMPIX® is the brand name of CHANTIX® sold in Canada
The product container label will display the text that is approved for marketing the products in Canada with both official languages, English and French. It is important to note that there are differences in the format and content of the labeling between Apo-Varenicline (varenicline tartrate), whether 0.5 mg or 1 mg varenicline free base equivalent, and CHANTIX® (varenicline tartrate) tablets. Please see the product comparison tables at the end of this letter.
CHANTIX® (varenicline tartrate) tablets are available only by prescription in the U.S. Please refer to the package insert for CHANTIX® (varenicline tartrate) tablets for full prescribing information. Ensure that your staff and others in your office and/or pharmacy who may be involved in the prescribing and/or dispensing of Apo-Varenicline (varenicline tartrate) receive a copy of this letter, review the information and instruct patients on the differences between CHANTIX® (varenicline tartrate) and Apo-Varenicline (varenicline tartrate).
The barcode on the imported product label may not register accurately on the U.S. scanning systems. Institutions should manually input the imported product information into their systems and confirm that the barcode, if scanned, provides correct information. Alternative procedures should be followed to assure that the correct drug product is being used and administered to individual patients. Barcodes for the U.S. NDCs for product identification are provided in Table 1 and Appendix 1 to assist with input into institutional systems.
In addition, Apotex Inc.’s Apo-Varenicline (varenicline tartrate) does not meet the product identifier requirements of the Drug Supply Chain Security Act (DSCSA) for the Interoperable Exchange of Information for Tracing of Human, Finished Prescription Drugs. Apo-Varenicline (varenicline tartrate) blister packs are not child resistant.
If you have any questions about the information contained in this letter, any quality related problems, or questions on the use of Apotex Inc.’s Apo-Varenicline (varenicline tartrate), please contact Apotex Corp. Customer Service at 1-800-706-5575.
For ordering information, please contact your primary wholesaler or distributor to place an order with Apotex Corp. at 1-800-706-5575.
Healthcare providers should report adverse events associated with the use of Apotex Inc.’s Apo-Varenicline (varenicline tartrate) to Apotex Corp. at 1-800-706-5575.
Adverse events or quality problems experienced with the use of this product may also be reported to the FDA’s MedWatch Adverse Event Reporting Program either online, by regular mail, or by fax:
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the preaddressed form or submit by fax to 1-800-FDA-0178.
We remain at your disposal to answer any questions you may have about our product and to provide more information if needed.
Sincerely,
Kiran Krishnan, PhD
Senior Vice President, Global Regulatory and Medical Affairs
Apotex Corp.
Enclosures:
Appendix 1 – Barcodes for Pharmacy Dispensing
Appendix 2 – Product Label and Product Characteristics Side-by-Side Comparison Table
Appendix 3 – Prescribing Information Side-by-Side Comparison Table: available at www1.apotex.com/us/apo-varenicline
Apo-Varenicline (varenicline tartrate),0.5 mg and 1 mg varenicline free base equivalent tablets product monograph: available at www1.apotex.com/us/apo-varenicline
Appendix 1: Barcodes for Pharmacy Dispensing
Product Name Quantity NDC Barcode Apo-Varenicline (varenicline tartrate) tablet, 0.5 mg (free base equivalent) Bottle of 56 tablets 60505-4765-5

Apo-Varenicline (varenicline tartrate) tablet, 1 mg (free base equivalent) Bottle of 56 tablets 60505-4766-6

Apo-Varenicline (varenicline tartrate) tablet, 0.5 mg and 1 mg (free base equivalent) Starter Blister Pack of 53 tablets 60505-4767-0

Apo-Varenicline (varenicline tartrate) tablet, 1 mg (free base equivalent) Continuation Blister Pack of 56 tablets 60505-4766-5

Appendix 2: Product Label and Product Characteristics Side-by-Side Comparison Table
U.S. FDA Approved Product Import Product Bottle Container Label (main panel) – 0.5 mg - 56 count 

Bottle Container Label (main panel) – 1 mg - 56 count 

Starter pack Label – 0.5mg & 1 mg – 53 count 

Continuation Pack – 1 mg - 56 count 

0.5 mg Tablet Images 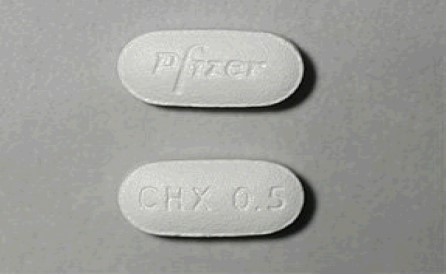
1 mg Tablet Images 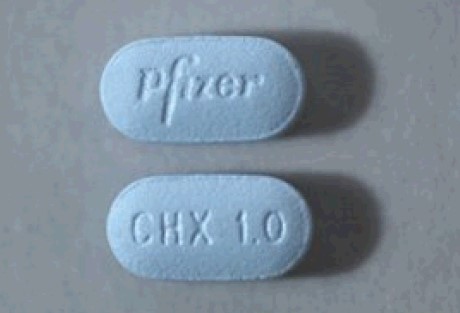

Product Name CHANTIX® 0.5 mg and 1 mg (varenicline) tablets Apo-Varenicline 0.5 mg and 1 mg (varenicline tartrate) tablets Route of Administration Oral Oral Ingredients Each 0.5 mg CHANTIX tablet contains 0.85 mg of varenicline tartrate equivalent to 0.5 mg of varenicline free base; each 1 mg CHANTIX tablet contains 1.71 mg of varenicline tartrate equivalent to 1 mg of varenicline free base.
Active ingredient: varenicline tartrate
Inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, Opadry® White (for 0.5 mg), Opadry® Blue (for 1 mg), and Opadry® Clear.Each 0.5 mg Apo-Varenicline tablet contains 0.5 mg of varenicline (as free base equivalent). Each 1 mg Apo-Varenicline tablet contains 1 mg of varenicline (as free base equivalent).
Active ingredient: Varenicline tartrate.
Inactive ingredients: anhydrous dibasic calcium phosphate, magnesium stearate and microcrystalline cellulose. The film-coating contains hydroxypropyl cellulose, hydroxypropyl methylcellulose, titanium dioxide and triacetin. The 1 mg tablet also contains indigotine aluminum lake 12% to 14% as a colouring agent.Storage Conditions Store at 25ºC (77ºF); excursions permitted to 15–30ºC (59–86ºF) (see USP Controlled Room Temperature). Store at room temperature (15ºC to 30ºC). Appendix 3: Prescribing Information Side-by-Side Comparison Table
U.S. FDA Approved Product Import Product Product Name CHANTIX® (varenicline) tablets APO-VARENICLINE (Varenicline Tablets) 1 INDICATIONS AND USAGE
CHANTIX is indicated for use as an aid to smoking cessation treatment.
INDICATIONS AND CLINICAL USE
Adults
APO-VARENICLINE (varenicline tartrate) is indicated for smoking-cessation treatment in adults, in conjunction with smoking-cessation counselling. Geriatrics (> 65 years of age): No dosage adjustment is necessary for healthy elderly patients. However, varenicline is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see WARNINGS AND PRECAUTIONS, Special Populations: Geriatrics).Pediatrics (< 18 years of age): Based on the data submitted and reviewed by Health Canada, the safety and efficacy of varenicline in pediatric patients has not been established; therefore, Health Canada has not authorized an indication for pediatric use (see WARNINGS AND PRECAUTIONS, Special Populations: Pediatrics).Dosage and Administration 2 DOSAGE AND ADMINISTRATION
2.1 Usual Dosage for Adults
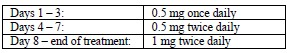
Smoking cessation therapies are more likely to succeed for patients who are motivated to stop smoking and who are provided additional advice and support. Provide patients with appropriate educational materials and counseling to support the quit attempt. The patient should set a date to stop smoking. Begin CHANTIX dosing one week before this date. Alternatively, the patient can begin CHANTIX dosing and then quit smoking between days 8 and 35 of treatment. CHANTIX should be taken orally after eating and with a full glass of water. The recommended dose of CHANTIX is 1 mg twice daily following a 1-week titration as follows:
Patients should be treated with CHANTIX for 12 weeks. For patients who have successfully stopped smoking at the end of 12 weeks, an additional course of 12 weeks treatment with CHANTIX is recommended to further increase the likelihood of long-term abstinence.
For patients who are sure that they are not able or willing to quit abruptly, consider a gradual approach to quitting smoking with CHANTIX. Patients should beginCHANTIX dosing and reduce smoking by 50% from baseline within the first four weeks, by an additional 50% in the next four weeks, and continue reducing with the goal of reaching complete abstinence by 12 weeks. Continue CHANTIX treatment for an additional 12 weeks, for a total of 24 weeks of treatment. Encourage patients to attempt quitting sooner if they feel ready [see Clinical Studies (14.5)].
Patients who are motivated to quit, and who did not succeed in stopping smoking during prior CHANTIX therapy for reasons other than intolerability due to adverse events or who relapsed after treatment, should be encouraged to make another attempt with CHANTIX once factors contributing to the failed attempt have been identified and addressed.
Consider a temporary or permanent dose reduction in patients who cannot tolerate the adverse effects of CHANTIX.DOSAGE AND ADMINISTRATION
Dosing Considerations
Smoking-cessation therapies are more likely to succeed for patients who are motivated to stop smoking and who are provided additional counselling and /or support services. In the clinical trials on which approval was based, varenicline was used with supportive counselling. Physicians should review the patient’s overall smoking-cessation plan that includes treatment with APO-VARENICLINE. The majority of clinical evidence in efficacy and safety was based on a 1 mg BID dose (see CLINICAL TRIALS). There is little clinical experience with doses above the maximum recommended dose of 1 mg BID.There is limited data available for dose comparison. In the one randomized clinical trial that included both 1 mg BID and 0.5 mg BID arms and that was designed to compare each of the two doses to placebo, and not to each other, the quit rates for 1 mg BID (n=253), 0.5 mg BID (n=253) and placebo (n=121) were:- for Weeks 9 to 12: 51%, 45%, and 12% respectively, and
- for Weeks 9 to 52: 23%, 19% and 4% respectively.
For further information on this study, see CLINICAL TRIAL, study 1.Based on the limited data available, it cannot be concluded that there is a difference between the two doses in the rate of serious neuropsychiatric events (see ADVERSE REACTIONS, Neuropsychiatric Adverse Events in Randomized Double Blind, Placebo Controlled Clinical Studies of Varenicline).
APO-VARENICLINE should be taken after eating and with a full glass of water.
Patients with Severe Renal Impairment
The maximum recommended dose for this population is 0.5 mg twice daily (see below: Special Populations, Patients with Impaired Renal Function).
Recommended Dose and Dosage Adjustment
Adults
Setting a quit date:
There are three ways to set a quit date with APO-VARENICLINE:-
Fixed quit approach: The patient sets a date to stop smoking. APO-VARENICLINE dosing should start 1 to 2 weeks before this date (see CLINICAL TRIALS).
OR
-
Flexible quit approach: The patient begins APO-VARENICLINE and then quits smoking between days 8 and 35 of treatment (i.e. between Weeks 2 and 5) (see ACTION AND CLINICAL PHARMACOLOGY, Special Populations, Flexibility in Setting a Quit Date).
OR
-
Gradual quit approach: The patient starts taking APO-VARENICLINE with a goal to quit smoking by end of 12 weeks of treatment. The patient should gradually reduce smoking during the first 12 weeks of treatment such as 50% reduction or more by 4 weeks of treatment, 75% or more by 8 weeks to reach 100% by 12 weeks (see ACTION AND CLINICAL PHARMACOLOGY, Special Populations).
Dosing Options
Following one week of titration, there is a choice of two doses for APO-VARENICLINE: 0.5 mg BID or 1 mg BID.As shown in the table below, the two titration schedules are identical from Day 1 to Day 7, separating at Day 8 when the patient either remains on 0.5 mg BID or moves up to 1 mg BID.

