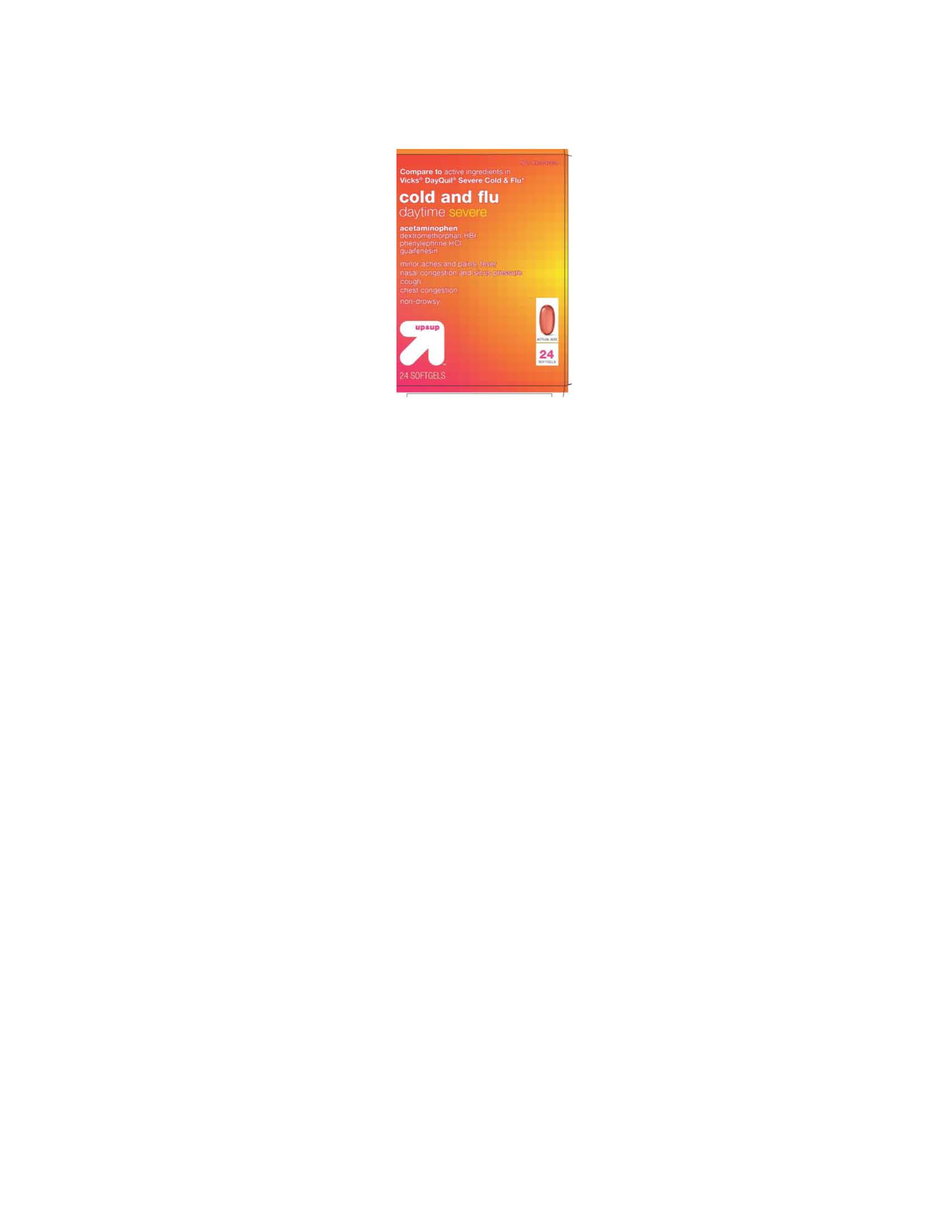
Label: COLD AND FLU DAYTIME SEVERE- acetaminophen, dextromethorphan hbr, guaifenesin, phenylephrine hcl capsule, liquid filled
- NDC Code(s): 11673-972-16, 11673-972-24
- Packager: TARGET CORP
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 15, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- PURPOSE
-
INDICATIONS & USAGE
Uses
temporarily relieves common cold/flu symptoms: nasal congestion - sinus congestion & pressure - cough due to minor throat & bronchial irritation - minor aches & pains - headache - fever - sore throat - reduces ...temporarily relieves common cold/flu symptoms:
nasal congestion
sinus congestion & pressure
cough due to minor throat & bronchial irritation
minor aches & pains
headache
fever
sore throat
reduces swelling of nasal passages
temporarily restores freer breathing through the nose
promotes nasal and/or sinus drainage
helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive -
DOSAGE & ADMINISTRATION
Directions
take only as directed - do not exceed 8 LiquiCaps per 24 hrs - adults & children 12 yrs & over 2 LiquiCaps with water every 4 hrs - children 4 to under 12 yrs ask a doctor ...take only as directed
do not exceed 8 softgels per 24 hrsadults & children 12 yrs & over 2 softgelswith water every 4 hrs
children 4 to under 12 yrs ask a doctor
children under 4 yrs do not use -
WARNINGS
Warnings
Liver warning - This product contains acetaminophen. Severe liver damage may occur if you take - more than 8 softgels in 24 hrs, which is the maximum daily amount for this product - with other ...Liver warning
This product contains acetaminophen.
Severe liver damage may occur if you take
more than 8 softgels in 24 hrs, which is the maximum daily amount for this product
with other drugs containing acetaminophen
3 or more alcoholic drinks every day while using this productAllergy Alert:
Acetaminophen may cause severe skin reactions. Symptoms may include:
Skin reddening
Blisters
RashIf a skin reaction occurs, stop use and seek medical help right away
Sore throat warning:
If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
- KEEP OUT OF REACH OF CHILDREN
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
COLD AND FLU DAYTIME SEVERE
acetaminophen, dextromethorphan hbr, guaifenesin, phenylephrine hcl capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:11673-972 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 200 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) FD&C RED NO. 40 (UNII: WZB9127XOA) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SORBITOL (UNII: 506T60A25R) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) POVIDONE (UNII: FZ989GH94E) SHELLAC (UNII: 46N107B71O) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) GELATIN (UNII: 2G86QN327L) Product Characteristics Color orange Score no score Shape BULLET Size 16mm Flavor Imprint Code 73 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:11673-972-16 16 in 1 BLISTER PACK; Type 0: Not a Combination Product 05/29/2020 2 NDC:11673-972-24 24 in 1 BLISTER PACK; Type 0: Not a Combination Product 05/29/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 05/18/2020 Labeler - TARGET CORP (006961700) Registrant - TIME CAP LABS,INC (037052099) Establishment Name Address ID/FEI Business Operations MARKSANS PHARMA LTD 925822975 manufacture(11673-972)