The choice of dosing regimen should be based on physician judgment and patient preference, following discussion with the patient (see also Dosing Considerations).Once APO-VARENICLINE treatment is initiated, the dose may be changed, temporarily or permanently, according to patient and physician judgments on tolerability and efficacy.
Patients who follow one of the first 2 approaches to setting a quit date (1 to 2 weeks after starting the treatment or between days 8 and 35 of treatment) should be treated with APO-VARENICLINE for 12 weeks. For patients who have successfully stopped smoking at the end of 12 weeks, an additional course of 12 weeks treatment with APO-VARENICLINE may be considered. No data are available on the efficacy of an additional 12 week course of treatment with varenicline for patients who have not successfully stopped smoking at the end of 12 weeks.Patients who follow the gradual quit approach (Week 12) should be treated with APO-VARENICLINE for 24 weeks.
Dose tapering may be considered. Regardless of whether the treatment course is 12 or 24 weeks, risk of smoking-cessation relapse is elevated in the period immediately following the end of drug treatment (see CLINICAL TRIALS). In addition, dose tapering may help minimize discontinuation symptoms (e.g., increase in irritability, urge to smoke, depression, and/or insomnia), observed in up to 3% of patients at the end of treatment. -
2.2 Dosage in Special Populations
Patients with Impaired Renal FunctionNo dosage adjustment is necessary for patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance less than 30 mL per min), the recommended starting dose of CHANTIX is 0.5 mg once daily. The dose may then be titrated as needed to a maximum dose of 0.5 mg twice daily. For patients with end-stage renal disease undergoing hemodialysis, a maximum dose of 0.5 mg once daily may be administered if tolerated [see Use in Specific Populations (8.6), Clinical Pharmacology (12.3)]. Elderly and Patients with Impaired Hepatic FunctionNo dosage adjustment is necessary for patients with hepatic impairment. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Use in Specific Populations (8.5)].
Special Populations
Psychiatric Patients
Patients with a history of psychiatric symptoms who are attempting to quit smoking should be monitored by their healthcare professional for new or worsened psychiatric events. Those with a current condition should be clinically stable. Patients should be instructed that if they develop worsened or new symptoms, to report these to their healthcare provider, so that dose adjustments of psychiatric medications and/or APO-VARENICLINE may be considered (see also WARNINGS AND PRECAUTIONS, Special Populations, Psychiatric Patients).
Patients with Impaired Renal Function:
No dosage adjustment is necessary for patients with mild (estimated creatinine clearance > 50 mL/min and ≤ 80 mL/min) to moderate (estimated creatinine clearance ≥ 30 mL/min and ≤ 50 mL/min) renal impairment. For patients who experience intolerable adverse events, dosing may be reduced.For patients with severe renal impairment, the recommended dose of APO-VARENICLINE is 0.5 mg twice daily. Dosing should begin at 0.5 mg once daily for the first 3 days then increased to 0.5 mg twice daily. Based on insufficient clinical experience with varenicline in patients with end-stage renal disease, treatment is not recommended in this patient population (see also WARNINGS AND PRECAUTIONS, Special Populations: Renal Impairment).
Patients with Hepatic Impairment:
No dosage adjustment is necessary for patients with hepatic impairment.Patients with Epilepsy, Patients undergoing Chemotherapy, and Patients with GI disturbances such as irritable bowel: The use of vareniclinehas not been studied inthese patient populations (see WARNINGS AND PRECAUTIONS, Special Populations). Dosing in Elderly Patients:No dosage adjustment is necessary for elderly patients with normal renal function. However, varenicline is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see WARNINGS AND PRECAUTIONS, Special Populations: Geriatrics).Dosage Form and Strengths 3 DOSAGE FORMS AND STRENGTHS
Capsular, biconvex tablets: 0.5 mg (white to off-white, debossed with "Pfizer" on one side and "CHX 0.5" on the other side) and 1 mg (light blue, debossed with"Pfizer" on one side and "CHX 1.0" on the other side).
DOSAGE FORMS, COMPOSITION AND PACKAGING
APO-VARENICLINE is supplied for oral administration in two strengths: 0.5 mg: White colored, modified capsule-shaped, biconvex, film coated tablets, with engraved “APO” on one side and “VAR” over “0.5” on the other side. Each tablet contains 0.5 mg of varenicline (as tartrate). Supplied in high-density polyethylene (HDPE) bottles of 56, 60 &100 tablets and in blisters of 28 & 30 tablets.1 mg: Blue colored, modified capsule-shaped, biconvex, film coated tablets, engraved with “APO” on one side and “VAR” over “1” on the other side. Each tablet contains 1 mg of varenicline (as tartrate). Supplied in high-density polyethylene (HDPE) bottles of 30, 56, 1000 & 10000 tablets and in blister pack of 28 & 30 tablets.Initial dosing pack: 53 counts - Includes 0.5 mg tablets in blister strips of 11 tablets and 1 mg tablets in blister strips of 42 tablets.
Continuation dosing pack: 1 mg tablets in blister strips of 56 tablets
Nonmedicinal ingredients are anhydrous dibasic calcium phosphate, magnesium stearate, and microcrystalline cellulose. The film-coating contains hydroxypropyl cellulose, hydroxypropyl methylcellulose, titanium dioxide and triacetin. The 1 mg tablet also contains indigotine aluminum lake 12-14% as a colouring agent.Contraindications 4 CONTRAINDICATIONS
CHANTIX is contraindicated in patients with a known history of serious hypersensitivity reactions or skin reactions to CHANTIX.CONTRAINDICATIONS
Patients who are hypersensitive to varenicline or to any ingredient in the formulation or component of the container.Warnings and Precautions 5 WARNINGS AND PRECAUTIONS
5.1 Neuropsychiatric Adverse Events including Suicidality
Serious neuropsychiatric adverse events have been reported in patients being treated with CHANTIX [see Adverse Reactions (6.2)]. These postmarketing reports have included changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide. Some patients who stopped smoking may have been experiencing symptoms of nicotine withdrawal, including depressed mood. Depression, rarely including suicidal ideation, has been reported in smokers undergoing a smoking cessation attempt without medication. However, some of these adverse events occurred in patients taking CHANTIX who continued to smoke.
Neuropsychiatric adverse events occurred in patients without and with pre-existing psychiatric disease; some patients experienced worsening of their psychiatric illnesses. Some neuropsychiatric adverse events, including unusual and sometimes aggressive behavior directed to oneself or others, may have been worsened by concomitant use of alcohol [see Warnings and Precautions (5.3), Adverse Reactions (6.2)]. Observe patients for the occurrence of neuropsychiatric adverse events. Advise patients and caregivers that the patient should stop taking CHANTIX and contact a healthcare provider immediately if agitation, depressed mood, or changes in behavior or thinking that are not typical for the patient are observed, or if the patient develops suicidal ideation or suicidal behavior. The healthcare provider should evaluate the severity of the symptoms and the extent to which the patient is benefiting from treatment, and consider options including dose reduction, continued treatment under closer monitoring, or discontinuing treatment. In many postmarketing cases, resolution of symptoms after discontinuation of CHANTIX was reported. However, the symptoms persisted in some cases; therefore, ongoing monitoring and supportive care should be provided until symptoms resolve.
The neuropsychiatric safety of CHANTIX was evaluated in a randomized, double-blind, active and placebo-controlled study that included patients without a history of psychiatric disorder (non-psychiatric cohort, N=3912) and patients with a history of psychiatric disorder (psychiatric cohort, N=4003). In the non-psychiatric cohort, CHANTIX was not associated with an increased incidence of clinically significant neuropsychiatric adverse events in a composite endpoint comprising anxiety, depression, feeling abnormal, hostility, agitation, aggression, delusions, hallucinations, homicidal ideation, mania, panic, and irritability. In the psychiatric cohort, therewere more events reported in each treatment group compared to the non-psychiatric cohort, and the incidence of events in the composite endpoint was higher for each of the active treatments compared to placebo: Risk Differences (RDs) (95%CI) vs. placebo were 2.7% (-0.05, 5.4) for CHANTIX, 2.2% (-0.5, 4.9) for bupropion, and 0.4% (-2.2, 3.0) for transdermal nicotine. In the non-psychiatric cohort, neuropsychiatric adverse events of a serious nature were reported in 0.1% of CHANTIX-treated patients and 0.4% of placebo-treated patients. In the psychiatric cohort, neuropsychiatric events of a serious nature were reported in 0.6% of CHANTIX-treated patients, with 0.5% involving psychiatric hospitalization. In placebo-treated patients, serious neuropsychiatric events occurred in 0.6%, with 0.2% requiring psychiatric hospitalization [see Clinical Studies (14.10)].
WARNINGS AND PRECAUTIONS
Psychiatric Symptoms (in Patients with and without Pre-existing Psychiatric Disorder or Symptoms) (see also ADVERSE REACTIONS, Post-Marketing Experience)
There have been post-marketing reports of serious neuropsychiatric symptoms in patients being treated with varenicline, including anxiety, psychosis, mood swings, depressed mood, agitation, aggression, hostility, changes in behavior or thinking, suicidal ideation, suicidal behavior and suicide, as well as worsening of pre-existing psychiatric disorder (previously diagnosed or not). Not all patients had stopped smoking at the time of onset of symptoms, and not all patients had known pre-existing psychiatric illness, or were using concomitant CNS drugs.
Randomized Study Data: A large randomized, double-blind, active and placebo-controlled study (“EAGLES” study) was conducted to compare the risk of serious neuropsychiatric events in patients with and without a history of psychiatric disorder treated for smoking cessation with varenicline, bupropion, nicotine replacement therapy patch (NRT) or placebo. The primary safety endpoint was a composite of neuropsychiatric adverse events that have been reported in post-marketing experience. The findings were that the use of varenicline, in patients with or without a history of psychiatric disorder, was not associated with an increased risk of serious neuropsychiatric adverse events in the composite primary endpoint compared with placebo (See ACTION AND CLINICAL PHARMACOLOGY, Special Populations and Conditions, Neuropsychiatric Safety Study in Subjects with and without a History of Psychiatric Disorder).
Recommendations: Clinicians should be aware of the possible emergence of serious neuropsychiatric symptoms in patients attempting to quit smoking, with or without treatment.
Alcohol Intake: There have been post-marketing reports of patients experiencing increased intoxicating effects of alcohol while taking varenicline. Some cases described unusual and sometimes aggressive behaviour, and were often accompanied by amnesia for the events.
Pre-existing Psychiatric Disorder or Symptoms: Smoking cessation, with or without pharmacotherapy, has been associated with exacerbation of underlying psychiatric illness (e.g. depression, anxiety). Patients with a history of psychiatric symptoms should be monitored for worsening or new symptoms when attempting to quit smoking, regardless of how well controlled symptoms may be when starting smoking cessation treatment. Patients should be instructed to report strongly atypical and concerning symptoms to their healthcare provider, so that dose adjustments of psychiatric medications or APO-VARENICLINE may be considered.General: Patients should be informed that if they experience thoughts, moods or behaviours that are strongly atypical and concerning while on smoking-cessation medication, including APO-VARENICLINE, the medication should be discontinued immediately, with urgent medical help sought as needed, and the symptoms reported to their healthcare provider.
5.2 Seizures
During clinical trials and the postmarketing experience, there have been reports of seizures in patients treated with CHANTIX. Some patients had no history of seizures, whereas others had a history of seizure disorder that was remote or well-controlled. In most cases, the seizure occurred within the first month of therapy. Weigh this potential risk against the potential benefits before prescribing CHANTIX in patients with a history of seizures or other factors that can lower the seizure threshold. Advise patients to discontinue CHANTIX and contact a healthcare provider immediately if they experience a seizure while on treatment [see Adverse Reactions (6.2)].Seizures
In clinical trials and post-marketing experience there have been reports of seizures in patients treated with varenicline. Some patients had no history of seizures, whereas others had a history of seizure disorder that was remote or well-controlled. APO-VARENICLINE should be used cautiously in patients with a history of seizures or other conditions that potentially lower the seizure threshold. Advise patients to discontinue APO-VARENICLINE and immediately contact a healthcare provider if they experience a seizure while on treatment (see Special Populations, Use of APO-VARENICLINE in Patients with Concomitant Conditions).5.3 Interaction with Alcohol
There have been postmarketing reports of patients experiencing increased intoxicating effects of alcohol while taking CHANTIX. Some cases described unusual and sometimes aggressive behavior, and were often accompanied by amnesia for the events. Advise patients to reduce the amount of alcohol they consume while taking CHANTIX until they know whether CHANTIX affects their tolerance for alcohol [see Adverse Reactions (6.2)].5.4 Accidental Injury
There have been postmarketing reports of traffic accidents, near-miss incidents in traffic, or other accidental injuries in patients taking CHANTIX. In some cases, the patients reported somnolence, dizziness, loss of consciousness or difficulty concentrating that resulted in impairment, or concern about potential impairment, in driving or operating machinery. Advise patients to use caution driving or operating machinery or engaging in other potentially hazardous activities until they know how CHANTIX may affect them.Accidental Injury, including while Driving, Operating Machinery
There have been post-marketing reports of traffic accidents, near-miss incidents in traffic, and other accidental injuries in patients taking varenicline. In some cases, the patients reported somnolence, dizziness, loss of consciousness (blackouts), seizures or difficulty concentrating.
Therefore, patients should be advised not to engage in potentially hazardous activities, such as driving a car or operating dangerous machines, until they know how APO-VARENICLINE may affect them.5.5 Cardiovascular Events
A comprehensive evaluation of cardiovascular (CV) risk with CHANTIX suggests that patients with underlying CV disease may be at increased risk; however, these concerns must be balanced with the health benefits of smoking cessation. CV risk has been assessed for CHANTIX in randomized controlled trials (RCT) and meta-analyses of RCTs. In a smoking cessation trial in patients with stable CV disease, CV events were infrequent overall; however, nonfatal myocardial infarction (MI) and nonfatal stroke occurred more frequently in patients treated with CHANTIX compared to placebo. All-cause and CV mortality was lower in patients treated with CHANTIX [see Clinical Studies (14.8)]. This study was included in a meta-analysis of 15 CHANTIX efficacy trials in various clinical populations that showed an increased hazard ratio for Major Adverse Cardiovascular Events (MACE) of 1.95; however, the finding was not statistically significant (95% CI: 0.79, 4.82). In the large postmarketing neuropsychiatric safety outcome trial, an analysis of adjudicated MACE events was conducted for patients while in the trial and during a 28-week non-treatment extension period. Few MACE events occurred during the trial; therefore, the findings did not contribute substantively to the understanding of CV risk with CHANTIX. Instruct patients to notify their healthcare providers of new or worsening CV symptoms and to seek immediate medical attention if they experience signs and symptoms of MI or stroke [see Clinical Studies (14.10)].
Cardiovascular Events
In a placebo-controlled smoking cessation clinical trial in patients with stable cardiovascular disease (CVD), patients were treated with varenicline 1 mg BID or placebo for 12 weeks, and then followed for another 40 weeks. There were approximately 350 patients per arm. Serious cardiovascular (CV) events that were reported more frequently in varenicline compared to placebo (difference > 2 subjects) were: non-fatal myocardial infarctions (4 vs. 1, on-treatment phase) and need for coronary revascularization (7 vs. 2, post-treatment phase). The total number of patients that experienced serious CV events in varenicline compared to placebo was: 10 vs. 9 on treatment phase, 16 vs. 11 post-treatment phase, for a total of 25 vs. 20 over the 52 week duration. The serious CV events occurring during the treatment and post-treatment phases were adjudicated by an independent blinded committee.
The study was powered for assessing efficacy (i.e. quit rates) but not for assessing differences in the occurrence of serious CV events between varenicline and placebo. Therefore, the study was not large enough to allow conclusions regarding the difference in the incidence of CV events reported in the two arms (See also ADVERSE EVENTS, Clinical Trial in Special Populations; and ACTION AND CLINICAL PHARMACOLOGY, Special Population). Physicians are to inform patients of the symptoms of a heart attack and stroke, and instruct them to get emergency medical help right away if they experience any of these symptoms (see also Patient Counselling Information).
The CV safety of varenicline was also evaluated in the Cardiovascular Safety Assessment Study in subjects with and without a history of psychiatric disorder that randomized subjects 1:1:1:1 to varenicline 1 mg BID, bupropion SR 150 mg BID, nicotine replacement therapy patch (NRT) 21 mg/day with taper or placebo for a treatment period of 12 weeks. Subjects were then followed post-treatment through a period of up to a total of 52 weeks (See ACTION AND CLINICAL PHARMACOLOGY, Special Populations and Conditions, Cardiovascular Safety Assessment Study in Subjects with and without a History of Psychiatric Disease). Major CV events (CV death, non-fatal MI, non-fatal stroke) were infrequent overall (1/2016 and 4/2014, for patients treated with varenicline and placebo, respectively) during the treatment period. However, because of the relatively low number of events overall and the lack of power for assessing differences between varenicline and placebo, an association between the use of varenicline and an increased risk of CV adverse events cannot be entirely ruled out.Varenicline has not been studied in patients with unstable cardiovascular disease or those with cardiovascular events occurring within two months before study screening. Patients should be advised to notify a health care provider of new or worsening symptoms of cardiovascular disease. The risks of APO-VARENICLINE should be weighed against the benefits of its use in smokers with cardiovascular disease. Smoking is an independent and major risk factor for cardiovascular disease. Varenicline has been demonstrated to increase the likelihood of abstinence from smoking for as long as one year compared to treatment with placebo.
5.6 Somnambulism
Cases of somnambulism have been reported in patients taking CHANTIX. Some cases described harmful behavior to self, others, or property. Instruct patients to discontinue CHANTIX and notify their healthcare provider if they experience somnambulism [see Adverse Reactions (6.2)].Somnambulism
Cases of somnambulism have been reported post-marketing in patients taking varenicline. Some cases described harmful behavior to self, others, or property. Instruct patients to discontinue APO-VARENICLINE and notify their healthcare provider if they experience somnambulism.5.7 Angioedema and Hypersensitivity Reactions
There have been postmarketing reports of hypersensitivity reactions including angioedema in patients treated with CHANTIX [see Adverse Reactions (6.2), Patient Counseling Information (17)]. Clinical signs included swelling of the face, mouth (tongue, lips, and gums), extremities, and neck (throat and larynx). There were infrequent reports of life-threatening angioedema requiring emergent medical attention due to respiratory compromise. Instruct patients to discontinue CHANTIX and immediately seek medical care if they experience these symptoms.Angioedema and Hypersensitivity reactions
There have been post-marketing reports of hypersensitivity reactions, including angioedema, in patients treated with varenicline (see ADVERSE REACTIONS, Post-Marketing Experience). Clinical signs included swelling of the face, mouth (tongue, lips and gums), neck (pharynx and larynx) and extremities. There were rare reports of life-threatening angioedema requiring urgentmedical attention due to respiratory compromise. Patients experiencing these symptoms should be instructed to discontinue treatment with APO-VARENICLINE and contact a healthcare provider immediately.5.8 Serious Skin Reactions
There have been postmarketing reports of rare but serious skin reactions, including Stevens-Johnson Syndrome and erythema multiforme, in patients using CHANTIX [see Adverse Reactions (6.2)]. As these skin reactions can be life-threatening, instruct patients to stop taking CHANTIX and contact a healthcare provider immediately at the first appearance of a skin rash with mucosal lesions or any other signs of hypersensitivity.Serious Skin Reactions
There have also been post-marketing reports of rare but severe cutaneous reactions, including Stevens-Johnson syndrome and erythema multiforme, in patients using varenicline (see ADVERSE REACTIONS, Post-Marketing Experience). As these skin reactions can be life-threatening, patients should be instructed to discontinue treatment at the first sign of rash or skin reaction and contact a healthcare provider immediately.5.9 Nausea
Nausea was the most common adverse reaction reported with CHANTIX treatment. Nausea was generally described as mild or moderate and often transient; however, for some patients, it was persistent over several months. The incidence of nausea was dose-dependent. Initial dose-titration was beneficial in reducing the occurrence of nausea. For patients treated to the maximum recommended dose of 1 mg twice daily following initial dosage titration, the incidence of nausea was 30% compared with 10% in patients taking a comparable placebo regimen. In patients taking CHANTIX 0.5 mg twice daily following initial titration, the incidence was 16% compared with11% for placebo. Approximately 3% of patients treated with CHANTIX 1 mg twice daily in studies involving 12 weeks of treatment discontinued treatment prematurely because of nausea. For patients with intolerable nausea, a dose reduction should be considered.Nausea
Nausea was the most common adverse event associated with varenicline treatment. Nausea was generally described as mild or moderate and often transient; however, for some subjects, it was persistent over several months. The incidence of nausea was dose-dependent. Initial dose-titration was beneficial in reducing the occurrence of nausea. Nausea was reported by approximately 30% of patients treated with varenicline 1 mg BID after an initial week of dose titration. In patients taking varenicline 0.5 mg BID, the incidence of nausea was 16% following initial titration. Approximately 3% of subjects treated with varenicline 1 mg BID in studies involving 12 weeks of treatment discontinued treatment prematurely because of nausea. For patients with intolerable nausea, dose reduction should be considered (see DOSAGE AND ADMINISTRATION, Recommended Dose and Dosage Adjustment).
Adverse Reactions
6 ADVERSE REACTIONSThe following serious adverse reactions were reported in postmarketing experience and are discussed in greater detail in other sections of the labeling: - Neuropsychiatric Adverse Events including Suicidality [see Warnings and Precautions (5.1)]
- Seizures [see Warnings and Precautions (5.2)]
- Interaction with Alcohol [see Warnings and Precautions (5.3)]
- Accidental Injury [see Warnings and Precautions (5.4)]
- Cardiovascular Events [see Warnings and Precautions (5.5)]
- Somnambulism [see Warnings and Precautions (5.6)]
- Angioedema and Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Serious Skin Reactions [see Warnings and Precautions (5.8)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, the adverse reactions rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During the premarketing development of CHANTIX, over 4500 subjects were exposed to CHANTIX, with over 450 treated for at least 24 weeks and approximately 100 for a year. Most study participants were treated for 12 weeks or less.
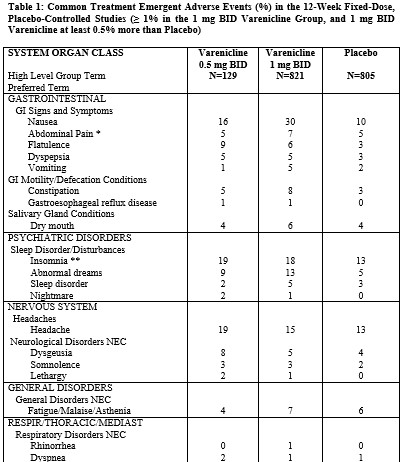
The most common adverse event associated with CHANTIX treatment is nausea, occurring in 30% of patients treated at the recommended dose, compared with 10% in patients taking a comparable placebo regimen [see Warnings and Precautions (5.9)]. Table 1 shows the adverse events for CHANTIX and placebo in the 12- week fixed dose premarketing studies with titration in the first week [Studies 2 (titrated arm only), 4, and 5]. Adverse events were categorized using the Medical Dictionary for Regulatory Activities (MedDRA, Version 7.1).
MedDRA High Level Group Terms (HLGT) reported in ≥5% of patients in the CHANTIX 1 mg twice daily dose group, and more commonly than in the placebo group,are listed, along with subordinate Preferred Terms (PT) reported in ≥1% of CHANTIX patients (and at least 0.5% more frequent than placebo). Closely related Preferred Terms such as ‘Insomnia’, ‘Initial insomnia’, ‘Middle insomnia’, ‘Early morning awakening’ were grouped, but individual patients reporting two or more grouped events are only counted once.
Clinical Trial Adverse Drug Reactions
Smoking-cessation with or without treatment is associated with various symptoms. For example, dysphoric or depressed mood, insomnia, irritability, frustration or anger, anxiety, difficulty concentrating, restlessness, decreased heart rate, increased appetite or weight gain have been reported in patients attempting to stop smoking.
Overview
Pre-marketing clinical trials included approximately 2300 patients treated for at least 12 weeks, approximately 700 for 6 months, and approximately 100 for one year. In general, onset of adverse events was in the first few weeks of therapy and severity was generally mild to moderate. No differences were observed by age, race or gender with regard to the incidence of adverse reactions, although patient numbers in elderly, and in non-caucasian races were too limited to allow conclusions.Commonly Observed Adverse Events
The most commonly observed adverse events associated with varenicline (> 5% and twice the rate seen in placebo-treated patients) were nausea, abnormal dreams, constipation, flatulence, and vomiting. For patients exposed to the maximum recommended dose of 1 mg BID following initial dosage titration, the incidence of nausea was 30%, compared with 16% in 0.5 mg BID and approximately 10% in placebo-treated patients. Nausea was generally described as mild to moderate and often transient; however, for some subjects, it was persistent throughout the treatment period.
Adverse Events Leading to Discontinuation
In Phase 2 and 3 placebo-controlled studies, the treatment discontinuation rate due to adverse events in patients randomized to 12 weeks treatment with the recommended maximum dose of 1 mg BID was 12% for varenicline compared to 10% for placebo. In this group, the adverse events most frequently resulting in treatment discontinuation in varenicline treated patients were as follows: nausea (2.7% vs 0.6% for placebo), insomnia (1.3% vs 1.2% for placebo), fatigue/malaise/asthenia (1.0% vs 0.5% for placebo), and dizziness (0.7% vs 0.4% for placebo).
Table 1 shows the adverse events for varenicline and placebo in the 12-week fixed dose studies with titration in the first week (Studies 1 (titrated arm only), 3, and 4). MedDRA High Level Group Terms (HLGT) reported in ≥ 5% of patients in the varenicline 1 mg BID dose group, and more commonly than in the placebo group, are listed, along with subordinate Preferred Terms (PT) reported in ≥ 1% of varenicline patients (and at least 0.5% more frequently than placebo). Closely related Preferred Terms such as ‘Insomnia’, ‘Initial insomnia’, ‘Middle insomnia’, ‘Early morning awakening’ were grouped, but individual patients reporting two or more grouped events were only counted once.Table 1. Common Treatment Emergent AEs (%) in the Fixed-Dose, Placebo-Controlled Studies (HLGTs >5% of Patients in the 1 mg BID CHANTIX Group and More Commonly than Placebo and PT ≥1% in the 1 mg BID CHANTIX Group, and 1 mg BID CHANTIX at Least 0.5% More than Placebo)
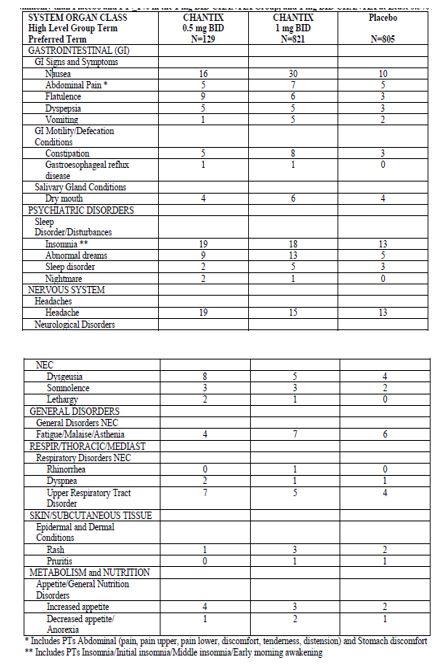
The overall pattern and frequency of adverse events during the longer-term premarketing trials was similar to those described in Table 1, though several of the most common events were reported by a greater proportion of patients with long-term use (e.g., nausea was reported in 40% of patients treated with CHANTIX 1 mg twice daily in a one year study, compared to 8% of placebo-treated patients).
Following is a list of treatment-emergent adverse events reported by patients treated with CHANTIX during all premarketing clinical trials and updated based on pooleddata from 18 placebo-controlled pre- and postmarketing studies, including approximately 5,000 patients treated with varenicline. Adverse events were categorized using MedDRA, Version 16.0. The listing does not include those events already listed in the previous tables or elsewhere in labeling, those events for which a drug cause was remote, those events which were so general as to be uninformative, and those events reported only once which did not have a substantial probability of being acutely life-threatening.Additional Clinical Trial Adverse Drug Reactions
The adverse drug reactions listed below are based on evaluation of data from pre-marketing phase 2-3 studies and updated based on a pooled database of a total of 18 placebo-controlled, pre- and post-marketing smoking cessation studies, with approximately 5,000 patients treated with varenicline. All reported events are included except those already listed in Table 1, those too general to be informative, and those not reasonably possibly associated with the use of the drug. In some cases, separate event terms have been consolidated to facilitate meaningful presentation. It is important to emphasize that although the events reported occurred during treatment with varenicline they were not necessarily caused by it.The ADRs listed below are presented by the Medical Dictionary for Regulatory Activities (MedDRA, Version 16) System Organ Class (SOC). The variability associated with adverse event reporting and the terminology used to describe adverse events limit the value of the quantitative frequency estimates provided. Events are further classified within system organ class categories and enumerated in order of decreasing frequency using the following definitions: very frequent (occurring in at least 1/10 patients), frequent (occurring in at least 1/100 patients), infrequent (occurring in < 1/100 to 1/1000 patients) and rare (occurring in fewer than 1/1000 patients).Blood and Lymphatic System Disorders. Infrequent: anemia, lymphadenopathy. Rare: leukocytosis, splenomegaly, thrombocytopenia.
Cardiac Disorders.Infrequent: angina pectoris, myocardial infarction, palpitations, tachycardia. Rare: acute coronary syndrome, arrhythmia, atrial fibrillation,bradycardia, cardiac flutter, cor pulmonale, coronary artery disease, ventricular extrasystoles.
Ear and Labyrinth Disorders.Infrequent: tinnitus, vertigo. Rare: deafness, Meniere’s disease.
Endocrine Disorders.Infrequent: thyroid gland disorders.
Eye Disorders.Infrequent: conjunctivitis, eye irritation, eye pain, vision blurred, visual impairment. Rare: blindness transient, cataract subcapsular, dry eye, nightblindness, ocular vascular disorder, photophobia, vitreous floaters.
Gastrointestinal Disorders.Frequent: diarrhea, toothache. Infrequent: dysphagia, eructation, gastritis, gastrointestinal hemorrhage, mouth ulceration. Rare: enterocolitis, esophagitis, gastric ulcer, intestinal obstruction, pancreatitis acute.
General Disorders and Administration Site Conditions. Frequent: chest pain. Infrequent: chest discomfort, chills, edema, influenza-like illness, pyrexia.
Hepatobiliary Disorders.Rare: gall bladder disorder.Investigations. Frequent: liver function test abnormal, weight increased. Infrequent: electrocardiogram abnormal. Rare: muscle enzyme increased, urine analysisabnormal.
Metabolism and Nutrition Disorders. Infrequent: diabetes mellitus, hypoglycemia. Rare: hyperlipidemia, hypokalemia.
Musculoskeletal and Connective Tissue Disorders. Frequent: arthralgia, back pain, myalgia. Infrequent: arthritis, muscle cramp, musculoskeletal pain. Rare: myositis, osteoporosis.
Nervous System Disorders.Frequent: disturbance in attention, dizziness. Infrequent: amnesia, convulsion, migraine, parosmia, syncope, tremor. Rare: balance disorder, cerebrovascular accident, dysarthria, mental impairment, multiple sclerosis, VIIth nerve paralysis, nystagmus, psychomotor hyperactivity, psychomotor skills impaired, restless legs syndrome, sensory disturbance, transient ischemic attack, visual field defect.
Psychiatric Disorders.Infrequent: dissociation, libido decreased, mood swings, thinking abnormal. Rare: bradyphrenia, disorientation, euphoric mood.
Renal and Urinary Disorders.Infrequent: nocturia, pollakiuria, urine abnormality. Rare: nephrolithiasis, polyuria, renal failure acute, urethral syndrome, urinaryretention.
Reproductive System and Breast Disorders.Frequent: menstrual disorder. Infrequent: erectile dysfunction. Rare: sexual dysfunction.
Respiratory, Thoracic and Mediastinal Disorders.Frequent: respiratory disorders. Infrequent: asthma, epistaxis, rhinitis allergic, upper respiratory tract inflammation. Rare: pleurisy, pulmonary embolism.
Skin and Subcutaneous Tissue Disorders.Infrequent: acne, dry skin, eczema, erythema, hyperhidrosis, urticaria.
Rare: photosensitivity reaction, psoriasis.
Vascular Disorders.Infrequent: hot flush. Rare: thrombosis.Blood and Lymphatic System Disorders: Infrequent: Anemia, Lymphadenopathy. Rare: Leukocytosis, Platelet count decreased, Thrombocytopenia, Splenomegaly.
Cardiac Disorders: Infrequent:Angina pectoris, Electrocardiogram abnormal, Heart rate increased, Myocardial infarction, Palpitations, Tachycardia. Rare:Arrhythmia, Atrial fibrillation, Bradycardia, Cardiac flutter, Coronary artery disease, Cor pulmonale, Acute coronary syndrome, Electrocardiogram ST segment depression, Electrocardiogram T wave amplitude decreased, Ventricular extrasystoles.
Ear and Labyrinth Disorders: Infrequent:Tinnitus, Vertigo. Rare:Deafness, Meniere’s disease. Endocrine Disorders: Infrequent:Thyroid gland disorders.
Eye Disorders: Infrequent: Conjunctivitis, Eye irritation, Vision blurred, Visual impairment, Eye pain. Rare:Acquired night blindness, Blindness transient, Cataract subcapsular, Dry eye, Mydriasis, Myopia, Lacrimation increased, Ocular vascular disorder, Photophobia, Scleral discolouration, Scotoma, Vitreous floaters.
Gastrointestinal Disorders: Frequent:Diarrhea, Toothache. Infrequent:Change of bowel habit, Aphthous stomatitis, Gingival pain, Dysphagia, Eructation, Gastritis, Gastrointestinal hemorrhage, Hematochezia, Mouth ulceration. Rare:Abnormal feces, Enterocolitis, Esophagitis, Gastric ulcer, Hematemesis, Intestinal obstruction, Pancreatitis acute, Tongue coated.
General Disorders and Administration Site Conditions: Frequent:Chest pain, Irritability. Infrequent:Chest discomfort, Chills, Edema, Influenza like illness, Pyrexia, Thirst. Rare: Cyst, Feeling cold.
Hepatobiliary Disorders: Rare:Gall bladder disorder, Worsening of existing autoimmune hepatitis.
Immune System Disorders: Infrequent:Hypersensitivity. Rare: Drug hypersensitivity.
Infections and Infestations: Very frequent:Nasopharyngitis. Frequent: Bronchitis, Sinusitis. Infrequent: Fungal infection, Gingivitis, Viral infection, Tooth abscess, Urinary Tract Infection.
Investigations: Frequent:Liver function test abnormal, alanine aminotransferase increased, Rare:Muscle enzyme increased, Semen abnormal, C-reactive protein increased, Blood calcium decreased, Urine analysis abnormal.
Metabolism and Nutrition Disorders: Frequent: Weight increased. Infrequent: Diabetes mellitus, Hypoglycemia. Rare: Hyperkalemia, Hyperlipidemia, Hypokalemia, Polydipsia.
Musculoskeletal and Connective Tissue Disorders: Frequent: Arthralgia, Back pain, Myalgia. Infrequent: Arthritis, Musculoskeletal chest pain, Muscle cramp, Musculoskeletal pain, Muscle spasms. Rare:Costochondritis, Joint stiffness,Myositis, Osteoporosis.
Nervous System Disorders: Frequent:Disturbance in attention, Dizziness, Somnolence. Infrequent:Amnesia, Convulsion, Hypoesthesia, Migraine, Parosmia, Syncope, Tremor. Rare: Balance disorder, Cerebrovascular accident, Circadian rhythm sleep disorder, Coordination abnormal, Dysarthria, Hypertonia, Hypogeusia, Mental impairment, Multiple sclerosis, VIIth nerve paralysis, Nystagmus, Psychomotor hyperactivity, Psychomotor skills impaired, Restless legs syndrome, Sensory disturbance, Transient ischemic attack, Visual field defect.
Psychiatric Disorders: Frequent:Agitation,Anxiety, Depression. Infrequent:Aggression, Dissociation, Libido decreased, Libido increased, Mood swings, Panic reaction, Restlessness, Suicidal ideation, Thinking abnormal. Rare:Bradyphrenia, Disorientation, Dysphoria, Emotional disorder, Euphoric mood, Hallucination, Psychotic disorder, Suicide attempt.
Renal and Urinary Disorders: Infrequent: Nocturia, Pollakiuria, Urine abnormality.Rare:Glycosuria, Nephrolithiasis, Polyuria,Renal failure acute, Urethral syndrome, Urinary retention.
Reproductive System and Breast Disorders: Frequent:Menstrual disorder. Infrequent:Erectile dysfunction, Menorrhagia. Rare: Sexual dysfunction, Vaginal discharge.
Respiratory, Thoracic and Mediastinal Disorders: Frequent:Cough, Respiratory disorders. Infrequent:Asthma, Dysphonia, Epistaxis, Rhinitis allergic, Throat irritation, Respiratory tract congestion, Sinus congestion, Rhinorrhea, Upper-airway cough syndrome, Upper respiratory tract inflammation. Rare: Laryngeal pain, Pleurisy, Pulmonary embolism, Snoring.
Skin and Subcutaneous Tissue Disorders: Frequent:Rash. Infrequent:Acne, Dry skin, Eczema, Erythema, Hyperhidrosis, Night sweats, Urticaria. Rare: Dermatitis, Photosensitivity reaction, Psoriasis.
Vascular Disorders: Frequent: Hypertension. Infrequent:Blood pressure increased, Hot flush, Hypotension.Rare: Peripheral ischemia, Thrombosis.
CHANTIX has also been studied in postmarketing trials including (1) a trial conducted in patients with chronic obstructive pulmonary disease (COPD), (2) a trial conducted in generally healthy patients (similar to those in the premarketing studies) in which they were allowed to select a quit date between days 8 and 35 of treatment (“alternative quit date instruction trial”), (3) a trial conducted in patients who did not succeed in stopping smoking during prior CHANTIX therapy, or who relapsed after treatment (“re-treatment trial”), (4) a trial conducted in patients with stable cardiovascular disease, (5) a trial conducted in patients with stable schizophrenia or schizoaffective disorder, (6) a trial conducted in patients with major depressive disorder, (7) a postmarketing neuropsychiatric safety outcome trial in patients without or with a history of psychiatric disorder, (8) a non-treatment extension of the postmarketing neuropsychiatric safety outcome trial that assessed CV safety, (9) a trial in patients who were not able or willing to quit abruptly and who were instructed to quit gradually (“gradual approach to quitting smoking trial”).
Adverse events in the trial of patients with COPD (1), in the alternative quit date instruction trial (2), and in the gradual approach to quitting smoking trial (9) were similar to those observed in premarketing studies. In the re-treatment trial (3), the profile of common adverse events was similar to that previously reported, but, in addition, varenicline-treated patients also commonly reported diarrhea (6% vs. 4% in placebo-treated patients), depressed mood disorders and disturbances (6% vs. 1%), and other mood disorders and disturbances (5% vs. 2%).
In the trial of patients with stable cardiovascular disease (4), more types and a greater number of cardiovascular events were reported compared to premarketing studies,as shown in Table 1 and in Table 2 below.Cardiovascular Adverse Events in Pooled Clinical Studies of Varenicline
In pooled data of 14 completed randomized double-blind placebo controlled smoking cessation trials (not including the study in patients with stable cardiovascular disease), the rate of reported treatment-emergent myocardial infarction (MI) or cerebrovascular accident (CVA) related adverse events was: 8 of 3317 (0.24%) patients on varenicline (> 1 mg), compared to 4 of 2542 (0.16%) patients on placebo.
Study in patients with Cardiovascular Disease
Varenicline was evaluated in a randomized, double-blind, placebo-controlled study of 703 subjects aged 35 to 75 years with stable, documented cardiovascular disease (other than or in addition to hypertension) that had been diagnosed for more than 2 months. Patients were treated with varenicline 1 mg BID or placebo for 12 weeks, and then followed for another 40 weeks post-treatment (See WARNINGS AND PRECAUTIONS, Cardiovascular Events).
There are two partially overlapping data sets of cardiovascular events from the study:
i) Treatment-emergent CV AEs captured via standard clinical trial AE reporting, while on drug treatment, (including, 30 days post-dose); and
ii) Pre-specified serious CV events that were adjudicated by an independent blinded committee captured throughout the 52 week duration (i.e., both “on-treatment” [including 30 days post-dose], and “post-treatment”).
The study was powered for assessing efficacy (i.e. quit rates) but not for assessing differences in the occurrence of serious CV events between varenicline and placebo. More cardiovascular events were reported in both arms compared to other studies, as expected due to underlying conditions.
Treatment-emergent cardiovascular events which occurred within 30 days after the last dose, and in at least 3 subjects in either arm, are shown in Table 5.
Table 2. Cardiovascular Mortality and Nonfatal Cardiovascular Events (%) with a Frequency >1% in Either Treatment Group in the Trial of Patients with Stable Cardiovascular Disease
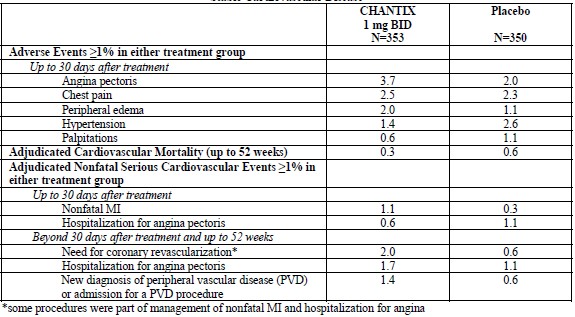

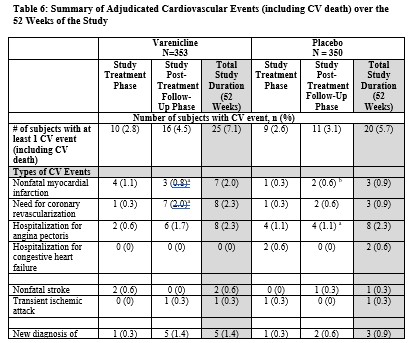
The adjudicated serious cardiovascular events are shown below in Table 6.Patients are counted only once within each row per study phase.
As shown in Table 6, the individual serious cardiovascular (CV) events that were reported more frequently in varenicline compared to placebo (difference > 2 subjects) were: non-fatal myocardial infarctions (4 vs. 1, on-treatment phase) and need for coronary revascularization (7 vs. 2, post-treatment phase). Some of the patients requiring coronary revascularization underwent the procedure as part of management of nonfatal MI and hospitalization for angina.
Varenicline was not studied in patients with unstable cardiovascular disease or those with cardiovascular events occurring within two months before screening. (See also: WARNINGS AND PRECAUTIONS, Cardiovascular Events, and ACTION AND CLINICAL PHARMACOLOGY, Special Populations and Conditions)Cardiovascular Safety Assessment Study in Patients with and without a History of Psychiatric Disorder
The cardiovascular (CV) safety of varenicline was evaluated in the Cardiovascular Safety Assessment Study in subjects with and without a history of psychiatric disorder. Subjects aged 18 to 75 years, smoking 10 or more cigarettes per day (N=8058) were randomized 1:1:1:1 to varenicline 1 mg BID, bupropion SR 150 mg BID, nicotine replacement therapy patch (NRT) 21 mg/day with taper or placebo for a treatment period of 12 weeks; they were then followed another 12 weeks post-treatment through a period of up to a total of 52 weeks. Of all treated subjects, 1749 (21.7%) had a medium CV risk and 644 (8.0%) had a high CV risk, as defined by Framingham score.
Major adverse cardiovascular event (MACE), were defined as cardiovascular death, non-fatal myocardial infarction or non-fatal stroke during treatment.Deaths and cardiovascular events were adjudicated by a blinded, independent committee. The study was not powered for assessing differences between varenicline and placebo in the time to MACE.The following table shows the incidence of MACE for all treatment groups during treatment, and cumulative for treatment plus 30 days and through end of study.
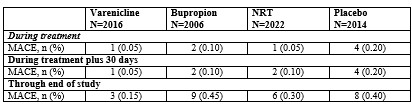
Because of the relatively low number of events overall and the lack of power for assessing differences between varenicline and placebo, an association between the use of varenicline and an increased risk of CV adverse events cannot be entirely ruled out.In the trial of patients with stable schizophrenia or schizoaffective disorder (5), 128 smokers on antipsychotic medication were randomized 2:1 to varenicline (1 mg twice daily) or placebo for 12 weeks with 12-week non-drug follow-up. The most common treatment emergent adverse events reported in this trial are shown in Table 3 below. Patients with Stable Schizophrenia or Schizoaffective Disorder (See also below: Neuropsychiatric Safety Study in Subjects with and without a History of Psychiatric Disorder)
Varenicline safety and tolerability was assessed in a double-blind study of 128 smokers with stable schizophrenia or schizoaffective disorder, on antipsychotic medication, randomized 2:1 to varenicline (1 mg twice daily) or placebo for 12 weeks with 12-week non-drug follow-up.
Assessments including the Positive and Negative Symptom Scale (PANSS), standard questioning regarding adverse events, and the Columbia Suicide Severity Rating Scale (C-SSRS) occurred weekly through week 13 and at weeks 16, 20 and 24.
Based on adverse event rates, including neuropsychiatric, there were no new safety concerns compared to studies in the general population of smokers. The study discontinuation rate due to neuropsychiatric adverse events in the varenicline arm was 4% (3 /84), compared to 0 (0 /43) in the placebo group.
In this study, there was no overall worsening of schizophrenia in either treatment group as measured by PANSS scores nor worsening of extra-pyramidal signs.
Evaluation of suicidal ideation and behavior (including C-SSRS): Reported lifetime history of suicidality was higher in the patients randomized to the varenicline arm compared to placebo [62% (52 /84) and 51% (22/43) respectively]. During the active treatment period, the rate of C-SSRS endorsement was 11% (9/82) in the varenicline arm and 9% (4/43) in the placebo arm. There were two suicide-related actions by two patients treated with varenicline (attempt through overdose, and preparatory act of collecting pills); both patients had a lifetime history of similar behaviours.
During the 12 week post-treatment phase, the rate of C-SSRS endorsement decreased in the placebo arm to 5% (2/39), while the rate in the varenicline arm remained at 11% (8 / 70). For six of the cases, all in the varenicline arm, the C-SSRS endorsements were the first in the study for those individuals and occurred more than 30 days after last treatment dose.
All incidences of suicidal ideation or behavior during the study, except for one patient treated with varenicline, occurred in patients with a prior history of suicidality.Table 3. Common Treatment Emergent AEs (%) in the Trial of Patients with Stable Schizophrenia or Schizoaffective Disorder
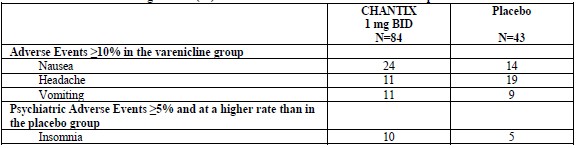
For the trial of patients with major depressive disorder (6), the most common treatment emergent adverse events reported are shown in Table 4 below. Additionally, in this trial, patients treated with varenicline were more likely than patients treated with placebo to report one of events related to hostility and aggression (3% vs. 1%). Table 4. Common Treatment Emergent AEs (%) in the Trial of Patients with Major Depressive Disorder
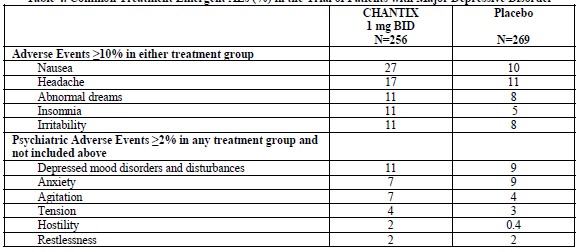
In the trial of patients without or with a history of psychiatric disorder (7), the most common adverse events in subjects treated with varenicline were similar to thoseobserved in premarketing studies. Most common treatment-emergent adverse events reported in this trial are shown in Table 5 below. Table 5. Treatment Emergent Common AEs (%) in the Trial of Patients without or with a History of Psychiatric Disorder
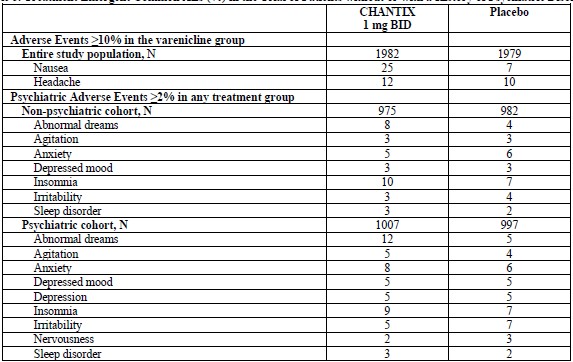
In the non-treatment extension of the postmarketing neuropsychiatric safety outcomes trial that assessed CV safety (8), the most common adverse events in subjects treated with varenicline and occurring up to 30 days after last dose of treatment were similar to those observed in premarketing studies. 6.2 Postmarketing Experience
The following adverse events have been reported during post-approval use of CHANTIX. Because these events are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
There have been reports of depression, mania, psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, anxiety, and panic, as well as suicidal ideation, suicide attempt, and completed suicide in patients attempting to quit smoking while taking CHANTIX [see Warnings and Precautions (5.1)].
There have been postmarketing reports of new or worsening seizures in patients treated with CHANTIX [see Warnings and Precautions (5.2)].
There have been postmarketing reports of patients experiencing increased intoxicating effects of alcohol while taking CHANTIX. Some reported neuropsychiatric events, including unusual and sometimes aggressive behavior [see Warnings and Precautions (5.1) and (5.3)].
There have been reports of hypersensitivity reactions, including angioedema [see Warnings and Precautions (5.7)].
There have also been reports of serious skin reactions, including Stevens-Johnson Syndrome and erythema multiforme, in patients taking CHANTIX [see Warnings and Precautions (5.8)].
There have been reports of myocardial infarction (MI) and cerebrovascular accident (CVA) including ischemic and hemorrhagic events in patients taking CHANTIX. In the majority of the reported cases, patients had pre-existing cardiovascular disease and/or other risk factors. Although smoking is a risk factor for MI and CVA, based on temporal relationship between medication use and events, a contributory role of varenicline cannot be ruled out [see Warnings and Precautions (5.5)].
There have been reports of hyperglycemia in patients following initiation of CHANTIX. There have been reports of somnambulism, some resulting in harmful behavior to self, others, or property in patients treated with CHANTIX [see Warnings and Precautions (5.6)].Post-Marketing Experience
The following adverse events have been reported during post-approval use of varenicline. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Psychiatric Symptoms
There have been reports of depressed mood, agitation, aggression, hostility, anxiety, changes in behavior or thinking, mania, psychosis, hallucinations, paranoia, delusions, homicidal ideation, mood swings, suicidal ideation and completed suicide in patients attempting to quit smoking while taking varenicline (see WARNINGS AND PRECAUTIONS, Psychiatric Symptoms in Patients with and without Pre-existing Psychiatric Disorder or Symptoms). Of the cases with information provided, the majority reported possible contributing factors, including primarily prior psychiatric history and/or concurrent psychiatric medications. Smoking status at the time of event onset was not reported in most cases. Patients should be advised that drinking alcohol may increase the risk of experiencing psychiatric adverse events. Smoking cessation with or without treatment is associated with nicotine withdrawal symptoms and the exacerbation of underlying psychiatric illness. The role of varenicline in these reports is not known (see also WARNINGS AND PRECAUTIONS, Psychiatric Symptoms in Patients with and without Pre-existing Psychiatric Disorder or Symptoms).
Hypersensitivity and Serious Skin Reactions
There have also been reports of hypersensitivity reactions, including angioedema and of rare but severe cutaneous reactions including Stevens-Johnson syndrome and erythema multiforme in patients taking varenicline (see WARNINGS AND PRECAUTIONS, Angioedema and Hypersensitivity Reactions and Serious Skin Reactions).
Myocardial Infarction and Cerebrovascular Accident
There have been reports of myocardial infarction (MI) and cerebrovascular accident (CVA) including ischemic and hemorrhagic events in patients taking varenicline. In the majority of the reported cases, patients had preexisting cardiovascular disease and/or other risk factors. Although smoking is a risk factor for MI and CVA, a contributory role of varenicline cannot be ruled out, based on temporal relationship between medication use and events.
Hyperglycemia and Diabetes Mellitus
Smoking cessation, with or without treatment, may be associated with altered glycemic control. There have been reports of hyperglycemia in patients taking varenicline. While the majority of these cases involved diabetic patients experiencing loss of glycemic control (see Special Populations, Patients with Diabetes), there have also been reports of new onset diabetes in patients with no pre-existing diabetes or pre-diabetes.Drug Interactions 7 DRUG INTERACTIONS
Based on varenicline characteristics and clinical experience to date, CHANTIX has no clinically meaningful pharmacokinetic drug interactions [see Clinical Pharmacology (12.3)].
DRUG INTERACTIONS
Overview
Based on varenicline pharmacokinetic characteristics, and clinical experience to date, it appears unlikely that varenicline would produce or be subject to clinically meaningful drug interactions. Drug interaction studies were performed with varenicline and: cimetidine, metformin, digoxin, warfarin, transdermal nicotine and bupropion.No clinically meaningful pharmacokinetic drug interactions have been identified, other than potential for interaction with cimetidine in patients with severe renal impairment (see Cimetidine, below).Drugs cleared by, or which affect, cytochrome P450 enzymes
In vitro studies demonstrated that varenicline does not inhibit cytochrome P450 enzymes (IC50 > 6400 ng/mL). The P450 enzymes tested for inhibition were: 1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A4/5. Also, in human hepatocytes in vitro, varenicline did not induce the activity of cytochrome P450 enzymes 1A2 and 3A4. Therefore, varenicline is unlikely to alter the pharmacokinetics of compounds that are primarily metabolized by cytochrome P450 enzymes.
Furthermore, since metabolism of varenicline represents less than 10% of its clearance, drugs known to affect the cytochrome P450 system are unlikely to alter the pharmacokinetics of varenicline (see ACTION AND CLINICAL PHARMACOLOGY: Pharmacokinetics) and therefore a dose adjustment of APO-VARENICLINE should not be required for these types of drugs.
Drugs cleared by, or which affect, renal secretion
In vitro studies demonstrated that varenicline does not inhibit human renal transport proteins at therapeutic concentrations. Therefore, drugs that are cleared by renal secretion (e.g., metformin - see below) are unlikely to be affected by varenicline.In vitro studies demonstrated the active renal secretion of varenicline is mediated by the human organic cation transporter, hOCT2. In patients with normal renal function coadministration with inhibitors of hOCT2 does not require a dose adjustment of varenicline as the increase in systemic exposure to varenicline is not expected to be clinically meaningful except in cases of severe renal impairment (see Cimetidine, and Other Inhibitors of hOCT2 below).7.1 Use with Other Drugs for Smoking Cessation
Safety and efficacy of CHANTIX in combination with other smoking cessation therapies have not been studied.
Bupropion
Varenicline (1 mg twice daily) did not alter the steady-state pharmacokinetics of bupropion (150 mg twice daily) in 46 smokers. The safety of the combination of bupropion and varenicline has not been established.
Nicotine replacement therapy (NRT)
Although co-administration of varenicline (1 mg twice daily) and transdermal nicotine (21 mg/day) for up to 12 days did not affect nicotine pharmacokinetics, the incidence of nausea, headache, vomiting, dizziness, dyspepsia, and fatigue was greater for the combination than for NRT alone. In this study, eight of twenty-two (36%) patients treated with the combination of varenicline and NRT prematurely discontinued treatment due to adverse events, compared to 1 of 17 (6%) of patients treated with NRT and placebo.
Use with other therapies for smoking-cessation:
Safety and efficacy of varenicline in combination with other smoking-cessation therapies, such as bupropion or nicotine replacement therapy, have not been studied.
Bupropion: Varenicline (1 mg BID) did not alter the steady-state pharmacokinetics of bupropion (150 mg BID) in 46 smokers. Steady-state pharmacokinetics of varenicline remained unchanged by bupropion co-administration.
Nicotine replacement therapy (NRT): When varenicline (1 mg BID) and NRT (transdermal, 21 mg/day) were co-administered to 24 smokers for 12 days, there was a statistically significant decrease in average systolic blood pressure (mean 2.6 mmHg) measured on the final day of the study. In this study, the incidence of nausea, headache, vomiting, dizziness, dyspepsia and fatigue were greater for the combination of varenicline and NRT than for NRT alone. Due to the partial agonist nicotinic activity of varenicline, it is not anticipated that co-administration with NRT would confer additional benefits compared with varenicline alone, and may result in increased side effects (see WARNINGS AND PRECAUTIONS).
Nicotine replacement therapy (NRT)
The concomitant use of NRT with APO-VARENICLINE (varenicline tartrate) may result in an increase in adverse reactions. In a clinical drug interaction study (N=24), the incidences of nausea, headache, vomiting, dizziness, dyspepsia and fatigue were greater for the combination of NRT and varenicline than for NRT alone (see DRUG INTERACTIONS). The safety and efficacy of the combination treatment with varenicline and NRT have not been studied. Due to the proposed mechanism of action of varenicline, it is not anticipated that co-administration with NRT would confer additional benefit compared with varenicline alone.
7.2 Effect of Smoking Cessation on Other Drugs
Physiological changes resulting from smoking cessation, with or without treatment with CHANTIX, may alter the pharmacokinetics or pharmacodynamics of certain drugs (e.g., theophylline, warfarin, insulin) for which dosage adjustment may be necessary.Effect of smoking-cessation
Physiological changes resulting from smoking-cessation, with or without treatment with APO-VARENICLINE, may alter the pharmacokinetics or pharmacodynamics of some drugs for which dosage adjustment may be necessary (examples include theophylline, warfarin and insulin). As smoking induces cytochrome P450 (CYP) isoenzyme 1A2, smoking-cessation may result in an increase of plasma levels of CYP1A2 substrates.Special Populations 8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Available data have not suggested an increased risk for major birth defects following exposure to varenicline in pregnancy, compared with women who smoke [see Data]. Smoking during pregnancy is associated with maternal, fetal, and neonatal risks (see Clinical Considerations). In animal studies, varenicline did not result in major malformations but caused decreased fetal weights in rabbits when dosed during organogenesis at exposures equivalent to 50 times the exposure at the maximum recommended human dose (MRHD). Additionally, administration of varenicline to pregnant rats during organogenesis through lactation produced developmental toxicity in offspring at maternal exposures equivalent to 36 times human exposure at the MRHD [see Data]. The estimated background risk of oral clefts is increased by approximately 30% in infants of women who smoke during pregnancy, compared to pregnant women who do not smoke. The background risk of other major birth defects and miscarriage for the indicated population are unknown. In the US general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
Disease-Associated Maternal and/or Embryo/Fetal Risk
Smoking during pregnancy causes increased risks of orofacial clefts, premature rupture of membranes, placenta previa, placental abruption, ectopic pregnancy, fetal growth restriction and low birth weight, stillbirth, preterm delivery and shortened gestation, neonatal death, sudden infant death syndrome and reduction of lung function in infants. It is not known whether quitting smoking with CHANTIX during pregnancy reduces these risks.
Data
Human Data
A population-based observational cohort study using the national registers of Denmark and Sweden compared pregnancy and birth outcomes among women exposed to varenicline (N=335, includes 317 first trimester exposed) with women who smoked during pregnancy (N=78,412) and with non-smoking pregnant women (N=806,438). The prevalence of major malformations, the primary outcome, was similar in all groups, including between smoking and non-smoking groups. The prevalence of adverse perinatal outcomes in the varenicline-exposed cohort was not greater than in the cohort of women who smoked, and differed somewhat between the three cohorts. The prevalences of the primary and secondary outcomes are shown in Table 6.Pregnant Women
Studies in animals have shown reproductive toxicity (see TOXICOLOGY). The potential risk for humans is not fully known (See ACTION AND CLINICAL PHARMACOLOGY, Special Populations: Pregnant Women). APO-VARENICLINE should not be used during pregnancy.
Pregnant Women
A population-based cohort study compared infants exposed to varenicline in utero (N=335) with infants born to mothers who smoked during pregnancy (N=78,412) and infants born to non-smoking mothers (N=806,438). In this study, infants exposed to varenicline in utero were no more likely to have major congenital malformations (3.6%) than infants born to mothers who smoked during pregnancy (4.3%) or to non-smoking mothers (4.2%). Similarly, infants exposed to varenicline in utero, as compared to infants of smoking and non-smoking mothers, were not at increased risk of stillbirth, (0.3%, 0.5%, 0.3%, respectively), small for gestational age (12.5%,17.1%, 9.1%), preterm birth (7.5%, 7.9%, 5.8%), or premature rupture of membrane (3.6%,5.4%, 3.8%).Table 6. Summary of Primary and Secondary Outcomes for Three Birth Cohorts
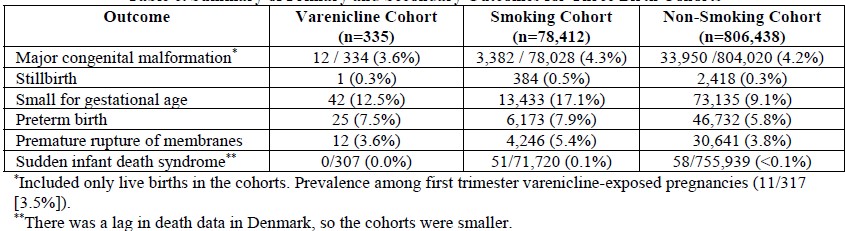
The study limitations include the inability to capture malformations in pregnancies that do not result in a live birth, and possible misclassification of outcome and of exposure to varenicline or to smoking.
Other small epidemiological studies of pregnant women exposed to varenicline did not identify an association with major malformations, consistent with the Danish and Swedish observational cohort study. Methodological limitations of these studies include small samples and lack of adequate controls.
Overall, available studies cannot definitely establish or exclude any varenicline-associated risk during pregnancy.
Animal Data
Pregnant rats and rabbits received varenicline succinate during organogenesis at oral doses up to 15 and 30 mg/kg/day, respectively. While no fetal structural abnormalities occurred in either species, maternal toxicity, characterized by reduced body weight gain, and reduced fetal weights occurred in rabbits at the highest dose (exposures 50 times the human exposure at the MRHD of 1 mg twice daily based on AUC). Fetal weight reduction did not occur in rabbits at exposures 23 times the human exposure at the MRHD based on AUC.
In a pre- and postnatal development study, pregnant rats received up to 15 mg/kg/day of oral varenicline succinate from organogenesis through lactation. Maternal toxicity, characterized by a decrease in body weight gain was observed at 15 mg/kg/day (36 times the human exposure at the MRHD based on AUC). However, decreased fertility and increased auditory startle response occurred in offspring at the highest maternal dose of 15 mg/kg/day8.2 Lactation
Risk Summary
There are no data on the presence of varenicline in human milk, the effects on the breastfed infant, or the effects on milk production. In animal studies varenicline was present in milk of lactating rats [see Data]. However, due to species-specific differences in lactation physiology, animal data may not reliably predict drug levels in human milk. The lack of clinical data during lactation precludes a clear determination of the risk of CHANTIX to an infant during lactation; however the developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CHANTIX and any potential adverse effects on the breastfed child from CHANTIX or from the underlying maternal condition.
Clinical Considerations
Because there are no data on the presence of varenicline in human milk and the effects on the breastfed infant, breastfeeding women should monitor their infant for seizures and excessive vomiting, which are adverse reactions that have occurred in adults that may be clinically relevant in breastfeeding infants.
Data
In a pre- and postnatal development study, pregnant rats received up to 15 mg/kg/day of oral varenicline succinate through gestation and lactation Mean serum concentrations of varenicline in the nursing pups were 5-22% of maternal serum concentrations.Nursing Women
Animal studies have shown that varenicline can be transferred to nursing pups. It is not known whether varenicline is excreted in human milk. Because many drugs are excreted in human milk and because the potential for adverse reactions in nursing infants from APO-VARENICLINE is unknown, a decision should be made whether to discontinue nursing or to discontinue the drug.8.4 Pediatric Use
CHANTIX is not recommended for use in pediatric patients 16 years of age or younger because its efficacy in this population was not demonstrated.
Single and multiple-dose pharmacokinetics of varenicline have been investigated in pediatric patients aged 12 to 17 years old (inclusive) and were approximately dose proportional over the 0.5 mg to 2 mg daily dose range studied. Steady-state systemic exposure in adolescent patients of bodyweight >55 kg, as assessed by AUC (0-24), was comparable to that noted for the same doses in the adult population. When 0.5 mg BID was given, steady-state daily exposure of varenicline was, on average, higher (by approximately 40%) in adolescent patients with bodyweight ≤ 55 kg compared to that noted in the adult population.
The efficacy and safety of varenicline was evaluated in a randomized, double-blind, placebo-controlled study of 312 patients aged 12 to 19 years, who smoked an average of at least 5 cigarettes per day during the 30 days prior to recruitment, had a score of at least 4 on the Fagerstrom Test for Nicotine Dependence scale, and at least one previous failed quit attempt. Patients were stratified by age (12 to 16 years of age, n = 216 and 17 to 19 years of age, n = 96) and by body weight (≤55 kg and >55 kg). Patients were randomized to one of two doses of varenicline, adjusted by weight to provide plasma levels in the efficacious range (based on adult studies) and placebo. Patients received treatment for 12 weeks, followed by a non-treatment period of 40 weeks, along with age-appropriate counseling throughout the study. Results from this study showed that varenicline, at either dose studied, did not improve continuous abstinence rates at weeks 9 through 12 of treatment compared with placebo in subjects 12 to 19 years of age. The varenicline safety profile in this study was consistent with that observed in adult studies.Pediatrics (< 18 years of age)
Based on the data submitted and reviewed by Health Canada, the safety and efficacy of varenicline in pediatric patients has not been established; therefore, Health Canada has not authorized an indication for pediatric use (see WARNINGS AND PRECAUTIONS, Special Populations: Pediatrics).
8.5 Geriatric Use
A combined single- and multiple-dose pharmacokinetic study demonstrated that the pharmacokinetics of 1 mg varenicline given once daily or twice daily to 16 healthy elderly male and female smokers (aged 65-75 years) for 7 consecutive days was similar to that of younger subjects. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Varenicline is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function [see Dosage and Administration (2.2)].
No dosage adjustment is recommended for elderly patients.Geriatrics (> 65 years of age)
A combined single and multiple-dose pharmacokinetic study demonstrated that the pharmacokinetics of 1 mg varenicline given once daily (QD) or BID to 16 healthy elderly male and female smokers (aged 65 to 75 years) for 7 consecutive days was similar to that of younger subjects. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Varenicline is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see DOSAGE AND ADMINISTRATION, Special Populations: Geriatrics).
8.6 Renal Impairment
Varenicline is substantially eliminated by renal glomerular filtration along with active tubular secretion. Dose reduction is not required in patients with mild to moderate renal impairment. For patients with severe renal impairment (estimated creatinine clearance <30 mL/min), and for patients with end-stage renal disease undergoing hemodialysis, dosage adjustment is needed [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
Renal Impairment
A multiple dose pharmacokinetic study was conducted in patients with normal renal function, with mild, moderate, or severe renal impairment (estimated creatinine clearance: > 80 mL/min, > 50 and ≤ 80 mL/min, ≥ 30 and ≤ 50 mL/min, and < 30 mL/min, respectively) or end-stage renal disease (ESRD). Varenicline pharmacokinetics was unchanged in subjects with mild renal impairment. Relative to subjects with normal renal function, varenicline exposure increased 1.5-fold in patients with moderate renal impairment and 2.1-fold in patients with severe renal impairment. In subjects with ESRD, varenicline was efficiently removed by hemodialysis. The recommended dose of varenicline is reduced in patients with severe renal impairment. APO-VARENICLINE is not recommended in patients with ESRD (see ACTION AND CLINICAL PHARMACOLOGY, Special Populations and Conditions: Renal Impairment, and DOSAGE AND ADMINISTRATION, Special Populations: Patients with Impaired Renal Function).Drug Abuse Dependence 9 DRUG ABUSE AND DEPENDENCE
9.1 Controlled Substance
Varenicline is not a controlled substance.9.3 Dependence
Humans
Fewer than 1 out of 1,000 patients reported euphoria in clinical trials with CHANTIX. At higher doses (greater than 2 mg), CHANTIX produced more frequent reports of gastrointestinal disturbances such as nausea and vomiting. There is no evidence of dose-escalation to maintain therapeutic effects in clinical studies, which suggests that tolerance does not develop. Abrupt discontinuation of CHANTIX was associated with an increase in irritability and sleep disturbances in up to 3% of patients. This suggests that, in some patients, varenicline may produce mild physical dependence which is not associated with addiction.
In a human laboratory abuse liability study, a single oral dose of 1 mg varenicline did not produce any significant positive or negative subjective responses in smokers. In non-smokers, 1 mg varenicline produced an increase in some positive subjective effects, but this was accompanied by an increase in negative adverse effects, especially nausea. A single oral dose of 3 mg varenicline uniformly produced unpleasant subjective responses in both smokers and non-smokers.
Animals
Studies in rodents have shown that varenicline produces behavioral responses similar to those produced by nicotine. In rats trained to discriminate nicotine from saline, varenicline produced full generalization to the nicotine cue. In self-administration studies, the degree to which varenicline substitutes for nicotine is dependent upon the requirement of the task. Rats trained to self-administer nicotine under easy conditions continued to self-administer varenicline to a degree comparable to that of nicotine; however in a more demanding task, rats self-administered varenicline to a lesser extent than nicotine. Varenicline pretreatment also reduced nicotine self-administration.Dependence/Tolerance
Human Studies
The rewarding potential of varenicline (1 mg and 3 mg doses) was compared with that of amphetamines in subjects experienced with psychomotor stimulants. The pattern for both smokers and non-smokers was consistent with a profile of a drug that, while having some pharmacological activity, did not produce amphetamine-like subjective effects.
Animal Studies
The subjective nicotine-like effects of varenicline were investigated in drug discrimination studies. At 1 mg/kg, there was complete substitution of varenicline for nicotine in a paradigm of nicotine-associated lever pressing for food reward. In an efficacy model, varenicline pretreatment dose-dependently reduced nicotine self-administration under a fixed-ratio schedule. Under a progressive ratio schedule rats worked harder for nicotine than for varenicline.Overdosage 10 OVERDOSAGE
In case of overdose, standard supportive measures should be instituted as required.
Varenicline has been shown to be dialyzed in patients with end-stage renal disease [see Clinical Pharmacology (12.3)], however, there is no experience in dialysis following overdose.OVERDOSAGE
Symptoms Consistent with its pharmacological profile, varenicline resulted in increased incidences of nausea and vomiting when given at doses greater than the recommended dose of 1 mg BID.
Treatment
Varenicline has been shown to be dialyzed in patients with end-stage renal disease (see ACTION AND CLINICAL PHARMACOLOGY, Special Populations and Conditions: Renal Insufficiency), however, there is no experience with dialysis following overdose.
For management of a suspected drug overdose, contact your regional Poison Control Centre.Description 11 DESCRIPTION
CHANTIX tablets contain varenicline (as the tartrate salt), which is a partial nicotinic agonist selective for α4β2 nicotinic acetylcholine receptor subtypes.
Varenicline, as the tartrate salt, is a powder which is a white to off-white to slightly yellow solid with the following chemical name: 7,8,9,10-tetrahydro-6,10-methano-6H-pyrazino[2,3- h][3]benzazepine, (2R,3R)-2,3-dihydroxybutanedioate (1:1). It is highly soluble in water. Varenicline tartrate has a molecular weight of 361.35Daltons, and a molecular formula of C13H13N3 • C4H6O6. The chemical structure is:
 CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with "Pfizer" on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with "Pfizer" on one side and "CHX 1.0" on the other side. Each 0.5 mg CHANTIX tablet contains 0.85 mg of varenicline tartrate equivalent to 0.5 mg of varenicline free base; each 1 mg CHANTIX tablet contains 1.71 mg of varenicline tartrate equivalent to 1 mg of varenicline free base. The following inactive ingredients are included in the tablets: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, Opadry® White (for 0.5 mg), Opadry® Blue (for 1 mg), and Opadry® Clear.
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with "Pfizer" on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with "Pfizer" on one side and "CHX 1.0" on the other side. Each 0.5 mg CHANTIX tablet contains 0.85 mg of varenicline tartrate equivalent to 0.5 mg of varenicline free base; each 1 mg CHANTIX tablet contains 1.71 mg of varenicline tartrate equivalent to 1 mg of varenicline free base. The following inactive ingredients are included in the tablets: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, Opadry® White (for 0.5 mg), Opadry® Blue (for 1 mg), and Opadry® Clear.
PHARMACEUTICAL INFORMATION
Drug Substance
Proper name: Varenicline Tartrate
Chemical name: 7,8,9,10-Tetrahydro-6,10-methano-6H-pyrazino [2,3-h] [3] benzazepine; (2R,3R)-2,3-dihydroxybutanedioate OR 5,8,14-triazatetracyclo [10.3.1.02,11.04,9]hexadeca-2(11)-3,5,7,9
Molecular formula: C13H13N3.C4H6O6
Molecular weight: 361.35 g/mol
Clinical Pharmacology 12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Varenicline binds with high affinity and selectivity at α4β2 neuronal nicotinic acetylcholine receptors. The efficacy of CHANTIX in smoking cessation is believed to be the result of varenicline’s activity at α4β2 sub-type of the nicotinic receptor where its binding produces agonist activity, while simultaneously preventing nicotine binding to these receptors.
Electrophysiology studies in vitro and neurochemical studies in vivo have shown that varenicline binds to α4β2 neuronal nicotinic acetylcholine receptors and stimulates receptor-mediated activity, but at a significantly lower level than nicotine. Varenicline blocks the ability of nicotine to activate α4β2 receptors and thus to stimulate the central nervous mesolimbic dopamine system, believed to be the neuronal mechanism underlying reinforcement and reward experienced upon smoking. Varenicline is highly selective and binds more potently to α4β2 receptors than to other common nicotinic receptors (>500-fold α3β4, >3,500-fold α7, >20,000-fold α1βγδ), or to non-nicotinic receptors and transporters (>2,000-fold). Varenicline also binds with moderate affinity (Ki = 350 nM) to the 5-HT3 receptor.ACTION AND CLINICAL PHARMACOLOGY
Mechanism of Action
The efficacy of varenicline in smoking-cessation is believed to be a result of varenicline's partial agonist activity at the α4β2 nicotinic acetylcholine receptor (ie, agonist activity to a lesser degree than nicotine), while simultaneously preventing nicotine binding (ie, antagonist activity).In vitro, varenicline binds with higher affinity to the α4β2 receptor subtype than to other common nicotinic receptors (> 500-fold α3β4; > 3,500-fold α7; > 20,000-fold α1βγδ), or to non-nicotinic receptors and transporters (> 2,000-fold).Electrophysiology studies in vitro and neurochemical studies in vivo have shown that varenicline acts as a partial agonist at α4β2 nicotinic acetylcholine receptors. In the absence of nicotine, varenicline’s agonist activity is at a significantly lower level than nicotine, but sufficient to activate the central nervous mesolimbic dopamine system, believed to be the neuronal mechanism underlying reinforcement and reward experienced upon smoking. In the presence of nicotine, which competes for the same human α4β2 nicotinic acetylcholine receptor (nAChR) binding site, varenicline prevented nicotine from activating the α4β2 receptor, since it has higher affinity for this site and this prevented full stimulation of the central nervous mesolimbic dopamine system.
Varenicline is also a partial agonist at α3β4 receptors, but a full agonist at α7 receptors and a full agonist at 5-HT3 receptors.
Varenicline has moderate affinity for the 5-HT3 serotonergic receptor (Ki=350 nM), at which it acts as a weak, full agonist (EC50=0.96 mcM).
Varenicline-induced nausea shortly after dosing, when gastrointestinal levels are predicted to be temporarily high, may be due to activation of this peripheral receptor, in addition to a possible role for peripheral α3β4 and/or central α4β2 nAChRs12.3 Pharmacokinetics
Absorption
Maximum plasma concentrations of varenicline occur typically within 3-4 hours after oral administration. Following administration of multiple oral doses of varenicline, steady-state conditions were reached within 4 days. Over the recommended dosing range, varenicline exhibits linear pharmacokinetics after single or repeated doses.
In a mass balance study, absorption of varenicline was virtually complete after oral administration and systemic availability was ~90%.
Food Effect
Oral bioavailability of varenicline is unaffected by food or time-of-day dosing.
Distribution
Plasma protein binding of varenicline is low (≤20%) and independent of both age and renal function.
Elimination
The elimination half-life of varenicline is approximately 24 hours.
Metabolism
Varenicline undergoes minimal metabolism, with 92% excreted unchanged in the urine.
Excretion
Renal elimination of varenicline is primarily through glomerular filtration along with active tubular secretion possibly via the organic cation transporter, OCT2.
Specific Populations
There are no clinically meaningful differences in varenicline pharmacokinetics due to age, race, gender, smoking status, or use of concomitant medications, as demonstrated in specific pharmacokinetic studies and in population pharmacokinetic analyses.
Age: Geriatric Patients
A combined single- and multiple-dose pharmacokinetic study demonstrated that the pharmacokinetics of 1 mg varenicline given once daily or twice daily to 16 healthy elderly male and female smokers (aged 65-75 years) for 7 consecutive days was similar to that of younger subjects.
Age: Pediatric Patients
CHANTIX is not recommended for use in pediatric patients 16 years of age or younger because its efficacy in this population was not demonstrated [see Use in Specific Populations (8.4)].
Renal Impairment
Varenicline pharmacokinetics were unchanged in subjects with mild renal impairment (estimated creatinine clearance >50 mL/min and ≤80 mL/min). In subjects with moderate renal impairment (estimated creatinine clearance ≥30 mL/min and ≤50 mL/min), varenicline exposure increased 1.5-fold compared with subjects with normal renal function (estimated creatinine clearance >80 mL/min). In subjects with severe renal impairment (estimated creatinine clearance <30 mL/min), varenicline exposure was increased 2.1-fold. In subjects with end-stage-renal disease (ESRD) undergoing a three-hour session of hemodialysis for three days a week, varenicline exposure was increased 2.7-fold following 0.5 mg once daily administration for 12 days. The plasma Cmax and AUC of varenicline noted in this setting were similar to those of healthy subjects receiving 1 mg twice daily [see Dosage and Administration (2.2), Use in Specific Populations (8.6)]. Additionally, in subjects with ESRD, varenicline was efficiently removed by hemodialysis [see Overdosage (10)].
Hepatic Impairment
Due to the absence of significant hepatic metabolism, varenicline pharmacokinetics should be unaffected in patients with hepatic impairment.
Drug-Drug Interactions
In vitro studies demonstrated that varenicline does not inhibit the following cytochrome P450 enzymes (IC50 >6400 ng/mL): 1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A4/5. Also, in human hepatocytes in vitro, varenicline does not induce the cytochrome P450 enzymes 1A2 and 3A4. In vitro studies demonstrated that varenicline does not inhibit human renal transport proteins at therapeutic concentrations. Therefore, drugs that are cleared by renal secretion (e.g., metformin [see below]) are unlikely to be affected by varenicline.
In vitro studies demonstrated the active renal secretion of varenicline is mediated by the human organic cation transporter OCT2. Co-administration with inhibitors of OCT2 (e.g., cimeditine [see below]) may not necessitate a dose adjustment of CHANTIX as the increase in systemic exposure to CHANTIX is not expected to be clinically meaningful. Furthermore, since metabolism of varenicline represents less than 10% of its clearance, drugs known to affect the cytochrome P450 system are unlikely to alter the pharmacokinetics of CHANTIX [see Clinical Pharmacology (12.3)]; therefore, a dose adjustment of CHANTIX would not be required.
Drug interaction studies were performed with varenicline and digoxin, warfarin, transdermal nicotine, bupropion, cimetidine, and metformin. No clinically meaningful pharmacokinetic drug-drug interactions have been identified.
Metformin
When co-administered to 30 smokers, varenicline (1 mg twice daily) did not alter the steady-state pharmacokinetics of metformin (500 mg twice daily), which is a substrate of OCT2. Metformin had no effect on varenicline steady-state pharmacokinetics.
Cimetidine
Co-administration of an OCT2 inhibitor, cimetidine (300 mg four times daily), with varenicline (2 mg single dose) to 12 smokers increased the systemic exposure of varenicline by 29% (90% CI: 21.5%, 36.9%) due to a reduction in varenicline renal clearance.
Digoxin *Varenicline (1 mg twice daily) did not alter the steady-state pharmacokinetics of digoxin administered as a 0.25 mg daily dose in 18 smokers.
Warfarin
Varenicline (1 mg twice daily) did not alter the pharmacokinetics of a single 25 mg dose of (R, S)-warfarin in 24 smokers. Prothrombin time (INR) was not affected by varenicline. Smoking cessation itself may result in changes to warfarin pharmacokinetics [see Drug Interactions (7.2)].
Use with Other Drugs for Smoking Cessation
Bupropion: Varenicline (1 mg twice daily) did not alter the steady-state pharmacokinetics of bupropion (150 mg twice daily) in 46 smokers [see Drug Interactions (7.1)].
NRT: Although co-administration of varenicline (1 mg twice daily) and transdermal nicotine (21 mg/day) for up to 12 days did not affect nicotine pharmacokinetics, the incidence of adverse reactions was greater for the combination than for NRT alone [see Drug Interactions (7.1)].
Pharmacokinetics
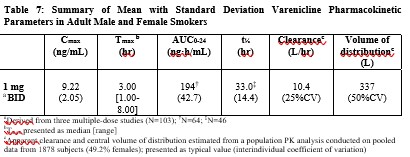
Absorption: Maximum plasma concentrations of varenicline occur typically within 3 to 4 hours after oral administration. Following administration of multiple oral doses of varenicline to healthy volunteers, steady-state conditions were reached within 4 days. Varenicline exhibits linear kinetics when given as single (0.1 to 3 mg) or repeated (1 to 3 mg/day) doses. In a mass balance study, absorption of varenicline is virtually complete after oral administration and systemic availability is high. Oral bioavailability of varenicline is unaffected by food or time-of-day dosing.
Distribution: Plasma protein binding of varenicline is low (≤ 20%) and independent of both age and renal function.
Metabolism: Varenicline tartrate undergoes minimal metabolism, with approximately 92% of recovered drug-related entity in urine being unchanged varenicline. Metabolite profiles (for circulation and urine) were similar for smokers and non-smokers, and are from the following minor routes of metabolism: N-carbomyl glucuronidation, N-formylation and conjugation with a hexose sugar.
Elimination: The elimination half-life of varenicline tartrate is approximately 24 hours. Renal elimination of varenicline is the major elimination route, primarily through glomerular filtration along with active tubular secretion via the organic cationic transporter, OCT2.
Special Populations and Conditions
There were no clinically meaningful differences seen in varenicline tartrate pharmacokinetics due to being elderly, race, gender, smoking status, or use of concomitant medications, as demonstrated in specific pharmacokinetic studies and in population pharmacokinetic analyses.
Pediatrics: Based on the data submitted and reviewed by Health Canada, the safety and efficacy of varenicline in pediatric patients has not been established; therefore, Health Canada has not authorized an indication for pediatric use.
Two pharmacokinetic studies have been conducted in adolescent smokers, aged 12 to 17 inclusive: a single dose study (n = 27), and a multiple dose study (n = 72). Pharmacokinetics were approximately dose-proportional over the 0.5 mg to 2 mg daily dose range studied. (see INDICATIONS AND CLINICAL USE, Special population: Pediatrics).
Steady-state systemic exposure: In the multiple-dose study, patients were stratified by bodyweight (> 55 kg; ≤ 55 kg), and within each bodyweight group, were randomized into three treatment arms (low dose of varenicline, high dose of varenicline and placebo) using a 2:2:1 randomization scheme. Dosing was as follows:- > 55 kg: 0.5 mg BID (n = 14), 1 mg BID (n = 14) and placebo (n = 7);
- ≤ 55 kg 0.5 mg QD (n = 15), 0.5 mg BID (n = 14) and placebo (n = 8).
The dosing period was 14 days, with all arms at target dose by Day 8. Patients were allowed to continue smoking at will throughout the study.
In adolescent patients of bodyweight > 55 kg, steady-state systemic exposures, as assessed by AUC (0 to 24), were consistent with those previously observed in the adult population. In adolescent patients of ≤ 55 kg, steady-state systemic exposure for the 0.5 mg BID was on average approximately 40% higher compared to that previously observed in the adult population.
Individual adverse event terms (MedDRA-coded preferred terms) that were reported in more than one patient taking varenicline and more frequently than for placebo were: nausea (most frequent), headache, vomiting, dizziness, pharyngolaryngeal pain, abdominal pain upper, anorexia, flatulence, abnormal dreams, arthralgia, fatigue, and somnolence. Patients ≤ 55 kg reported more adverse events than patients > 55 kg. Mood-related events were reported for three patients of 57 in the varenicline arms (anger, mood swings, irritability; none severe), compared with 0 reports in 15 patients in the placebo arms.
Geriatrics: A combined single and multiple-dose pharmacokinetic study demonstrated that the pharmacokinetics of 1 mg varenicline given once or twice daily to 16 healthy elderly male and female smokers (aged 65 to 75 years) for 7 consecutive days was similar to that of younger subjects.
Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see DOSAGE AND ADMINISTRATION, Special Populations: Dosing in Elderly Patients).
Hepatic Insufficiency: Due to the absence of significant hepatic metabolism, varenicline tartrate pharmacokinetics should be unaffected in patients with hepatic insufficiency, except in the case that there is accompanying renal compromise (see DOSAGE AND ADMINISTRATION). The potential for clinically meaningful drug interactions between varenicline and metabolic inhibitors/inducers is low.
Renal Impairment: Varenicline tartrate pharmacokinetics were studied in subjects with normal, mild, moderate, severe renal impairment and end-stage renal disease (n=6 per arm), following 0.5 mg once daily administration for 12 days.
Varenicline pharmacokinetics were essentially unchanged in subjects with mild renal impairment (estimated creatinine clearance > 50 mL/min and ≤ 80 mL/min). In patients with moderate renal impairment (estimated creatinine clearance ≥ 30 mL/min and ≤ 50 mL/min), varenicline exposure [AUCτ] increased 1.5-fold compared with subjects with normal renal function (estimated creatinine clearance > 80 mL/min).
In subjects with severe renal impairment (estimated creatinine clearance < 30 mL/min), varenicline exposure [AUCτ] was increased 2.1-fold.
In subjects with end-stage renal disease (ESRD), undergoing a three-hour session of hemodialysis for three days a week, varenicline exposure [AUCτ] was increased 2.7-fold; varenicline was efficiently removed by hemodialysis (see DOSAGE AND ADMINISTRATION, Recommended Dose and Dosage Adjustment: Special Populations, Patients with Impaired Renal Function).
Drug-drug interaction studies were limited to approximately two-week studies in healthy young adult volunteers who smoked.
Cimetidine: Co-administration of varenicline (2 mg single dose) with an hOCT2 inhibitor, cimetidine (300 mg four times daily (QID) at steady-state) to 12 smokers increased the systemic exposure of varenicline by 29% (90% CI: 21.5%, 36.9%) due to a reduction in varenicline renal clearance. No dosage adjustment is recommended based on concomitant cimetidine administration in subjects with normal renal function or in patients with mild to moderate renal impairment. In patients with severe renal impairment, the concomitant use of cimetidine and varenicline should be avoided (see DOSAGE AND ADMINISTRATION, Recommended Dose and Dosage Adjustment: Special Populations, Patients with Impaired Renal Function).
Other inhibitors of hOCT2: Other inhibitors of hOCT2 have not been directly studied. Cimetidine causes greater in vivo drug interactions with renally cleared compounds than other inhibitors of hOCT2. Consequently, co-administration of other inhibitors of hOCT2 with varenicline would not require dosage adjustment in patients with normal renal function or moderate renal impairment. In patients with severe renal impairment, the concomitant use of varenicline and other inhibitors of hOCT2, such as trimethoprim, ranitidine or levofloxacin should be avoided (see DOSAGE AND ADMINISTRATION, Recommended Dose and Dosage Adjustment: Special Populations, Patients with Impaired Renal Function).
Co-administration with Other Drugs Eliminated via hOCT2: Based on the lack of interaction between varenicline and metformin, interactions between varenicline and other cationic drugs eliminated via hOCT2 are unlikely.
Warfarin: Varenicline (1 mg BID steady-state) did not alter the pharmacokinetics of a single 25 mg dose of (R, S)-warfarin in 24 smokers. Prothrombin time (INR) was not affected by varenicline. Smoking-cessation itself may result in changes to warfarin pharmacokinetics (see WARNINGS AND PRECAUTIONS).
Multiple dosing for both drugs:
Metformin: When co-administered to 30 smokers, varenicline (1 mg BID) did not alter the steady-state pharmacokinetics of metformin (500 mg BID), which is a substrate of hOCT2. Metformin had no effect on varenicline steady-state pharmacokinetics.
Digoxin: Varenicline (1 mg BID) did not alter the steady-state pharmacokinetics of digoxin administered as a 0.25 mg daily dose in 18 smokers. Steady-state pharmacokinetics of varenicline remained unchanged by digoxin co-administration.
Nonclinical Toxicology 13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Lifetime carcinogenicity studies were performed in CD-1 mice and Sprague-Dawley rats. There was no evidence of a carcinogenic effect in mice administered varenicline by oral gavage for 2 years at doses up to 20 mg/kg/day (47 times the maximum recommended human daily (MRHD) exposure based on AUC). Rats were administered varenicline (1, 5, and 15 mg/kg/day) by oral gavage for 2 years. In male rats (n = 65 per sex per dose group), incidences of hibernoma (tumor of the brown fat) were increased at the mid dose (1 tumor, 5 mg/kg/day, 23 times the MRHD exposure based on AUC) and maximum dose (2 tumors, 15 mg/kg/day, 67 times the MRHD exposure based on AUC). The clinical relevance of this finding to humans has not been established. There was no evidence of carcinogenicity in female rats.
Mutagenesis
Varenicline was not genotoxic, with or without metabolic activation, in the following assays: Ames bacterial mutation assay; mammalian CHO/HGPRT assay; and tests for cytogenetic aberrations in vivo in rat bone marrow and in vitro in human lymphocytes.
Impairment of Fertility
There was no evidence of impairment of fertility in either male or female Sprague-Dawley rats administered varenicline succinate up to 15 mg/kg/day (67 and 36 times, respectively, the MRHD exposure based on AUC at 1 mg twice daily). Maternal toxicity, characterized by a decrease in body weight gain, was observed at 15 mg/kg/day. However, a decrease in fertility was noted in the offspring of pregnant rats who were administered varenicline succinate at an oral dose of 15 mg/kg/day. This decrease in fertility in the offspring of treated female rats was not evident at an oral dose of 3 mg/kg/day (9 times the MRHD exposure based on AUC at 1 mg twice daily).TOXICOLOGY
Carcinogenesis: Lifetime carcinogenicity studies were performed in CD-1 mice and Sprague-Dawley rats. There was no evidence of a carcinogenic effect in mice administered varenicline by oral gavage for 2 years at doses up to 20 mg/kg/day (47 times the maximum recommended human daily exposure based on the area under the curve (AUC). Rats were administered varenicline (1, 5, and 15 mg/kg/day) by oral gavage for 2 years. In male rats (n=65 per sex per dose group), incidences of hibernoma (tumor of the brown fat) was increased at the mid dose (1 tumor, 5 mg/kg/day, 23 times the maximum recommended human daily exposure based on AUC) and at the maximum dose (2 tumors, 15 mg/kg/day, 67 times the maximum recommended human daily exposure based on AUC). The clinical relevance of this finding to humans has not been established. There was no evidence of carcinogenicity in female rats.
Mutagenesis: Varenicline was not genotoxic, with or without metabolic activation, in the following assays: Ames bacterial mutation assay; mammalian CHO/HGPRT assay; and tests for cytogenetic aberrations in vivo in rat bone marrow and in vitro in human lymphocytes.
Sexual Function / Reproduction
Impairment of Fertility: There was no evidence of impairment of fertility in either male or female Sprague-Dawley rats administered varenicline succinate up to 15 mg/kg/day (67 and 36 times, respectively, the maximum recommended human daily exposure based on AUC at 1 mg BID). However, a decrease in fertility was noted in the offspring of pregnant rats who were administered varenicline succinate at an oral dose of 15 mg/kg/day (36 times the maximum recommended human daily exposure based on AUC at 1 mg BID). This decrease in fertility in the offspring of treated female rats was not evident at an oral dose of 3 mg/kg/day (9 times the maximum recommended human daily exposure based on AUC at 1 mg BID).Clinical Studies 14 CLINICAL STUDIES
The efficacy of CHANTIX in smoking cessation was demonstrated in six clinical trials in which a total of 3659 chronic cigarette smokers (≥10 cigarettes per day) were treated with CHANTIX. In all clinical studies, abstinence from smoking was determined by patient self-report and verified by measurement of exhaled carbon monoxide (CO≤10 ppm) at weekly visits. Among the CHANTIX-treated patients enrolled in these studies, the completion rate was 65%. Except for the dose-ranging study (Study 1) and the maintenance of abstinence study (Study 6), patients were treated for 12 weeks and then were followed for 40 weeks post-treatment. Most patients enrolled in these trials were white (79-96%). All studies enrolled almost equal numbers of men and women. The average age of patients in these studies was 43 years. Patients on average had smoked about 21 cigarettes per day for an average of approximately 25 years. Patients set a date to stop smoking (target quit date) with dosing starting 1 week before this date.
Seven additional studies evaluated the efficacy of CHANTIX in patients with cardiovascular disease, in patients with chronic obstructive pulmonary disease [see Clinical Studies (14.7)], in patients instructed to select their quit date within days 8 and 35 of treatment [see Clinical Studies (14.4)], patients with major depressive disorder [see Clinical Studies (14.9)], patients who had made a previous attempt to quit smoking with CHANTIX, and either did not succeed in quitting or relapsed after treatment [see Clinical Studies (14.6)], in patients without or with a history of psychiatric disorder enrolled in a postmarketing neuropsychiatric safety outcome trial [see Warnings and Precautions (5.1), Clinical Studies (14.10)], and in patients who were not able or willing to quit abruptly and were instructed to quit gradually [see Clinical studies (14.5)].
In all studies, patients were provided with an educational booklet on smoking cessation and received up to 10 minutes of smoking cessation counseling at each weekly treatment visit according to Agency for Healthcare Research and Quality guidelines.CLINICAL TRIALS
Comparative Bioavailability Study
A randomized, single dose, 2-way crossover comparative bioavailability study, conducted under fasting conditions on healthy male volunteers (N=23). The rate and extent of absorption of varenicline was measured and compared following a single oral dose (1 x 1 mg tablet) of APO-VARENICLINE and CHAMPIX®/MD (Varenicline). The results are summarized in the following table:
Summary table of the comparative bioavailability data for APO-VARENICLINE (fasting conditions)
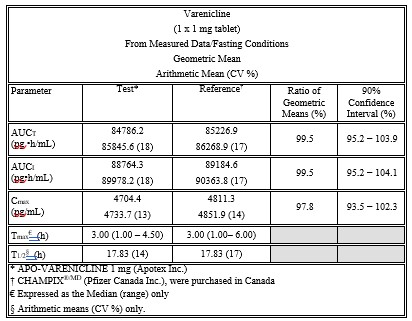
14.1 Initiation of Abstinence
Study 1
This was a six-week dose-ranging study comparing CHANTIX to placebo. This study provided initial evidence that CHANTIX at a total dose of 1 mg per day or 2 mg per day was effective as an aid to smoking cessation.
Study 2
This study of 627 patients compared CHANTIX 1 mg per day and 2 mg per day with placebo. Patients were treated for 12 weeks (including one-week titration) and then were followed for 40 weeks post-treatment. CHANTIX was given in two divided doses daily. Each dose of CHANTIX was given in two different regimens, with and without initial dose-titration, to explore the effect of different dosing regimens on tolerability. For the titrated groups, dosage was titrated up over the course of one week, with full dosage achieved starting with the second week of dosing. The titrated and nontitrated groups were pooled for efficacy analysis.
Forty-five percent of patients receiving CHANTIX 1 mg per day (0.5 mg twice daily) and 51% of patients receiving 2 mg per day (1 mg twice daily) had CO-confirmed continuous abstinence during weeks 9 through 12 compared to 12% of patients in the placebo group (Figure 1). In addition, 31% of the 1 mg per day group and 31% of the 2 mg per day group were continuously abstinent from one week after TQD through the end of treatment as compared to 8% of the placebo group.
Study 3
This flexible-dosing study of 312 patients examined the effect of a patient-directed dosing strategy of CHANTIX or placebo. After an initial one-week titration to a dose of 0.5 mg twice daily, patients could adjust their dosage as often as they wished between 0.5 mg once daily to 1 mg twice daily per day. Sixty-nine percent of patients titrated to the maximum allowable dose at any time during the study. For 44% of patients, the modal dose selected was 1 mg twice daily; for slightly over half of the study participants, the modal dose selected was 1 mg/day or less.
Of the patients treated with CHANTIX, 40% had CO-confirmed continuous abstinence during weeks 9 through 12 compared to 12% in the placebo group. In addition, 29% of the CHANTIX group were continuously abstinent from one week after TQD through the end of treatment as compared to 9% of the placebo group.
Study 4 and Study 5
These identical double-blind studies compared CHANTIX 2 mg per day, bupropion sustained-release (SR) 150 mg twice daily, and placebo. Patients were treated for 12 weeks and then were followed for 40 weeks post-treatment. The CHANTIX dosage of 1 mg twice daily was achieved using a titration of 0.5 mg once daily for the initial 3 days followed by 0.5 mg twice daily for the next 4 days. The bupropion SR dosage of 150 mg twice daily was achieved using a 3-day titration of 150 mg once daily. Study 4 enrolled 1022 patients and Study 5 enrolled 1023 patients. Patients inappropriate for bupropion treatment or patients who had previously used bupropion were excluded.
In Study 4, patients treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (44%) compared to patients treated with bupropion SR (30%) or placebo (17%). The bupropion SR quit rate was also superior to placebo. In addition, 29% of the CHANTIX group were continuously abstinent from one week after TQD through the end of treatment as compared to 12% of the placebo group and 23% of the bupropion SR group.
Similarly in Study 5, patients treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (44%) compared to patients treated with bupropion SR (30%) or placebo (18%). The bupropion SR quit rate was also superior to placebo. In addition, 29% of the CHANTIX group were continuously abstinent from one week after TQD through the end of treatment as compared to 11% of the placebo group and 21% of the bupropion SR group.Other Clinical Studies
The efficacy of varenicline (varenicline tartrate) in smoking-cessation was demonstrated in five double-blind, placebo-controlled clinical trials in which a total of 4190 chronic cigarette smokers (about 10 cigarettes per day) received varenicline. Patients set a date to stop smoking (target quit date, or TQD) of 1 week after treatment initiation. For four of the studies, the primary outcome was based on 12 weeks of drug treatment, with a subsequent 40 weeks of double-blind assessment, post drug-treatment. Of these four, two included a bupropion SR arm. The fifth study assessed the effect of 12 weeks of double-blind treatment on maintenance of abstinence achieved during a prior 12 weeks of open-label varenicline.
The four smoking cessation studies with 12 weeks treatment:
Primary objective: A comparison of varenicline to placebo, and additionally in each of the two studies with a bupropion SR arm comparison of varenicline (1 mg BID) to buproprion SR.
Primary endpoint: Abstinence Responder rate was defined as % of patients for whom 4-week continuous abstinence from Week 9 through Week 12 (4 Week-Continuous Quit Rate, or 4W-CQR) was recorded. Abstinence from smoking was determined on a weekly basis, by patient self-report and measurement of expired carbon monoxide levels (CO). Abstinence was defined as self-report of not even a puff of a cigarette, and by having CO measurements of ≤ 10 ppm. Intent-to-treat population was used, and patients who discontinued drug treatment early were eligible as responders, provided they chose to remain in the study.
Key secondary endpoint: Continuous Abstinence Rate (CAR) was defined as the proportion of all patients who reported that they did not smoke (not even a puff of a cigarette) from Week 9 through to Week 52 (i.e., including the 40-week, non-drug treatment period), and had an exhaled CO measurement of ≤ 10 ppm.
Study 1; 12-week randomized dose comparison:
This study compared varenicline 0.5 mg BID (n=253) and 1 mg BID (n=253) with placebo (n=121). Each treatment arm had two different regimens - with or without a week of dose titration – in order to explore the effect on tolerability. The titrated and non-titrated groups were pooled for efficacy analysis.
Study 2; 12-week flexible dose study:
This study (n=312) examined the effect of patient-directed dosing strategy of varenicline or placebo. After an initial one week titration to a dose 0.5 mg BID, patients could adjust their dosage as often as they wished between 0.5 mg QD to 1 mg BID. Sixty-nine percent (69%) of patients titrated to the maximum allowable dose at any time during the study. For 44% of patients, the modal dose selected was 1 mg BID; for 52% of the study patients, the modal dose selected was 1 mg/day or less.
Study 3 and Study 4; Identical 12-week studies with active comparator arm:
Two identical double-blinded clinical trials prospectively compared the efficacy of varenicline (1 mg BID) to placebo, and to sustained release bupropion (150 mg BID) in the absence of NRT in smoking-cessation. Patients received treatment for 12 weeks and then were followed for a total study duration of 52 weeks. The varenicline dosage of 1 mg BID was achieved using a titration of 0.5 mg once daily for the initial 3 days followed by 0.5 mg BID for the next 4 days. The bupropion dosage of 150 mg BID was achieved using a 3-day titration of 150 mg once daily.
Study Results
Primary Endpoint
In all four studies, the primary endpoint for varenicline (i.e., 4W-CQR from Week 9 to Week 12) demonstrated statistical superiority to placebo and in the subset of the two identical studies, statistical superiority to bupropion SR was also demonstrated with varenicline 1 mg BID dose. No patients were allowed to use NRT during the drug treatment phase, and those who did were considered treatment failures. The 4W-CQR (Weeks 9 to 12) for all four studies are shown in Table 11.Figure 1: Continuous Abstinence, Weeks 9 through 12
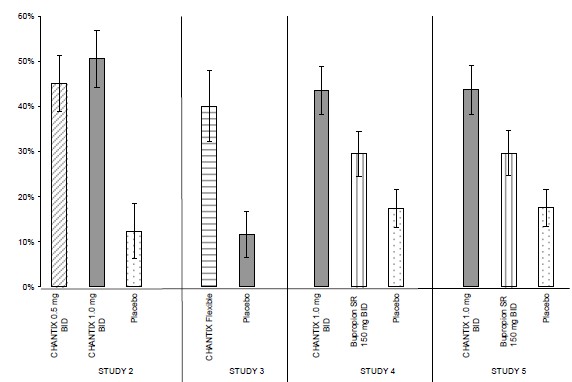
Table 7. Continuous Abstinence, Weeks 9 through 12 (95% confidence interval)
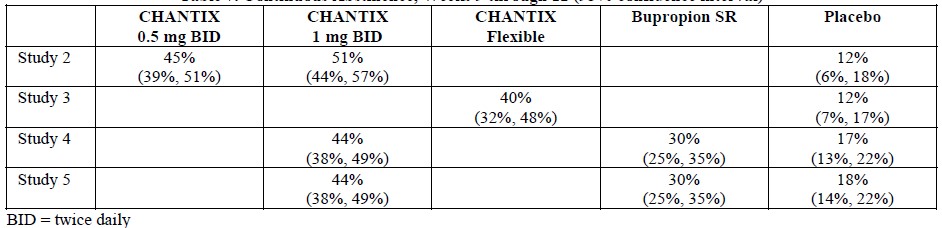
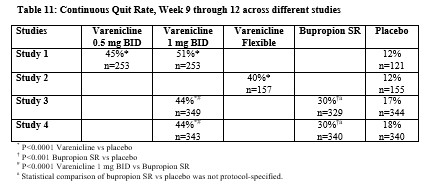
14.2 Urge to Smoke
Based on responses to the Brief Questionnaire of Smoking Urges and the Minnesota Nicotine Withdrawal scale “urge to smoke” item, CHANTIX reduced urge to smoke compared to placebo.Urge to Smoke and Withdrawal SymptomsBased on the responses to the Brief Questionnaire of Smoking Urges and the Minnesota Nicotine Withdrawal Scale, as measured in the 12-week treatment period, craving and urge to smoke were significantly reduced in patients randomized to varenicline compared to those randomized to placebo, as were negative affect withdrawal symptoms (depressed mood; irritability, frustration, or anger; anxiety; difficulty concentrating). 14.3 Long-Term Abstinence
Studies 1 through 5 included 40 weeks of post-treatment follow-up. In each study, CHANTIX-treated patients were more likely to maintain abstinence throughout the follow-up period than were patients treated with placebo (Figure 2, Table 8).Secondary Endpoints:
In all four studies, a key secondary endpoint for varenicline (i.e., CAR Week 9 through 52) demonstrated statistical superiority to placebo. The CAR Weeks 9 through 52 for all four studies are shown in Table 12.
Maintenance of Abstinence StudyThe fifth study assessed the benefit of an additional 12 weeks of varenicline therapy on the maintenance of abstinence. Patients received open-label varenicline 1 mg BID for 12 weeks. Patients who were abstinent for 7 continuous days at Week 12 were then randomized to double-blind treatment with either varenicline (1 mg BID, n=602) or placebo (n=604) for an additional 12 weeks, and then followed for a total study duration of 52 weeks.
The primary study endpoint was the CO-confirmed CAR (defined as above) from Week 13 through Week 24 in the double-blind treatment phase. A key secondary endpoint was the CAR for Week 13 through Week 52.
Superiority to placebo was shown for both the primary and secondary endpoints (see Table 9).
The CAR from Week 13 through Week 24 was higher for patients continuing treatment with varenicline (70.6%) than for patients switching to placebo (49.8%). Superiority to placebo was also maintained during the 28-week, post-treatment follow-up (varenicline 44.0% versus placebo 37.1% at Week 52). This study showed the benefit of an additional 12 weeks of treatment with varenicline 1 mg BID for the maintenance of smoking-cessation, compared to placebo. A statistically significant difference was maintained at Week 52, the final week of the study.
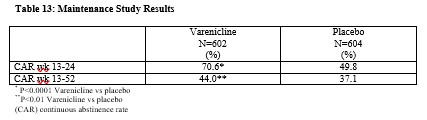
Figure 2: Continuous Abstinence, Weeks 9 through 52
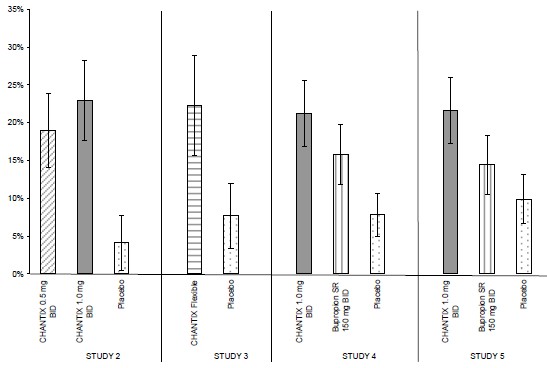
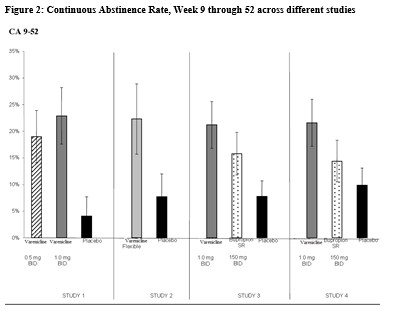
Table 8. Continuous Abstinence, Weeks 9 through 52 (95% confidence interval) Across Different Studies
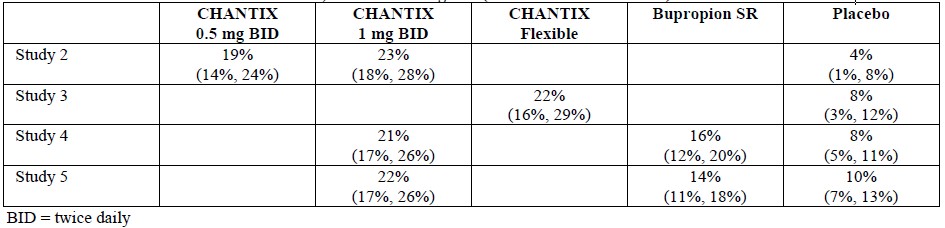
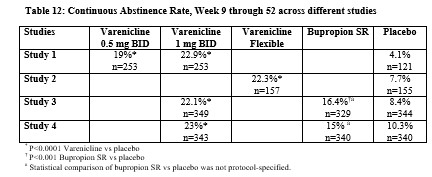
Study 6
This study assessed the effect of an additional 12 weeks of CHANTIX therapy on the likelihood of long-term abstinence. Patients in this study (N=1927) were treated with open-label CHANTIX 1 mg twice daily for 12 weeks. Patients who had stopped smoking for at least a week by Week 12 (N= 1210) were then randomized to double-blind treatment with CHANTIX (1 mg twice daily) or placebo for an additional 12 weeks and then followed for 28 weeks post-treatment.
The continuous abstinence rate from Week 13 through Week 24 was higher for patients continuing treatment with CHANTIX (70%) than for patients switching to placebo (50%). Superiority to placebo was also maintained during 28 weeks post-treatment follow-up (CHANTIX 54% versus placebo 39%).
In Figure 3 below, the x-axis represents the study week for each observation, allowing a comparison of groups at similar times after discontinuation of CHANTIX; post-CHANTIX follow-up begins at Week 13 for the placebo group and Week 25 for the CHANTIX group. The y-axis represents the percentage of patients who had been abstinent for the last week of CHANTIX treatment and remained abstinent at the given timepoint.Figure 3: Continuous Abstinence Rate during Nontreatment Follow-Up
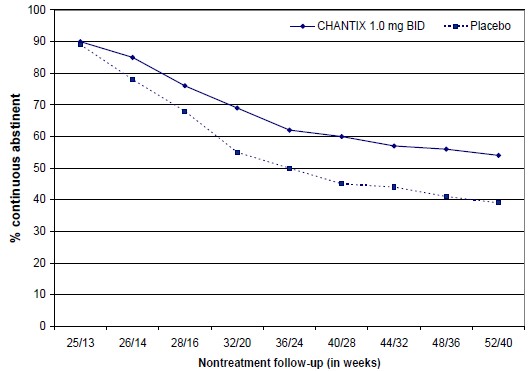
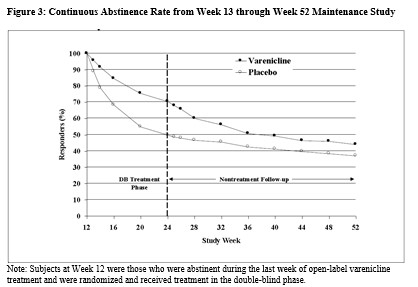
14.4 Alternative Instructions for Setting a Quit Date
CHANTIX was evaluated in a double-blind, placebo-controlled trial where patients were instructed to select a target quit date between Day 8 and Day 35 of treatment. Subjects were randomized 3:1 to CHANTIX 1 mg twice daily (N=486) or placebo (N=165) for 12 weeks of treatment and followed for another 12 weeks posttreatment. Patients treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (54%) compared to patients treated with placebo (19%) and from weeks 9 through 24 (35%) compared to subjects treated with placebo (13%).14.5 Gradual Approach to Quitting Smoking
CHANTIX was evaluated in a 52-week double-blind placebo-controlled study of 1,510 subjects who were not able or willing to quit smoking within four weeks, but were willing to gradually reduce their smoking over a 12 week period before quitting. Subjects were randomized to either CHANTIX 1 mg twice daily (N=760) or placebo (N=750) for 24 weeks and followed up post-treatment through week 52. Subjects were instructed to reduce the number of cigarettes smoked by at least 50 percent by the end of the first four weeks of treatment, followed by a further 50 percent reduction from week four to week eight of treatment, with the goal of reaching complete abstinence by 12 weeks. After the initial 12-week reduction phase, subjects continued treatment for another 12 weeks. Subjects treated with CHANTIX had a significantly higher Continuous Abstinence Rate compared with placebo at weeks 15 through 24 (32% vs. 7%) and weeks 15 through 52 (24% vs. 6%).14.6 Re-Treatment Study
CHANTIX was evaluated in a double-blind, placebo-controlled trial of patients who had made a previous attempt to quit smoking with CHANTIX, and either did not succeed in quitting or relapsed after treatment. Subjects were randomized 1:1 to CHANTIX 1 mg twice daily (N=249) or placebo (N=245) for 12 weeks of treatment and followed for 40 weeks post-treatment. Patients included in this study had taken CHANTIX for a smoking-cessation attempt in the past (for a total treatment duration of a minimum of two weeks), at least three months prior to study entry, and had been smoking for at least four weeks. Patients treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (45%) compared to patients treated with placebo (12%) and from weeks 9 through 52 (20%) compared to subjects treated with placebo (3%).Patients Re-treated with Varenicline
Varenicline was evaluated in a double-blind, placebo-controlled trial of 494 patients who had made a previous attempt to quit smoking with varenicline, and either did not succeed in quitting or relapsed after treatment. Subjects were randomized 1:1 to varenicline 1 mg BID (n=249) or placebo (n=245) for 12 weeks of treatment and followed for up to 40 weeks post-treatment. Patients included in this study had taken varenicline for a smoking-cessation attempt in the past (for a total treatment duration of a minimum of two weeks), at least three months prior to study entry, and had been smoking for at least four weeks. Quit rates in this study were in the range of those from studies in subjects at their first attempt to quit smoking with varenicline. Adverse events in this one-year study were quantitatively and qualitatively similar to those from studies in subjects at their first attempt to quit with varenicline
Table 9. Continuous Abstinence (95% confidence interval), Re-Treatment Study
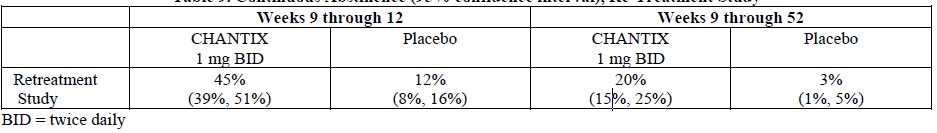
14.7 Subjects with Chronic Obstructive Pulmonary Disease
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged ≥ 35 years with mild-to-moderate COPD with post-bronchodilator FEV1/FVC <70% and FEV1 ≥ 50% of predicted normal value. Subjects were randomized to CHANTIX 1 mg twice daily (N=223) or placebo (N=237) for a treatment of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (41%) compared to subjects treated with placebo (9%) and from week 9 through 52 (19%) compared to subjects treated with placebo (6%).Patients with Chronic Obstructive Pulmonary Disease
Varenicline was evaluated in a randomized, double-blind, placebo-controlled smoking cessation study of 499 subjects with mild-to-moderate COPD with post-bronchodilator FEV1/FVC < 70% and FEV1 ≥ 50% of predicted normal value, aged > 35 years. Subjects were randomized and treated with varenicline 1 mg BID (n=248) or placebo (n=251) for 12 weeks and then followed for 40 weeks post-treatment. Quit rates were in the range of those from studies in the general population of smokers. Adverse events in this one-year study were quantitatively and qualitatively similar to those observed in studies in the general population of smokers.
Table 10. Continuous Abstinence (95% confidence interval), Studies in Patients with Chronic Obstructive Pulmonary Disease (COPD)
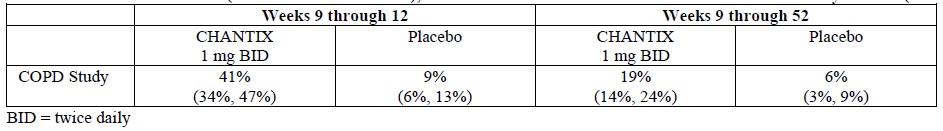
14.8 Subjects with Cardiovascular Disease and Other Cardiovascular Analyses
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 35 to 75 years with stable, documented cardiovascular disease (diagnoses other than, or in addition to, hypertension) that had been diagnosed for more than 2 months. Subjects were randomized to CHANTIX 1 mg twice daily (N=353) or placebo (N=350) for a treatment period of 12 weeks and then were followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (47%) compared to subjects treated with placebo (14%) and from week 9 through 52 (20%) compared to subjects treated with placebo (7%).Patients with Cardiovascular Disease
Varenicline was evaluated in a randomized, double-blind, placebo-controlled smoking cessation study of subjects aged 35 to 75 years with stable, documented cardiovascular disease (other than or in addition to hypertension) that had been diagnosed for > 2 months. Subjects were randomized to varenicline 1 mg BID (n=353) or placebo (n=350) for 12 weeks of treatment and then were followed for 40 weeks post-treatment. Quit rates were in the range of those from studies in the general population of smokers. Adverse events in this study were quantitatively and qualitatively similar to those observed in studies in the general population of smokers, other than cardiovascular-related events (see also WARNINGS AND PRECAUTIONS, Cardiovascular Events).Table 11. Continuous Abstinence (95% confidence interval), Studies in Patients with Cardiovascular Disease (CVD)
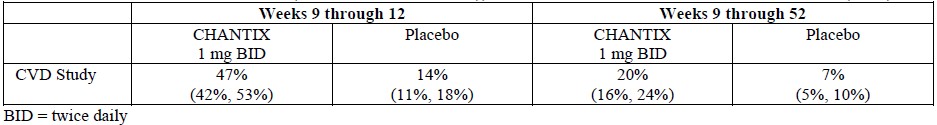
In this study, all-cause and CV mortality was lower in patients treated with CHANTIX, but certain nonfatal CV events occurred more frequently in patients treated with CHANTIX than in patients treated with placebo [see Warnings and Precautions (5.5), Adverse Reactions (6.1)]. Table 12 below shows mortality and the incidence of selected nonfatal serious CV events occurring more frequently in the CHANTIX arm compared to the placebo arm. These events were adjudicated by an independent blinded committee. Nonfatal serious CV events not listed occurred at the same incidence or more commonly in the placebo arm. Patients with more than one CV event of the same type are counted only once per row. Some of the patients requiring coronary revascularization underwent the procedure as part of management of nonfatal MI and hospitalization for angina. Table 12. Mortality and Adjudicated Nonfatal Serious Cardiovascular Events in the Placebo-Controlled CHANTIX Trial in Patients with Stable Cardiovascular Disease
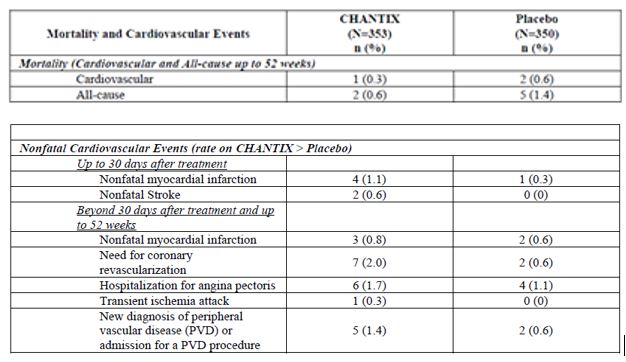
Following the CVD study, a meta-analysis of 15 clinical trials of ≥12 weeks treatment duration, including 7002 patients (4190 CHANTIX, 2812 placebo), was conducted to systematically assess the CV safety of CHANTIX. The study in patients with stable CV disease described above was included in the meta-analysis. There were lower rates of all-cause mortality (CHANTIX 6 [0.14%]; placebo 7 [0.25%]) and CV mortality (CHANTIX 2 [0.05%]; placebo 2 [0.07%]) in the CHANTIX arms compared with the placebo arms in the meta-analysis.
The key CV safety analysis included occurrence and timing of a composite endpoint of Major Adverse Cardiovascular Events (MACE), defined as CV death, nonfatal MI, and nonfatal stroke. These events included in the endpoint were adjudicated by a blinded, independent committee. Overall, a small number of MACE occurred in the trials included in the meta-analysis, as described in Table 13. These events occurred primarily in patients with known CV disease.Table 13. Number of MACE cases, Hazard Ratio and Rate Difference in a Meta-Analysis of 15 Clinical Trials Comparing CHANTIX to Placebo*
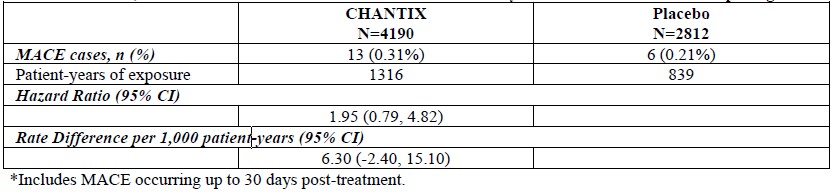
The meta-analysis showed that exposure to CHANTIX resulted in a hazard ratio for MACE of 1.95 (95% confidence interval from 0.79 to 4.82) for patients up to 30 days after treatment; this is equivalent to an estimated increase of 6.3 MACE events per 1,000 patient-years of exposure. The meta-analysis showed higher rates of CV endpoints in patients on CHANTIX relative to placebo across different time frames and pre-specified sensitivity analyses, including various study groupings and CV outcomes. Although these findings were not statistically significant they were consistent. Because the number of events was small overall, the power for finding a statistically significant difference in a signal of this magnitude is low.
Additionally, a cardiovascular endpoint analysis was added to the postmarketing neuropsychiatric safety outcome study along with a non-treatment extension, [see Warnings and Precautions (5.5), Adverse Reactions (6.1), Clinical Studies (14.10)].14.9 Subjects with Major Depressive Disorder
CHANTIX was evaluated in a randomized, double-blind, placebo-controlled study of subjects aged 18 to 75 years with major depressive disorder without psychotic features (DSM-IV TR). If on medication, subjects were to be on a stable antidepressant regimen for at least two months. If not on medication, subjects were to have experienced a major depressive episode in the past 2 years, which was successfully treated. Subjects were randomized to CHANTIX 1 mg twice daily (N=256) or placebo (N=269) for a treatment of 12 weeks and then followed for 40 weeks post-treatment. Subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 (36%) compared to subjects treated with placebo (16%) and from week 9 through 52 (20%) compared to subjects treated with placebo (10%).Patients with Major Depressive Disorder (See also below: Neuropsychiatric Safety Study in Subjects with and without a History of Psychiatric Disorder)
Varenicline was evaluated in a randomized, double-blind, placebo-controlled study of 525 subjects with major depressive disorder without psychotic features (DSM-IV TR), on stable antidepressant treatment and/or who experienced a major depressive episode (which was successfully treated) in the past 2 years. Subjects aged 18 to 75 years were randomized to varenicline 1 mg BID (n=256) or placebo (n=269) for a treatment of 12 weeks and then followed for 40 weeks post-treatment. Quit rates in this study were in the range of those from studies in the general population of smokers.In general, the adverse events in this one-year study were quantitatively and qualitatively similar to those observed in studies in the general population of smokers.The following psychiatric AEs were more frequent in the varenicline group vs placebo: agitation (6.6% vs. 4.1%), depression (6.6% vs. 4.8%), tension (3.5% vs. 3.0%), hostility (2.0% vs. 0.4%) and restlessness (2.0% vs. 1.9%). No overall worsening of depression was observed during the study in neither varenicline or placebo treatment groups.The percentage of subjects with suicidal ideation and/or behavior during treatment were 6.0% and 7.5% respectively for the varenicline and placebo groups and 6.2% vs 5.8% for the non-treatment follow-up period. There was one event of intentional self-injury/possible suicide attempt during treatment (Day 73) in a subject with history of alcohol abuse in the placebo group. A possible suicide could not be ruled out in one subject who died by an overdose of illicit drugs 76 days after last dose of study drug in the varenicline group.Table 14. Continuous Abstinence (95% confidence interval), Study in Patients with Major Depressive Disorder (MDD)
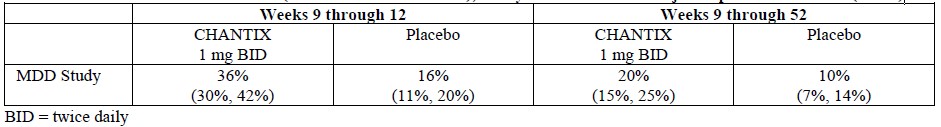
14.10 Postmarketing Neuropsychiatric Safety Outcome Trial
CHANTIX was evaluated in a randomized, double-blind, active and placebo-controlled trial that included subjects without a history of psychiatric disorder (non-psychiatric cohort, N=3912) and with a history of psychiatric disorder (psychiatric cohort, N=4003). Subjects aged 18-75 years, smoking 10 or more cigarettes per day were randomized 1:1:1:1 to CHANTIX 1 mg BID, bupropion SR 150 mg BID, NRT patch 21 mg/day with taper or placebo for a treatment period of 12 weeks; they were then followed for another 12 weeks post-treatment. [See Warnings and Precautions (5.1)]
A composite safety endpoint intended to capture clinically significant neuropsychiatric (NPS) adverse events included the following NPS adverse events: anxiety, depression, feeling abnormal, hostility, agitation, aggression, delusions, hallucinations, homicidal ideation, mania, panic, paranoia, psychosis, irritability, suicidal ideation, suicidal behavior or completed suicide.
As shown in Table 15, the use of CHANTIX, bupropion, and NRT in the non-psychiatric cohort was not associated with an increased risk of clinically significant NPS adverse events compared with placebo. Similarly, in the non-psychiatric cohort, the use of CHANTIX was not associated with an increased risk of clinically significant NPS adverse events in the composite safety endpoint compared with bupropion or NRT.Neuropsychiatric Safety Study in Subjects with and without a History of Psychiatric Disorder (see also WARNING AND PRECAUTIONS, Psychiatric Symptoms in Patients with and without Pre-existing Psychiatric Disorder or Symptoms)
Varenicline was evaluated in a randomized, double-blind, active and placebo-controlled study that included subjects with a history of psychiatric disorder (psychiatric cohort, N=4074) and subjects without a history of psychiatric disorder (non-psychiatric cohort, N=3984). Excluded psychiatric disorders included current substance abuse, dementias, impulse control and dissociative disorders. Subjects aged 18 to 75 years, smoking 10 or more cigarettes per day were randomized 1:1:1:1 to varenicline 1 mg BID, bupropion SR 150 mg BID, nicotine replacement therapy patch (NRT) 21 mg/day with taper or placebo for a treatment period of 12 weeks; they were then followed for another 12 weeks post-treatment.
The prospective primary safety endpoint was a composite of the following neuropsychiatric (NPS) adverse events (which mapped from 261 MedDRA preferred terms): severe events of anxiety, depression, feeling abnormal, or hostility; and moderate or severe events of agitation, aggression, delusions, hallucinations, homicidal ideation, mania, panic, paranoia, psychosis, suicidal ideation, suicidal behavior or completed suicide.
The primary diagnoses in the psychiatric cohort of the study were: Affective Disorders ~70%; Anxiety Disorders ~19%; Psychotic Disorders ~ 10%, and Borderline Personality Disorders ~ 1% with all patients judged to be clinically stable.
Table 8 shows the rates of the composite NPS adverse event primary end point by treatment group and the risk differences (RDs) (95% CI) vs placebo in each of the non-psychiatric and psychiatric cohort.
Table 15. Number of Patients with Clinically Significant or Serious NPS Adverse Events by Treatment Group Among Patients without a History of Psychiatric Disorder
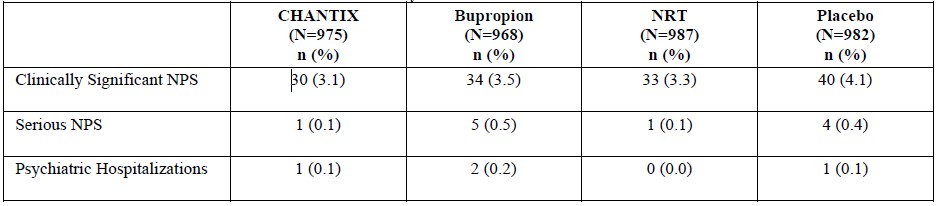
Table 8: Rates of Patients Reporting the Composite NPS AE Primary Endpoint by Treatment Group in Both Patient Cohorts
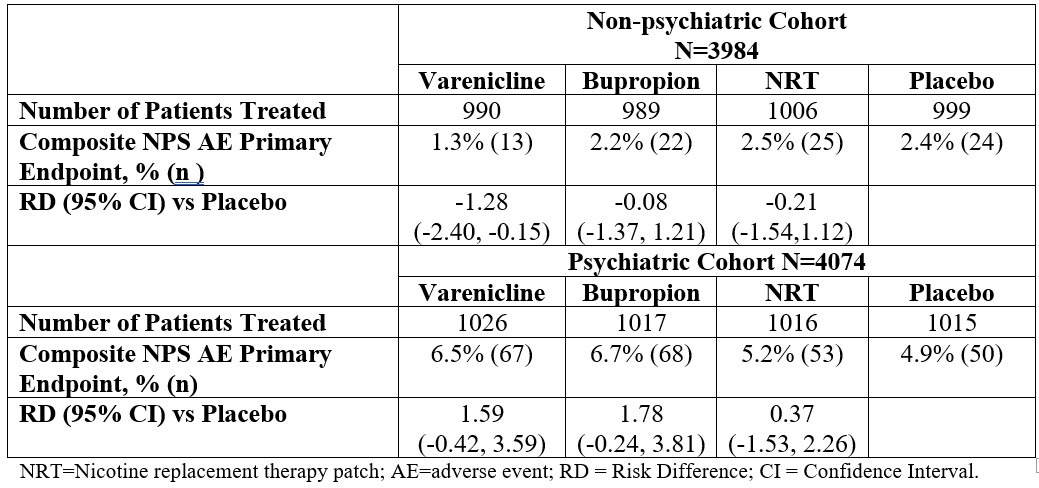
As shown in Table 16, there were more clinically significant NPS adverse events reported in patients in the psychiatric cohort in each treatment group compared with the non-psychiatric cohort. The incidence of events in the composite endpoint was higher for each of the active treatments compared to placebo: Risk Differences (RDs) (95%CI) vs placebo were 2.7% (-0.05, 5.4) for CHANTIX, 2.2% (-0.5, 4.9) for bupropion, and 0.4% (-2.2, 3.0) for NRT transdermal nicotine In the psychiatric cohort, there were more events reported in patients in each treatment group compared with the non-psychiatric cohort, and the incidence of events in the composite endpoint was higher for each of the active treatments compared to placebo.
However, in neither cohort (psychiatric or non-psychiatric) was the use of varenicline or bupropion associated with a significantly increased risk, compared with placebo, of NPS primary endpoint AEs (95% CIs were lower than or included zero).
Various sensitivity analyses were performed, including different expansions of the selected AE definitions. The sensitivity analyses did not reveal significantly increased rates of psychiatric adverse events for varenicline compared to placebo, nor compared to the two other treatments (bupropion, NRT).
The totality of psychiatric adverse events in the study is shown below (Table 9) for reference.Table 16. Number of Patients with Clinically Significant or Serious NPS Adverse Events by Treatment Group Among Patients with a History of Psychiatric Disorder
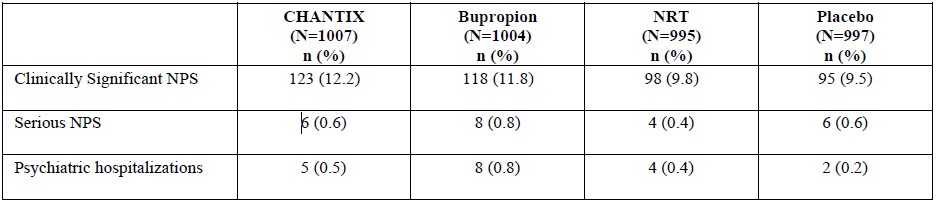
Table 9: Incidence of Adverse Events Coding to Preferred Terms in the Psychiatric Disorder System Organ Class (SOC) and/or Preferred Terms Pre-Specified for the Primary NPS Endpoint

There was one completed suicide, which occurred during treatment in a patient treated with placebo in the non-psychiatric cohort. There were no completed suicides reported in the psychiatric cohort.
In both cohorts, subjects treated with CHANTIX had a superior rate of CO-confirmed abstinence during weeks 9 through 12 and 9 through 24 compared to subjects treated with bupropion, nicotine patch and placebo.Suicidality
The percentage of subjects with suicidal ideation and/or behavior based on the Columbia-Suicide Severity Rating Scale (C-SSRS) was similar between the varenicline and placebo groups for both the non-psychiatric and psychiatric cohort, both during treatment and in the non-treatment follow-up, as shown in Table 10.
There was one completed suicide, which occurred during treatment in a subject treated with placebo, in the non-psychiatric cohort.Table 17 Continuous Abstinence (95% confidence interval), Study in Patients with or without a History of Psychiatric Disorder
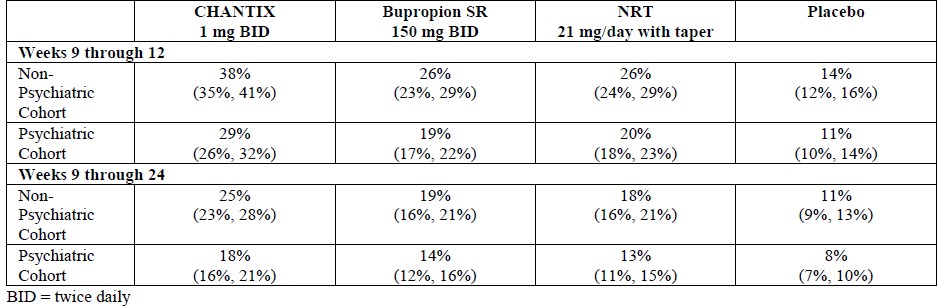
Cardiovascular Outcome Analysis
To obtain another source of data regarding the CV risk of CHANTIX, a cardiovascular endpoint analysis was added to the postmarketing neuropsychiatric safety outcome study along with a non-treatment extension. In the parent study (N=8027), subjects aged 18-75 years, smoking 10 or more cigarettes per day were randomized 1:1:1:1 to CHANTIX 1 mg BID, bupropion SR 150 mg BID, nicotine replacement therapy (NRT) patch 21 mg/day or placebo for a treatment period of 12 weeks; they were then followed for another 12 weeks post-treatment. The extension study enrolled 4590 (57.2%) of the 8027 subjects who were randomized and treated in the parent study and followed them for additional 28 weeks. Of all treated subjects, 1743 (21.7%) had a medium CV risk and 640 (8.0%) had a high CV risk, as defined by Framingham score. Note that one site from the parent study was excluded in the assessment of CV safety and two sites were excluded in the assessment of neuropsychiatric safety.
The primary CV endpoint was the time to major adverse CV event (MACE), defined as CV death, nonfatal myocardial infarction or nonfatal stroke during treatment. Deaths and CV events were adjudicated by a blinded, independent committee. Table 18 below shows the incidence of MACE and Hazard Ratios compared to placebo for all randomized subjects exposed to at least 1 partial dose of study treatment in the parent study.Table 18. The Incidence of MACE and Hazard Ratios in the Cardiovascular Safety Assessment Trial in Subjects without or with a History of Psychiatric Disorder
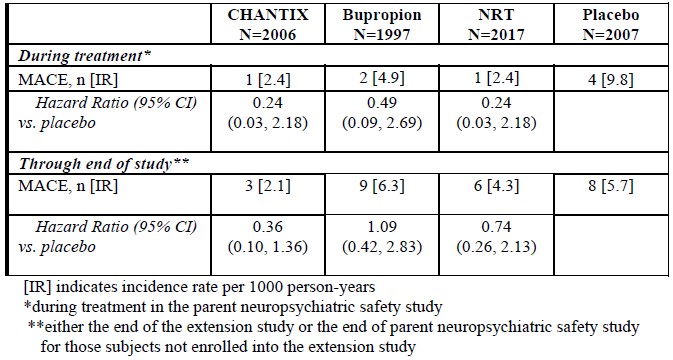
For this study, MACE+ was defined as any MACE or a new onset or worsening peripheral vascular disease (PVD) requiring intervention, a need for coronary revascularization, or hospitalization for unstable angina. Incidence rates of MACE+ and all-cause mortality for all randomized subjects exposed to at least 1 partial dose of study treatment in the parent study are shown for all treatment groups during treatment, and through end of study in the Table 19 below. Table 19. The Incidence of MACE+ and All-Cause Death in the Cardiovascular Safety Assessment Trial in Subjects without or with a History of Psychiatric
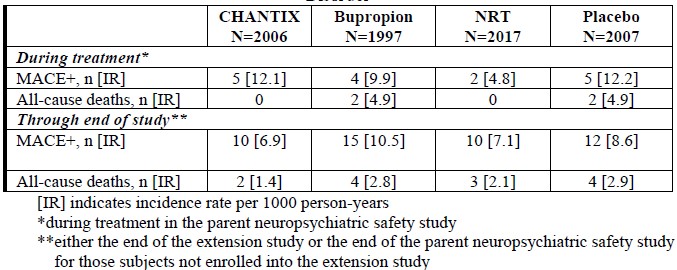
The number of subjects who experienced MACE, MACE+ and all-cause death was similar or lower among patients treated with CHANTIX than patients treated with placebo. The number of events observed overall was too low to distinguish meaningful differences between the treatment arms.
16 HOW SUPPLIED/STORAGE AND HANDLING
CHANTIX is supplied for oral administration in two strengths: a 0.5 mg capsular biconvex, white to off-white, film-coated tablet debossed with "Pfizer" on one side and "CHX 0.5" on the other side and a 1 mg capsular biconvex, light blue film-coated tablet debossed with "Pfizer" on one side and "CHX 1.0" on the other side. CHANTIX is supplied in the following package configurations:
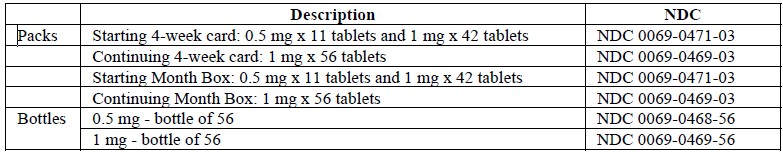
Store at 25ºC (77ºF); excursions permitted to 15–30ºC (59–86ºF) (see USP Controlled Room Temperature).
STORAGE AND STABILITY
APO-VARENICLINE is supplied for oral administration in two strengths: 0.5 mg: White colored, modified capsule-shaped, biconvex, film coated tablets, with engraved “APO” on one side and “VAR” over “0.5” on the other side. Each tablet contains 0.5 mg of varenicline (as tartrate). Supplied in high-density polyethylene (HDPE) bottles of 56, 60 &100 tablets and in blisters of 28 & 30 tablets.1 mg: Blue colored, modified capsule-shaped, biconvex, film coated tablets, engraved with “APO” on one side and “VAR” over “1” on the other side. Each tablet contains 1 mg of varenicline (as tartrate). Supplied in high-density polyethylene (HDPE) bottles of 30, 56, 1000 & 10000 tablets and in blister pack of 28 & 30 tablets. Initial dosing pack: 53 counts - Includes 0.5 mg tablets in blister strips of 11 tablets and 1 mg tablets in blister strips of 42 tablets.Continuation dosing pack: 1 mg tablets in blister strips of 56 tablets
Store at room temperature (15ºC to 30ºC).Patient Counseling Information 17 PATIENT COUNSELING INFORMATION
See FDA-approved patient labeling (Medication Guide)
Initiate Treatment and Continue to Attempt to Quit if Lapse
Instruct patients to set a date to quit smoking and to initiate CHANTIX treatment one week before the quit date. Alternatively, the patient can begin CHANTIX dosing and then set a date to quit smoking between days 8 and 35 of treatment. Encourage patients to continue to attempt to quit if they have early lapses after quit day [see Dosage and Administration (2.1)].
For patients who are sure that they are not able or willing to quit abruptly, a gradual approach to quitting smoking with CHANTIX may be considered. Patients should begin CHANTIX dosing and reduce smoking during the first 12 weeks of treatment, then quit by the end of that period and continue treatment for an additional 12 weeks for a total of 24 weeks [see Dosage and Administration (2.1)].
Encourage patients who are motivated to quit and who did not succeed in stopping smoking during prior CHANTIX therapy for reasons other than intolerability due to adverse events, or who relapsed after treatment to make another attempt with CHANTIX once factors contributing to the failed attempt have been identified and addressed [see Dosage and Administration (2.1), Clinical Studies (14.6)].
How to Take
Advise patients that CHANTIX should be taken orally after eating, and with a full glass of water [see Dosage and Administration (2.1)].
Starting Week Dosage
Instruct patients on how to titrate CHANTIX, beginning at a dose of 0.5 mg/day. Explain that one 0.5 mg tablet should be taken daily for the first three days, and that for the next four days, one 0.5 mg tablet should be taken in the morning and one 0.5 mg tablet should be taken in the evening [see Dosage and Administration (2.1)].
Continuing Weeks Dosage
Advise patients that, after the first seven days, the dose should be increased to one 1 mg tablet in the morning and one 1 mg tablet in the evening [see Dosage and Administration (2.1)].
Dosage Adjustment for CHANTIX or Other Drugs
Inform patients that nausea and insomnia are side effects of CHANTIX and are usually transient; however, advise patients that if they are persistently troubled by these symptoms, they should notify the prescribing physician so that a dose reduction can be considered.
Inform patients that some drugs may require dose adjustment after quitting smoking [see Dosage and Administration (2.1)].
Counseling and Support
Provide patients with educational materials and necessary counseling to support an attempt at quitting smoking [see Dosage and Administration (2.1)].
Neuropsychiatric Adverse Events
Inform patients that some patients have experienced changes in mood (including depression and mania), psychosis, hallucinations, paranoia, delusions, homicidal ideation, aggression, hostility, agitation, anxiety, and panic, as well as suicidal ideation and suicide when attempting to quit smoking while taking CHANTIX. Instruct patients to discontinue CHANTIX and contact a healthcare professional if they experience such symptoms [see Warnings and Precautions (5.1), Adverse Reactions (6.2)].
History of Psychiatric Illness
Encourage patients to reveal any history of psychiatric illness prior to initiating treatment.
Nicotine Withdrawal
Inform patients that quitting smoking, with or without CHANTIX, may be associated with nicotine withdrawal symptoms (including depression or agitation) or exacerbation of pre-existing psychiatric illness.
Seizures
Encourage patients to report any history of seizures or other factors that can lower seizure threshold. Instruct patients to discontinue CHANTIX and contact a healthcare provider immediately if they experience a seizure while on treatment [see Warnings and Precautions (5.2)].
Interaction with Alcohol
Advise patients to reduce the amount of alcohol they consume while taking CHANTIX until they know whether CHANTIX affects their tolerance for alcohol [see Warnings and Precautions (5.3), Adverse Reactions (6.2)].
Driving or Operating Machinery
Advise patients to use caution driving or operating machinery until they know how quitting smoking and/or varenicline may affect them [see Warnings and Precautions (5.4)].
Cardiovascular Events
Patients should be instructed to notify their healthcare providers of symptoms of new or worsening cardiovascular events and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke [see Warnings and Precautions (5.5), Adverse Reactions (6.1)].
Somnambulism
Patients should be instructed to discontinue CHANTIX and notify their healthcare providers if they experience somnambulism [see Warnings and Precautions (5.6)].
Angioedema
Inform patients that there have been reports of angioedema, with swelling of the face, mouth (lip, gum, tongue) and neck (larynx and pharynx) that can lead to life-threatening respiratory compromise. Instruct patients to discontinue CHANTIX and immediately seek medical care if they experience these symptoms [see Warnings and Precautions (5.7), Adverse Reactions (6.2)].
Serious Skin Reactions
Inform patients that serious skin reactions, such as Stevens-Johnson Syndrome and erythema multiforme, were reported by some patients taking CHANTIX. Advise patients to stop taking CHANTIX at the first sign of rash with mucosal lesions or skin reaction and contact a healthcare provider immediately [see Warnings and Precautions (5.8), Adverse Reactions (6.2)].
Vivid, Unusual, or Strange Dreams
Inform patients that they may experience vivid, unusual or strange dreams during treatment with CHANTIX.
Pregnancy and Lactation
Patients who are pregnant or breastfeeding or planning to become pregnant should be advised of: the risks of smoking to a pregnant mother and her developing baby, the potential risks of CHANTIX use during pregnancy and breastfeeding, and the benefits of smoking cessation with and without CHANTIX. Advise breastfeeding women to monitor the infant for seizures and vomiting [see Use in Specific Populations (8.1 and 8.2)].
Patient Counselling Information
Consumer Information is included in the package of APO-VARENICLINE dispensed to the patient.
Prior to prescribing APO-VARENICLINE, physicians should:- Discuss with the patient the expected benefits and risks of APO-VARENICLINE, as well as those of all smoking-cessation options.
- Inform the patients that quitting smoking, with or without treatment, may be associated with nicotine withdrawal symptoms (including depression, irritation or agitation) or exacerbation of pre-existing psychiatric disorder.
- Encourage the patient to reveal any history of psychiatric disorder prior to initiating treatment. Patients with such history who are trying to stop smoking should be monitored by their physician for new or worsened psychiatric events.
- Advise patients:
- not to engage in potentially hazardous tasks, such as driving a car or operating dangerous machines, until they know how APO-VARENICLINE may affect them. In some cases, patients have reported somnolence, dizziness, loss of consciousness, seizures or difficulty concentrating while driving.
- that some people have reported seizures while taking varenicline and encourage them to report any history of seizures or other factors that can lower seizure threshold. Instruct patients to discontinue APO-VARENICLINE and immediately contact a healthcare provider if they experience a seizure while on treatment.
- that there have been post-marketing reports of serious neuropsychiatric symptoms in patients being treated with varenicline, including anxiety, psychosis, mood swings, aggression, depressed mood, agitation, hallucinations, hostility, changes in behavior or thinking, suicidal ideation, suicidal behavior and suicide, as well as worsening of pre-existing psychiatric disorder.
- that i) new or worse cardiovascular events (heart and stroke) have been reported, primarily in those who already have cardiovascular problems and ii) based on available data, it is not possible to determine whether varenicline increases the risk of cardiovascular events.
- Patients should be instructed to read the patient information leaflet supplied with every APO-VARENICLINE prescription before starting their APO-VARENICLINE pills. This leaflet is approved by Health Canada and is Part III of the APO-VARENICLINE Product Monograph.
- Patients should also be provided with educational materials and necessary counselling to support an attempt at quitting smoking, including a review of the overall smoking cessation plan with the physician.
- Patients should call government-funded toll-free provincial Quit Lines which can be used to support a quit attempt.
- Patients should be informed that there are three choices in setting a quit date when using APO-VARENICLINE, and discuss with their physician which one is best for them.
- Patients should be instructed on how to titrate APO-VARENICLINE:
- Begin at a dose of 0.5 mg per day. Prescribers should explain that one 0.5 mg tablet should be taken daily for the first three days, and then for the next four days, two 0.5 mg tablets should be taken daily: one in the morning and one in the evening.Following this one week of titration, there are two dosing options: the dose can remain at 0.5 mg twice daily or can go up to 1 mg twice daily, depending on the physician judgment and patient preference. Based on the limited data available, the two doses do not appear different in terms of either quit rates, or rates of serious psychiatric side effects (see DOSAGE AND ADMINISTRATION, Dosing Considerations).
- If needed, the dose can be changed depending on how well the patient tolerates APO-VARENICLINE and how effective the doctor and patient consider it is in helping the patient quit smoking.
- Patients should be informed that the maximum dose of APO-VARENICLINE is 1 mg twice a day.
- Patients should be encouraged to continue in their quit attempt if they have early lapses after their quit date.
- Patients should be encouraged to inform friends and family members of their quit attempt which includes treatment with APO-VARENICLINE and ask for their support and help in monitoring for any changes in behavior or thinking that are not typical for the patient.
- Patients should be advised that drinking alcohol may increase the risk of experiencing psychiatric adverse events during treatment with APO-VARENICLINE.
- Patients with pre-existing psychiatric disorder should be instructed that if they develop worsened or new symptoms, to report these to their healthcare provider; dose adjustments of psychiatric medications or APO-VARENICLINE may be considered.
- Patients should be informed that if they experience thoughts, moods or behaviours that are strongly atypical and concerning while on smoking-cessation medication, including APO-VARENICLINE, the medication should be discontinued immediately, urgent medical help sought as needed, and the symptoms reported to their healthcare provider.
- Patients should be informed that:
- they may experience vivid, unusual or strange dreams during treatment with APO-VARENICLINE.
- nausea is the most common adverse event associated with varenicline and is usually transient. APO-VARENICLINE should be taken after eating and with a full glass of water. Patients should be advised that if they are persistently troubled by this symptom, a dose reduction may be considered.
- if they experience sleepwalking, they should discontinue APO-VARENICLINE and notify their healthcare provider.
- there have been reports of angioedema, with swelling of the face, mouth (tongue, lips and gums) and neck (pharynx and larynx) that can lead to life-threatening respiratory compromise. Patients should be instructed to discontinue APO-VARENICLINE and seek immediate emergency medical attention if they experience these symptoms.
- serious skin reactions, such as Stevens-Johnson syndrome and erythema multiforme, were reported by some patients taking varenicline. Patients should be advised to stop taking APO-VARENICLINE at the first sign of rash with mucosal lesions or skin reaction and seek immediate emergency medical attention.
- Patients should be instructed to notify their healthcare providers of symptoms of new or worsening cardiovascular events and to seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke.
Company Information This product’s label may have been updated. For full prescribing information, please visit www.pfizer.com

MEDICATION GUIDE PART III: CONSUMER INFORMATION Medication Guide CHANTIX® (CHANT-iks)
(varenicline)TabletsPrAPO-VARENICLINE
Varenicline Tablets
0.5 mg and 1 mg varenicline (as varenicline tartrate)Read this information each time you refill your prescription in case new information has been added.This leaflet is part III of a three-part "Product Monograph" published when APO-VARENICLINE was approved for sale in Canada and is designed specifically for Consumers. This leaflet is a summary and will not tell you everything about APO-VARENICLINE. Contact your doctor or pharmacist if you have any questions about the drug. What is the most important information I should know about CHANTIX?
When you try to quit smoking, with or without CHANTIX, you may have symptoms that may be due to nicotine withdrawal, including:

Some people have even experienced suicidal thoughts when trying to quit smoking without medication. Sometimes quitting smoking can lead to worsening of mental health problems that you already have, such as depression.
Some people have had serious side effects while taking CHANTIX to help them quit smoking, including:
New or worse mental health problems, such as changes in behavior or thinking, aggression, hostility, agitation, depressed mood, or suicidal thoughts or actions.
Some people had these symptoms when they began taking CHANTIX, and others developed them after several weeks of treatment, or after stopping CHANTIX. These symptoms happened more often in people who had a history of mental health problems before taking CHANTIX, than in people without a history of mental health problems.
Stop taking CHANTIX and call your healthcare provider right away if you, your family, or caregiver notice any of these symptoms. Work with your healthcare provider to decide whether you should continue to take CHANTIX. In many people, these symptoms went away after stopping CHANTIX, but in some people symptoms continued after stopping CHANTIX. It is important for you to follow-up with your healthcare provider until your symptoms go away.
Before taking CHANTIX, tell your healthcare provider if you have ever had depression or other mental health problems. You should also tell your healthcare provider about any symptoms you had during other times you tried to quit smoking, with or without CHANTIX.
What is the most important information I should know about APO-VARENICLINE?
When you try to quit smoking, with or without APO-VARENICLINE, you may have symptoms that may be due to nicotine withdrawal, including- the urge to smoke
- depressed mood
- trouble sleeping
- irritability
- frustration
- anger
- feeling anxious
- difficulty concentrating
- restlessness
- decreased heart rate
- increased appetite or weight gain.
Some people have even experienced suicidal thoughts when trying to quit smoking without medication. Sometimes quitting smoking can lead to worsening of mental health problems that you already have, such as depression.
Mental Health Problems
Some people have had serious side effects while taking APO-VARENICLINE to help them quit smoking, including changes in behavior or thinking, hostility, agitation, aggression, depressed mood, or suicidal thoughts or actions. These symptoms have occurred in people with previous mental health problems, as well as in those with no previous history. For some people, these symptoms began when they started taking APO-VARENICLINE while for others, they began after several weeks of treatment, or shortly after stopping APO-VARENICLINE. Before taking any quit-smoking treatment, including APO-VARENICLINE, tell your healthcare provider (doctor, pharmacist or nurse): - if you have ever had depression or other mental health problems;
- about any concerning symptoms you had during other times you tried to quit smoking, with or without medication.
Inform your friends and family members of your quit attempt with APO-VARENICLINE and ask for their support and help in monitoring for any changes in behavior or thinking that are not normal.
Drinking alcohol may increase the risk of having mental health problems during your treatment with APO-VARENICLINE.
Patients with history of mental health problems (e.g. depression, anxiety, schizophrenia): If you have had mental health problems before taking APO-VARENICLINE, your healthcare provider will monitor you while you try to quit smoking with APO-VARENICLINE. If you develop worsened or new symptoms, talk to your healthcare provider right away because changing the dose (of APO-VARENICLINE or other medications) may make a difference.
All patients/General: If you have thoughts, moods or behaviours that are severe, concerning or very abnormal for you, stop taking APO-VARENICLINE right away, seek medical help, and tell your healthcare provider about your symptoms. In many people, these symptoms went away after stopping APO-VARENICLINE, but not in all. It is important for you to follow up with your healthcare provider until your symptoms go away.
Allergic Reactions
Some people can have allergic reactions to APO-VARENICLINE. Some of these allergic reactions can be life-threatening and include: swelling of the face, mouth, and throat that can cause trouble breathing. If you have these symptoms, stop taking APO-VARENICLINE and seek immediate emergency medical attention.
Serious Skin Reactions
Some people can have serious skin reactions while taking APO-VARENICLINE. These can include rash, swelling, redness, and peeling of the skin. Some of these reactions can become life-threatening. If you have a rash with peeling skin or blisters in your mouth, around the eyes or genitals, stop taking APO-VARENICLINE and seek immediate emergency medical attention.
What is CHANTIX?
CHANTIX is a prescription medicine to help people stop smoking.Quitting smoking can lower your chances of having lung disease, heart disease or getting certain types of cancer that are related to smoking.CHANTIX has not been shown to be effective in children 16 years of age and under. CHANTIX should not be used in children 16 years of age and under.It is not known if CHANTIX is safe and effective when used with other stop smoking medicines.ABOUT THIS MEDICATION
What the medication is used for:
APO-VARENICLINE is a prescription medicine which is used in combination with supportive counselling to help motivated adults stop smoking.
What it does:
APO-VARENICLINE can help to relieve the craving and withdrawal symptoms associated with stopping smoking.
APO-VARENICLINE does not contain nicotine, but it has been shown to affect the nicotine receptor in the brain that is thought to be most related to smoking addiction.
APO-VARENICLINE can affect this receptor in two opposite ways: it acts like a weaker version of nicotine, and also blocks nicotine from getting to the receptor because it binds more tightly. Although it is thought that this may be, in part, how APO-VARENICLINE works, it is not known exactly how the drug works in people.
Who should not take CHANTIX?
Do not take CHANTIX if you have had a serious allergic or skin reaction to CHANTIX. Symptoms may include:

When it should not be used:
Do not take APO-VARENICLINE if you:- are allergic (hypersensitive) to varenicline tartrate or any of the other ingredients of APO-VARENICLINE (see list below of non-medicinal ingredients).
- are using nicotine replacement therapy, such as patches, gum or inhaler. The combination of APO-VARENICLINE and nicotine replacement therapy is not expected to improve your chances of quitting, and may result in more side effects than with APO-VARENICLINE alone.
What should I tell my healthcare provider before taking CHANTIX?
See “What is the most important information I should know about CHANTIX?”
Before you take CHANTIX, tell your healthcare provider if you:- use other treatments to quit smoking. Using CHANTIX with a nicotine patch may cause nausea, vomiting, headache, dizziness, upset stomach, and tiredness to happen more often than if you just use a nicotine patch alone.
- have kidney problems or get kidney dialysis. Your healthcare provider may prescribe a lower dose of CHANTIX for you.
- have a history of seizures
- drink alcohol
- have heart or blood vessel problems
- have any other medical conditions
- are pregnant or plan to become pregnant.
- are breastfeeding. It is not known if CHANTIX passes into breast milk. If you breastfeed and take CHANTIX, monitor your baby for seizures as well as spitting up or vomiting more than normal.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Your healthcare provider may need to change the dose of some of your medicines when you stop smoking.
You should not use CHANTIX while using other medicines to quit smoking. Tell your healthcare provider if you use other treatments to quit smoking.
Know the medicines you take. Keep a list of them with you to show your healthcare provider and pharmacist when you get a new medicine.
WARNINGS AND PRECAUTIONS
BEFORE you use APO-VARENICLINE talk to your healthcare provider if you:- have experienced depression or any other mental health problems. Your healthcare provider will monitor you for new or worsened emotional or behavioral problems during treatment with APO-VARENICLINE.
- have any problems with your kidneys, as you may need a lower dose of APO-VARENICLINE.
- have heart or blood vessel (cardiovascular) problems.
- have a history of seizures.
- have any other medical conditions.
- are pregnant, are breastfeeding or plan to become pregnant (see “Pregnancy” and “Breastfeeding”
- below).have diabetes. APO-VARENICLINE can potentially affect your blood sugar regulation, and you may need to monitor your blood sugar more often. If you notice changes, discuss this with your healthcare provider.
How should I take CHANTIX? - There are 3 ways that you can use CHANTIX to help you quit smoking. Talk to your healthcare provider about thefollowing 3 ways to use CHANTIX:
- Choose a quit date when you will stop smoking. Start taking CHANTIX 1 week (7 days) before your quit date. Take CHANTIX for 12 weeks.
OR
-
Start taking CHANTIX before you choose a quit date. Pick a date to quit smoking that is between days 8 and 35 of treatment. Take CHANTIX for 12 weeks.
OR
-
If you are sure that you are not able or willing to quit smoking right away, start taking CHANTIX and reduce smoking during the first 12 weeks of treatment, as follows:

Aim to quit by the end of the 12th week of treatment, or sooner if you feel ready.
Continue to take CHANTIX for another 12 weeks, for a total of 24 weeks of treatment.
Starting CHANTIX before your quit date gives CHANTIX time to build up in your body. You can keep smoking during this time. Take CHANTIX exactly as prescribed by your healthcare provider.
- Choose a quit date when you will stop smoking. Start taking CHANTIX 1 week (7 days) before your quit date. Take CHANTIX for 12 weeks.
- CHANTIX comes as a white tablet (0.5 mg) and a blue tablet (1 mg). You start with the white tablet and then usually go to the blue tablet. See the chart below for dosing instructions for adults.

- Make sure that you try to stop smoking on your quit date. If you slip-up and smoke, try again. Some people need to take CHANTIX for a few weeks for CHANTIX to work best.
- Most people will take CHANTIX for up to 12 weeks. If you have completely quit smoking by 12 weeks, your healthcare provider may prescribe CHANTIX for another 12 weeks to help you stay cigarette-free.
- Take CHANTIX after eating and with a full glass (8 ounces) of water.
- This dosing schedule may not be right for everyone. Talk to your healthcare provider if you are having side effects such as nausea, strange dreams, or sleep problems. Your healthcare provider may want to reduce your dose.
- If you miss a dose of CHANTIX, take it as soon as you remember. If it is almost time for your next dose, skip the missed dose. Just take your next dose at your regular time.
PROPER USE OF THIS MEDICATION
You are more likely to stop smoking if you are motivated to stop. Your healthcare provider can provide advice, support and sources of further information to help ensure your attempt to stop smoking is successful.
To increase the chances of success, APO-VARENICLINE should be used in combination with supportive counselling as recommended by your healthcare provider. Varenicline was used in combination with supportive counselling in the clinical trials. At any time, you can also call government-funded toll-free provincial Quit Lines, to speak to a knowledgeable and supportive specialist; these phone numbers are available on the Health Canada website.
Always take APO-VARENICLINE exactly as your healthcare provider has told you. You should check with your healthcare provider if you are not sure. REMEMBER: This medication has been prescribed specifically for you. Do not give it to anyone else.
Setting Your Quit Date: Starting treatment before your quit date lets APO-VARENICLINE build up in your body.
You can keep smoking until your quit date.
There are three ways to set your quit date when using APO-VARENICLINE. Talk to your healthcare provider about which way is best for you:- Fixed quit approach: Set a quit date when you will stop smoking. Start taking APO-VARENICLINE 8 to 14 days (1 to 2 weeks) before your quit date. You should take APO-VARENICLINE for 12 weeks. After 12 weeks of treatment, your healthcare provider may recommend an additional 12 weeks of treatment.
Or
-
Flexible quit approach: Start taking APO-VARENICLINE, then quit smoking between Day 8 and Day 35 after the start of your treatment (i.e. between Weeks 2 and 5). You should take APO-VARENICLINE for 12 weeks. After 12 weeks of treatment, your healthcare provider may recommend an additional 12 weeks of treatment.
Or
-
Gradual quit approach: Start taking APO-VARENICLINE and reduce smoking with a goal to quit smoking by end of 12 weeks of treatment. For example, reduce smoking by half by the 4th week, another half by the 8th week (down to 25%) and then quit by the end of the 12th week. You may quit any time before the end of 12 weeks of treatment, if you are able to. Continue treatment for an additional 12 weeks for a total of 24 weeks.
The maximum dose of APO-VARENICLINE is 1 mg twice a day.
Write down, and keep in a visible or convenient place (for example on the fridge or on the APO-VARENICLINE pack), the date that you started APO- VARENICLINE, your quit date, and the date to stop taking APO-VARENICLINE. Make sure that you try to stop smoking on your quit date. If you slip-up and smoke after that target date, keep trying. Some people need a few weeks on APO-VARENICLINE for it to work best.
Dosing Options:
APO-VARENICLINE should be taken after eating and with a full glass of water. Regardless of which dose is prescribed, the first week on APO-VARENICLINE is the same, and is described in the following table:
Week 1 Dosing Schedule:

After the first week, your healthcare provider may recommend to stay at 0.5 mg twice a day (OPTION 1) or go up to 1 mg twice a day (OPTION 2).Week 2 (day 8) to the end of treatment
OPTION 1: Continue on 0.5 mg twice a day

Or
OPTION 2: Start taking 1 mg twice a day

Based on the limited data available, the two doses do not appear different in terms of either quit rates, or rates of serious mental health side effects (your healthcare provider can provide more information).
Discussion with your healthcare provider is important in order to choose the dose that is best for you.
If needed, the dose can be changed depending on how well you tolerate APO-VARENICLINE and how effective your healthcare provider and you consider it is in helping you quit smoking. Your healthcare provider will help decide what dose is right for you.
Your healthcare provider may recommend to gradually lower the dose at the end of the treatment period rather than stopping abruptly.
What should I avoid while taking CHANTIX? - Use caution when driving or operating machinery until you know how CHANTIX affects you. CHANTIX may make you feel sleepy, dizzy, or have trouble concentrating, making it hard to drive or perform other activities safely.
- Decrease the amount of alcoholic beverages that you drink during treatment with CHANTIX until you know if CHANTIX affects your ability to tolerate alcohol. Some people have experienced the following when drinking alcohol during treatment with CHANTIX:

Can I smoke while taking APO-VARENICLINE? You can keep smoking prior to your quit date.Smoking after your quit date will reduce your chance of breaking your smoking addiction.Some people have reported a change in the taste of cigarettes after starting APO-VARENICLINE.Drinking alcohol during treatment with APO-VARENICLINE may increase the risk of mental health symptoms.Reported experiences include: - unusual or sometimes aggressive behavior;
- more intoxicated than expected from the amount of alcohol;
-
no memory of things that have happened.
Use of APO-VARENICLINE with other therapies for smoking-cessation:
The safety and benefits of taking APO-VARENICLINE in combination with other medicines for stopping smoking have not been studied. Taking APO-VARENICLINE in combination with other smoking-cessation therapies (e.g., nicotine replacement therapy) is therefore not recommended. Using APO-VARENICLINE in combination with nicotine replacement therapies (e.g., patch gum or inhaler) is not likely to increase your chances of quitting smoking, and it may result in more side effects than with APO-VARENICLINE alone.
What are the possible side effects of CHANTIX?
Serious side effects of CHANTIX may include:- See “What is the most important information I should know about CHANTIX?”
- Seizures. Some people have had seizures during treatment with CHANTIX. In most cases, the seizures have happened during the first month of treatment with CHANTIX. If you have a seizure during treatment with CHANTIX, stop taking CHANTIX and contact your healthcare provider right away.
-
New or worse heart or blood vessel (cardiovascular) problems, mostly in people, who already have cardiovascular problems. Tell your healthcare provider if you have any changes in symptoms during treatment with CHANTIX.
Get emergency medical help right away if you have any of the following symptoms of a heart attack, including:
- chest discomfort (uncomfortable pressure, squeezing, fullness or pain) that lasts more than a few minutes, or that goes away and comes back
- pain or discomfort in one or both arms, back, neck, jaw or stomach
- shortness of breath, sweating, nausea, vomiting, or feeling lightheaded associated with chest discomfort
- Sleepwalking can happen with CHANTIX, and can sometimes lead to behavior that is harmful to you or other people, or to property. Stop taking CHANTIX and tell your healthcare provider if you start sleepwalking.
- Allergic reactions can happen with CHANTIX. Some of these allergic reactions can be life-threatening.
-
Serious skin reactions, including rash, swelling, redness, and peeling of the skin. Some of these skin reactions can become life-threatening.
Stop taking CHANTIX and get medical help right away if you have any of the following symptoms:
- swelling of the face, mouth (tongue, lips, and gums), throat or neck
- trouble breathing
- rash with peeling skino blisters in your mouth
The most common side effects of CHANTIX include: - nausea
- sleep problems (trouble sleeping or vivid, unusual, or strange dreams)
- constipation
- gas
- vomiting
Tell your healthcare provider about side effects that bother you or that do not go away.
These are not all the side effects of CHANTIX. Ask your healthcare provider or pharmacist for more information.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
The effects of changes in your body resulting from stopping smoking, with or without treatment with APO-VARENICLINE, may alter the way other drugs work. Tell your healthcare provider about all your other medicines, including prescription and nonprescription medicines, vitamins and herbal supplements. Especially, tell your healthcare provider if you take: - Insulin
- Asthma medicines (theophylline)
- Blood thinner (warfarin)
as an adjustment of the dose of these medicines may be necessary once you are smoke-free
See “What is the most important information I should know about APO-VARENICLINE?”
Pregnancy
Talk to your healthcare provider if you are pregnant or planning to become pregnant.
You should not take APO-VARENICLINE while you are pregnant. It is unknown if APO-VARENICLINE will harm your unborn baby.
It is best to stop smoking before you get pregnant.
Breastfeeding
You should ask your healthcare provider for advice before taking any medication, including APO-VARENICLINE, if you are breastfeeding, as the medicine may pass into breast milk.
APO-VARENICLINE is not recommended for use in children under 18 years of age.
Accidental Injury, including while Driving, Operating Machinery
Do not engage in potentially hazardous tasks, such as driving a car or operating dangerous machines, until you know how APO-VARENICLINE may affect you. In some cases, people have reported sleepiness, dizziness, blackouts, seizures or difficulty concentrating while driving.
Seizures
Tell your healthcare provider if you have experienced seizures or have epilepsy before you start APO-VARENICLINE treatment. Some people have reported seizures while taking varenicline, both with and without a history of seizures.
Heart or Stroke Events
New or worse heart or blood vessel (cardiovascular) problems have been reported in people taking varenicline, primarily in those who already have cardiovascular problems.
From the information available to date, it is not possible to determine whether APO-VARENICLINE increases the risk of heart or stroke events.
Tell your healthcare provider if you have any changes in cardiovascular symptoms during treatment with APO-VARENICLINE. Get emergency medical help right away if you have symptoms of a heart attack, including any of the following:- Chest discomfort (uncomfortable pressure, squeezing, fullness or pain) that lasts more than a few minutes, or that goes away and comes back.
- Pain or discomfort in one or both arms, back, neck, jaw or stomach.
- Shortness of breath, sweating, nausea, vomiting, or feeling lightheaded associated with chest discomfort. Get emergency medical help right away if you have symptoms of a stroke, including any of the following:
- Weakness - Sudden loss of strength or sudden numbness in the face, arm or leg even if temporary.
- Trouble speaking - Sudden difficulty speaking or understanding or sudden confusion, even if temporary.
- Vision problems - Sudden trouble with vision, even if temporary.
- Headache - Sudden severe and unusual headache.
- Dizziness - Sudden loss of balance, especially with any of the above signs.
Sleepwalking
Sleepwalking has been reported in patients taking APO-VARENICLINE, and may sometimes lead to behaviour that is harmful to you or other people or property. Stop taking APO-VARENICLINE and tell your healthcare provider if you start sleepwalking. Whether you are taking medication to stop smoking or not, the following are symptoms you may feel: depressed, short-tempered, frustrated or angry, nervous, impatient; have difficulty concentrating. Your appetite may increase, and you may gain some weight.Like all medicines, APO-VARENICLINE can cause side effects, although not everybody gets them.The common side effects are mostly mild to moderate and these usually occur in the first weeks of treatment.Some of the most common side effects you should be aware of include:- Nausea, vomiting
- Trouble sleeping
- Headache
- Abnormal dreams (vivid, unusual, or increased dreaming; rarely may include nightmares)
- Sleepiness, tiredness, dizziness
- Constipation, diarrhea, gas
See “What is the most important information I should know about APO-VARENICLINE?”
Stop taking APO-VARENICLINE if you experience severe or unusual feelings of agitation, aggression, depressed mood, hostility, hallucinations, or if you have thoughts of self-harm or harm to others. Tell your healthcare provider about your symptoms.
Allergic Reactions
Some people have allergic reactions to APO-VARENICLINE. Some of these allergic reactions can be life-threatening and include: swelling of the face, mouth (lips, gums, tongue), and throat can cause trouble breathing. If you have these symptoms, stop taking APO-VARENICLINE and seek immediate emergency medical attention.
Serious Skin Reactions
Some people can have serious skin reactions while taking APO-VARENICLINE. These can include rash, swelling, redness, and peeling of the skin. Some of these reactions can become life-threatening. If you have a rash with peeling skin, or blistering of the mouth, around the eyes or genitals, stop taking APO-VARENICLINE and seek immediate emergency medical attention.
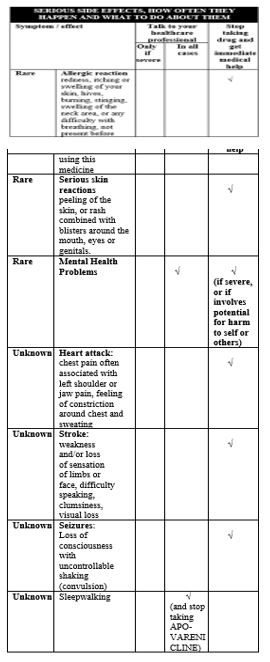
Overdose:
If you think you have taken too much APO-VARENICLINE, contact your healthcare professional, hospital emergency department or regional Poison Control Centre immediately, even if there are no symptoms.
Missed Dose:
Do not take a double dose to make up for a forgotten tablet. It is important that you take APO-VARENICLINE regularly at the same time each day. If you forget to take a dose, take it as soon as you remember, as long as it is within a few hours of the missed dose. If it has been longer than a few hours since the missed dose, or if you do not remember whether you took a dose or not, then skip that dose, and wait to take the next dose at the correct time. If you have any further questions on the use of this product, ask your healthcare provider - Dizziness - Sudden loss of balance, especially with any of the above signs.
How should I store CHANTIX? - Store CHANTIX at room temperature, between 68°F to 77°F (20°C to 25°C).
Keep CHANTIX and all medicines out of the reach of children.
Store APO-VARENICLINE at room temperature (15°C to 30°C).
Keep out of the reach and sight of children.General information about the safe and effective use of CHANTIX.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use CHANTIX for a condition for which it was not prescribed. Do not give your CHANTIX to other people, even if they have the same symptoms that you have. It may harm them. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about CHANTIX that is written for healthcare professionals.For more information about CHANTIX and tips on how to quit smoking, go to www.CHANTIX.com or call 1-877-242-6849. If you are motivated to quit smoking and did not succeed during prior CHANTIX treatment for reasons other than side effects, or if you returned to smoking after treatment, speak with your healthcare provider about whether another course of CHANTIX therapy may be right for you.If you want more information about APO-VARENICLINE: - Talk to your healthcare professional
Find the full Product Monograph that is prepared for healthcare professionals and includes this Consumer Information by visiting the Health Canada website (https://health-products.canada.ca/dpd-bdpp/index-eng.jsp). Find the Consumer Information on the manufacturer’s website (http://www.apotex.ca/products) or by calling 1-800-667-4708.
What are the ingredients in CHANTIX?
Active ingredient: varenicline tartrate
Inactive ingredients: microcrystalline cellulose, anhydrous dibasic calcium phosphate, croscarmellose sodium, colloidal silicon dioxide, magnesium stearate, Opadry® White (for 0.5 mg), Opadry® Blue (for 1 mg), and Opadry® Clear.What the medicinal ingredient is:
Varenicline tartrate.
What the nonmedicinal ingredients are:
The nonmedicinal ingredients are anhydrous dibasic calcium phosphate, magnesium stearate and microcrystalline cellulose. The film-coating contains hydroxypropyl cellulose, hydroxypropyl methylcellulose, titanium dioxide and triacetin. The 1 mg tablet also contains indigotine aluminum lake 12% to 14% as a colouring agent.
This leaflet was prepared by Apotex Inc., Toronto, Ontario M9L 1T9.
Last revised: April 09, 2019This Medication Guide has been approved by the U.S. Food and Drug Administration.
Revised: Feb 2019 -
PATIENT PACKAGE INSERT
PART III: CONSUMER INFORMATION
PrAPO-VARENICLINE
Varenicline Tablets
0.5 mg and 1 mg varenicline (as varenicline tartrate)
Read this information each time you refill your prescription in case new information has been added.
This leaflet is part III of a three-part "Product Monograph" published when APO-VARENICLINE was approved for sale in Canada and is designed specifically for Consumers. This leaflet is a summary and will not tell you everything about APO-VARENICLINE. Contact your doctor or pharmacist if you have any questions about the drug.
What is the most important information I should know about APO-VARENICLINE?
When you try to quit smoking, with or without APO-VARENICLINE, you may have symptoms that may be due to nicotine withdrawal, including
- the urge to smoke
- depressed mood
- trouble sleeping
- irritability
- frustration
- anger
- feeling anxious
- difficulty concentrating
- restlessness
- decreased heart rate
- increased appetite or weight gain.
Some people have even experienced suicidal thoughts when trying to quit smoking without medication. Sometimes quitting smoking can lead to worsening of mental health problems that you already have, such as depression.
Mental Health Problems
Some people have had serious side effects while taking APO-VARENICLINE to help them quit smoking, including changes in behavior or thinking, hostility, agitation, aggression, depressed mood, or suicidal thoughts or actions. These symptoms have occurred in people with previous mental health problems, as well as in those with no previous history. For some people, these symptoms began when they started taking APO-VARENICLINE while for others, they began after several weeks of treatment, or shortly after stopping APO-VARENICLINE.
Before taking any quit-smoking treatment, including APO-VARENICLINE, tell your healthcare provider (doctor, pharmacist or nurse):
- if you have ever had depression or other mental health problems;
- about any concerning symptoms you had during other times you tried to quit smoking, with or without medication.
Inform your friends and family members of your quit attempt with APO-VARENICLINE and ask for their support and help in monitoring for any changes in behavior or thinking that are not normal.
Drinking alcohol may increase the risk of having mental health problems during your treatment with APO-VARENICLINE.
Patients with history of mental health problems (e.g. depression, anxiety, schizophrenia): If you have had mental health problems before taking APO-VARENICLINE, your healthcare provider will monitor you while you try to quit smoking with APO-VARENICLINE. If you develop worsened or new symptoms, talk to your healthcare provider right away because changing the dose (of APO-VARENICLINE or other medications) may make a difference.
All patients/General: If you have thoughts, moods or behaviours that are severe, concerning or very abnormal for you, stop taking APO-VARENICLINE right away, seek medical help, and tell your healthcare provider about your symptoms. In many people, these symptoms went away after stopping APO-VARENICLINE, but not in all. It is important for you to follow up with your healthcare provider until your symptoms go away.
Allergic Reactions Some people can have allergic reactions to APO-VARENICLINE. Some of these allergic reactions can be life-threatening and include: swelling of the face, mouth, and throat that can cause trouble breathing. If you have these symptoms, stop taking APO-VARENICLINE and seek immediate emergency medical attention.
Serious Skin Reactions Some people can have serious skin reactions while taking APO-VARENICLINE. These can include rash, swelling, redness, and peeling of the skin. Some of these reactions can become life-threatening. If you have a rash with peeling skin or blisters in your mouth, around the eyes or genitals, stop taking APO-VARENICLINE and seek immediate emergency medical attention.
ABOUT THIS MEDICATION
What the medication is used for:
APO-VARENICLINE is a prescription medicine which is used in combination with supportive counselling to help motivated adults stop smoking.
What it does:
APO-VARENICLINE can help to relieve the craving and withdrawal symptoms associated with stopping smoking.
APO-VARENICLINE does not contain nicotine, but it has been shown to affect the nicotine receptor in the brain that is thought to be most related to smoking addiction. APO-VARENICLINE can affect this receptor in two opposite ways: it acts like a weaker version of nicotine, and also blocks nicotine from getting to the receptor because it binds more tightly. Although it is thought that this may be, in part, how APO-VARENICLINE works, it is not known exactly how the drug works in people.
When it should not be used:
Do not take APO-VARENICLINE if you:
- are allergic (hypersensitive) to varenicline tartrate or any of the other ingredients of APO-VARENICLINE (see list below of non-medicinal ingredients).
- are using nicotine replacement therapy, such as patches, gum or inhaler. The combination of APO-VARENICLINE and nicotine replacement therapy is not expected to improve your chances of quitting, and may result in more side effects than with APO-VARENICLINE alone.
What dosage forms it comes in:
APO-VARENICLINE is available as film-coated tablets. The 0.5 mg tablets are white and the 1 mg tablets are blue.
WARNINGS AND PRECAUTIONS
BEFORE you use APO-VARENICLINE talk to your healthcare provider if you:
- have experienced depression or any other mental health problems. Your healthcare provider will monitor you for new or worsened emotional or behavioral problems during treatment with APO-VARENICLINE.
- have any problems with your kidneys, as you may need a lower dose of APO-VARENICLINE.
- have heart or blood vessel (cardiovascular) problems.
- have a history of seizures.
- have any other medical conditions.
- are pregnant, are breastfeeding or plan to become pregnant (see “Pregnancy” and “Breastfeeding” below).
- have diabetes. APO-VARENICLINE can potentially affect your blood sugar regulation, and you may need to monitor your blood sugar more often. If you notice changes, discuss this with your healthcare provider.
The effects of changes in your body resulting from stopping smoking, with or without treatment with APO-VARENICLINE, may alter the way other drugs work. Tell your healthcare provider about all your other medicines, including prescription and nonprescription medicines, vitamins and herbal supplements. Especially, tell your healthcare provider if you take:
- Insulin
- Asthma medicines (theophylline)
- Blood thinner (warfarin)
as an adjustment of the dose of these medicines may be necessary once you are smoke-free.
Mental Health Symptoms
See “What is the most important information I should know about APO-VARENICLINE?”
Pregnancy
Talk to your healthcare provider if you are pregnant or planning to become pregnant.
You should not take APO-VARENICLINE while you are pregnant. It is unknown if APO-VARENICLINE will harm your unborn baby.
It is best to stop smoking before you get pregnant.
Breastfeeding
You should ask your healthcare provider for advice before taking any medication, including APO-VARENICLINE, if you are breastfeeding, as the medicine may pass into breast milk.
APO-VARENICLINE is not recommended for use in children under 18 years of age.
Accidental Injury, including while Driving, Operating Machinery
Do not engage in potentially hazardous tasks, such as driving a car or operating dangerous machines, until you know how APO-VARENICLINE may affect you. In some cases, people have reported sleepiness, dizziness, blackouts, seizures or difficulty concentrating while driving.
Seizures
Tell your healthcare provider if you have experienced seizures or have epilepsy before you start APO-VARENICLINE treatment. Some people have reported seizures while taking varenicline, both with and without a history of seizures.
Heart or Stroke Events
New or worse heart or blood vessel (cardiovascular) problems have been reported in people taking varenicline, primarily in those who already have cardiovascular problems. From the information available to date, it is not possible to determine whether APO-VARENICLINE increases the risk of heart or stroke events.
Tell your healthcare provider if you have any changes in cardiovascular symptoms during treatment with APO-VARENICLINE. Get emergency medical help right away if you have symptoms of a heart attack, including any of the following:
- Chest discomfort (uncomfortable pressure, squeezing, fullness or pain) that lasts more than a few minutes, or that goes away and comes back.
- Pain or discomfort in one or both arms, back, neck, jaw or stomach.
- Shortness of breath, sweating, nausea, vomiting, or feeling lightheaded associated with chest discomfort.
Get emergency medical help right away if you have symptoms of a stroke, including any of the following:
- Weakness - Sudden loss of strength or sudden numbness in the face, arm or leg even if temporary.
- Trouble speaking - Sudden difficulty speaking or understanding or sudden confusion, even if temporary.
- Vision problems - Sudden trouble with vision, even if temporary.
- Headache - Sudden severe and unusual headache.
- Dizziness - Sudden loss of balance, especially with any of the above signs.
Sleepwalking
Sleepwalking has been reported in patients taking APO-VARENICLINE, and may sometimes lead to behaviour that is harmful to you or other people or property. Stop taking APO-VARENICLINE and tell your healthcare provider if you start sleepwalking.
INTERACTIONS WITH THIS MEDICATION
Drinking alcohol during treatment with APO-VARENICLINE may increase the risk of mental health symptoms.
Reported experiences include:
- unusual or sometimes aggressive behavior;
- more intoxicated than expected from the amount of alcohol;
- no memory of things that have happened.
Use of APO-VARENICLINE with other therapies for smoking-cessation:
The safety and benefits of taking APO-VARENICLINE in combination with other medicines for stopping smoking have not been studied. Taking APO-VARENICLINE in combination with other smoking-cessation therapies (e.g., nicotine replacement therapy) is therefore not recommended. Using APO-VARENICLINE in combination with nicotine replacement therapies (e.g., patch gum or inhaler) is not likely to increase your chances of quitting smoking, and it may result in more side effects than with APO-VARENICLINE alone.
PROPER USE OF THIS MEDICATION
You are more likely to stop smoking if you are motivated to stop. Your healthcare provider can provide advice, support and sources of further information to help ensure your attempt to stop smoking is successful.
To increase the chances of success, APO-VARENICLINE should be used in combination with supportive counselling as recommended by your healthcare provider. Varenicline was used in combination with supportive counselling in the clinical trials. At any time, you can also call government-funded toll-free provincial Quit Lines, to speak to a knowledgeable and supportive specialist; these phone numbers are available on the Health Canada website.
Always take APO-VARENICLINE exactly as your healthcare provider has told you. You should check with your healthcare provider if you are not sure.
REMEMBER: This medication has been prescribed specifically for you. Do not give it to anyone else.
Setting Your Quit Date: Starting treatment before your quit date lets APO-VARENICLINE build up in your body. You can keep smoking until your quit date.
There are three ways to set your quit date when using APO-VARENICLINE. Talk to your healthcare provider about which way is best for you:
- Fixed quit approach: Set a quit date when you will stop smoking. Start taking APO-VARENICLINE 8 to 14 days (1 to 2 weeks) before your quit date. You should take APO-VARENICLINE for 12 weeks. After 12 weeks of treatment, your healthcare provider may recommend an additional 12 weeks of treatment.
Or
- Flexible quit approach: Start taking APO-VARENICLINE, then quit smoking between Day 8 and Day 35 after the start of your treatment (i.e. between Weeks 2 and 5). You should take APO-VARENICLINE for 12 weeks. After 12 weeks of treatment, your healthcare provider may recommend an additional 12 weeks of treatment.
Or
- Gradual quit approach: Start taking APO-VARENICLINE and reduce smoking with a goal to quit smoking by end of 12 weeks of treatment. For example, reduce smoking by half by the 4th week, another half by the 8th week (down to 25%) and then quit by the end of the 12th week. You may quit any time before the end of 12 weeks of treatment, if you are able to. Continue treatment for an additional 12 weeks for a total of 24 weeks.
Write down, and keep in a visible or convenient place (for example on the fridge or on the APO-VARENICLINE pack), the date that you started APO- VARENICLINE, your quit date, and the date to stop taking APO-VARENICLINE.
Make sure that you try to stop smoking on your quit date. If you slip-up and smoke after that target date, keep trying. Some people need a few weeks on APO-VARENICLINE for it to work best.
Dosing Options:
APO-VARENICLINE should be taken after eating and with a full glass of water.
Regardless of which dose is prescribed, the first week on APO-VARENICLINE is the same, and is described in the following table:
Week 1 Dosing Schedule:
Day Dose Day 1 to 3 Take one white APO-VARENICLINE 0.5 mg tablet once a day. Day 4 to 7 Take one white APO-VARENICLINE 0.5 mg tablet twice a day, once in the morning and once in the evening, at about the same time each day. After the first week, your healthcare provider may recommend to stay at 0.5 mg twice a day (OPTION 1) or go up to 1 mg twice a day (OPTION 2).
Week 2 (day 8) to the end of treatment
OPTION 1: Continue on 0.5 mg twice a day
Day Dose Day 8 - end of treatment 0.5 mg twice a day: Continue to take one white APO-VARENICLINE 0.5 mg pill in the morning, and one in the evening, at about the same time each day Or
OPTION 2: Start taking 1 mg twice a day
Day Dose Day 8 - end of treatment 1 mg twice a day: Take one light blue APO-VARENICLINE 1 mg pill in the morning, and one in the evening, at about the same time each day. The maximum dose of APO-VARENICLINE is 1 mg twice a day.
Based on the limited data available, the two doses do not appear different in terms of either quit rates, or rates of serious mental health side effects (your healthcare provider can provide more information).
Discussion with your healthcare provider is important in order to choose the dose that is best for you.
If needed, the dose can be changed depending on how well you tolerate APO-VARENICLINE and how effective your healthcare provider and you consider it is in helping you quit smoking. Your healthcare provider will help decide what dose is right for you.
Your healthcare provider may recommend to gradually lower the dose at the end of the treatment period rather than stopping abruptly.
Can I smoke while taking APO-VARENICLINE?
You can keep smoking prior to your quit date.
Smoking after your quit date will reduce your chance of breaking your smoking addiction.
Some people have reported a change in the taste of cigarettes after starting APO-VARENICLINE.
Overdose:
If you think you have taken too much APO-VARENICLINE, contact your healthcare professional, hospital emergency department or regional Poison Control Centre immediately, even if there are no symptoms. Missed Dose:
Do not take a double dose to make up for a forgotten tablet. It is important that you take APO-VARENICLINE regularly at the same time each day. If you forget to take a dose, take it as soon as you remember, as long as it is within a few hours of the missed dose. If it has been longer than a few hours since the missed dose, or if you do not remember whether you took a dose or not, then skip that dose, and wait to take the next dose at the correct time.
If you have any further questions on the use of this product, ask your healthcare provider.
SIDE EFFECTS AND WHAT TO DO ABOUT THEM
Whether you are taking medication to stop smoking or not, the following are symptoms you may feel: depressed, short-tempered, frustrated or angry, nervous, impatient; have difficulty concentrating.
Your appetite may increase, and you may gain some weight.
Like all medicines, APO-VARENICLINE can cause side effects, although not everybody gets them.
The common side effects are mostly mild to moderate and these usually occur in the first weeks of treatment. Some of the most common side effects you should be aware of include:
- Nausea, vomiting
- Trouble sleeping
- Headache
- Abnormal dreams (vivid, unusual, or increased dreaming; rarely may include nightmares)
- Sleepiness, tiredness, dizziness
- Constipation, diarrhea, gas
Mental Health Problems See “What is the most important information I should know about APO-VARENICLINE?”
Stop taking APO-VARENICLINE if you experience severe or unusual feelings of agitation, aggression, depressed mood, hostility, hallucinations, or if you have thoughts of self-harm or harm to others. Tell your healthcare provider about your symptoms.
Allergic Reactions
Some people have allergic reactions to APO-VARENICLINE. Some of these allergic reactions can be life-threatening and include: swelling of the face, mouth (lips, gums, tongue), and throat can cause trouble breathing. If you have these symptoms, stop taking APO-VARENICLINE and seek immediate emergency medical attention.
Serious Skin Reactions
Some people can have serious skin reactions while taking APO-VARENICLINE. These can include rash, swelling, redness, and peeling of the skin. Some of these reactions can become life-threatening. If you have a rash with peeling skin, or blistering of the mouth, around the eyes or genitals, stop taking APO-VARENICLINE and seek immediate emergency medical attention.
SERIOUS SIDE EFFECTS, HOW OFTEN THEY HAPPEN AND WHAT TO DO ABOUT THEM Symptom/effect Talk to your healthcare professional Stop taking drug and get immediate medical help Only if severe In all cases Rare Allergic reaction redness, itching or swelling of your skin, hives, burning, stinging, swelling of the neck area, or any difficulty with breathing, not present before using this medicine √ Rare Serious skin reactions peeling of the skin, or rash combined with blisters around the mouth, eyes or genitals. √ Rare Mental Health Problems √ √
(if severe, or if involves potential for harm to self or others)Unknown Heart attack: chest pain often associated with left shoulder or jaw pain, feeling of constriction around chest and sweating √ Unknown Stroke: weakness and/or loss of sensation of limbs or face, difficulty speaking, clumsiness, visual loss √ Unknown Seizures: Loss of consciousness with uncontrollable shaking (convulsion) √ Unknown Sleepwalking √
(and stop taking APO-VARENICLINE)This is not a complete list of side effects. For any unexpected effects while taking APO-VARENICLINE, contact your doctor or pharmacist.
HOW TO STORE IT
Store APO-VARENICLINE at room temperature (15°C to 30°C).
Keep out of the reach and sight of children.
Reporting Side Effects You can report any suspected side effects associated with the use of health products to Health Canada by: • Visiting the Web page on Adverse Reaction Reporting (https://www.canada.ca/en/health-canada/services/drugs-health-products/medeffect-canada/adverse-reaction-reporting.html) for information on how to report online, by mail or by fax; or • Calling toll-free at 1-866-234-2345. NOTE: Contact your health professional if you need information about how to manage your side effects. The Canada Vigilance Program does not provide medical advice. MORE INFORMATION
If you want more information about APO-VARENICLINE:
- Talk to your healthcare professional
- Find the full Product Monograph that is prepared for healthcare professionals and includes this Consumer Information by visiting the Health Canada website (https://health-products.canada.ca/dpd-bdpp/index-eng.jsp). Find the Consumer Information on the manufacturer’s website (http://www.apotex.ca/products) or by calling 1-800-667-4708.
This leaflet was prepared by Apotex Inc., Toronto, Ontario
M9L 1T9.
Last revised: April 09, 2019
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
APO-VARENICLINE
varenicline kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:60505-4767 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60505-4767-0 1 in 1 CARTON; Type 0: Not a Combination Product 07/16/2021 01/23/2023 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BLISTER PACK 11 Part 2 1 BLISTER PACK 42 Part 1 of 2 APO-VARENICLINE
apo-varenicline tablet, film coatedProduct Information Item Code (Source) NDC:60505-8282 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VARENICLINE TARTRATE (UNII: 82269ASB48) (VARENICLINE - UNII:W6HS99O8ZO) VARENICLINE 0.5 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) MAGNESIUM STEARATE (UNII: 70097M6I30) HYPROMELLOSE 2910 (5 MPA.S) (UNII: R75537T0T4) HYDROXYPROPYL CELLULOSE (110000 WAMW) (UNII: 5Y0974F5PW) TRIACETIN (UNII: XHX3C3X673) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE Score no score Shape OVAL (Capsule-Shaped) Size 8mm Flavor Imprint Code APO;VAR;0;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60505-8282-3 11 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug for use in drug shortage 07/16/2021 01/23/2023 Part 2 of 2 APO-VARENICLINE
apo-varenicline tablet, film coatedProduct Information Item Code (Source) NDC:60505-3283 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VARENICLINE TARTRATE (UNII: 82269ASB48) (VARENICLINE - UNII:W6HS99O8ZO) VARENICLINE 1 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) MAGNESIUM STEARATE (UNII: 70097M6I30) HYPROMELLOSE 2910 (5 MPA.S) (UNII: R75537T0T4) HYDROXYPROPYL CELLULOSE (110000 WAMW) (UNII: 5Y0974F5PW) TRIACETIN (UNII: XHX3C3X673) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) Product Characteristics Color BLUE Score no score Shape OVAL (Capsule-Shaped) Size 10mm Flavor Imprint Code APO;VAR;1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60505-3283-6 42 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug for use in drug shortage 07/16/2021 01/23/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug for use in drug shortage 07/16/2021 01/23/2023 APO-VARENICLINE
varenicline tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:60505-4765 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VARENICLINE TARTRATE (UNII: 82269ASB48) (VARENICLINE - UNII:W6HS99O8ZO) VARENICLINE 0.5 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) MAGNESIUM STEARATE (UNII: 70097M6I30) HYPROMELLOSE 2910 (5 MPA.S) (UNII: R75537T0T4) HYDROXYPROPYL CELLULOSE (110000 WAMW) (UNII: 5Y0974F5PW) TRIACETIN (UNII: XHX3C3X673) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color WHITE Score no score Shape OVAL (Capsule-Shaped) Size 8mm Flavor Imprint Code APO;VAR;0;5 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60505-4765-5 56 in 1 BOTTLE; Type 0: Not a Combination Product 07/16/2021 03/31/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug for use in drug shortage 07/16/2021 03/31/2025 APO-VARENICLINE
varenicline tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:60505-4766 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength VARENICLINE TARTRATE (UNII: 82269ASB48) (VARENICLINE - UNII:W6HS99O8ZO) VARENICLINE 1 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) ANHYDROUS DIBASIC CALCIUM PHOSPHATE (UNII: L11K75P92J) MAGNESIUM STEARATE (UNII: 70097M6I30) HYPROMELLOSE 2910 (5 MPA.S) (UNII: R75537T0T4) HYDROXYPROPYL CELLULOSE (110000 WAMW) (UNII: 5Y0974F5PW) TRIACETIN (UNII: XHX3C3X673) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) FD&C BLUE NO. 2 (UNII: L06K8R7DQK) Product Characteristics Color BLUE Score no score Shape OVAL (Capsule-Shaped) Size 10mm Flavor Imprint Code APO;VAR;1 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:60505-4766-6 56 in 1 BOTTLE; Type 0: Not a Combination Product 07/16/2021 07/31/2024 2 NDC:60505-4766-5 1 in 1 CARTON 01/20/2023 01/23/2023 2 56 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug for use in drug shortage 07/16/2021 07/31/2024 Labeler - Apotex Corp (845263701) Registrant - Apotex Inc (209429182)




