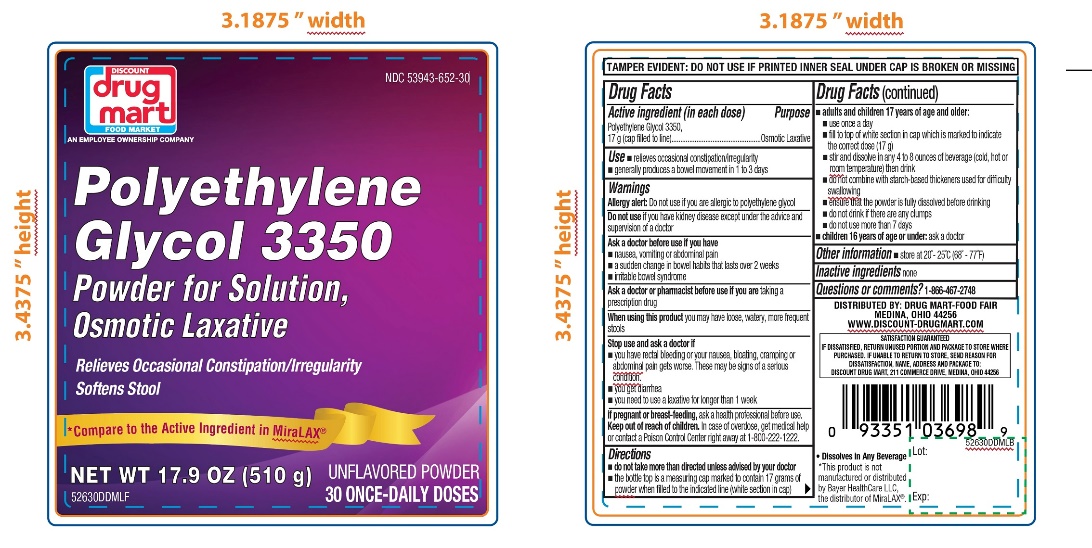
Label: DISCOUNT DRUG MART- polyethylene glycol 3350 powder, for solution
- NDC Code(s): 53943-652-14, 53943-652-30
- Packager: DISCOUNT DRUG MART
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated August 31, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each dose)
- Purpose
- Use
-
Warnings
Ask a doctor before use if you have
- ▪
- nausea, vomiting or abdominal pain
- ▪
- a sudden change in bowel habits that lasts over 2 weeks
- ▪
- irritable bowel syndrome
-
Directions
- ▪
- do not take more than directed unless advised by your doctor
- ▪
- the bottle top is a measuring cap marked to contain 17 grams of powder when filled to the indicated line (white section in cap)
- ▪
-
adults and children 17 years of age and older:
- ▪
- use once a day
- ▪
- fill to top of white section in cap which is marked to indicate the correct dose (17 g)
- ▪
- stir and dissolve in any 4 to 8 ounces of beverage (cold, hot or room temperature) then drink
- ▪
- do not combine with starch-based thickeners used for difficulty swallowing
- ▪
- ensure that the powder is fully dissolved before drinking
- ▪
- do not drink if there are any clumps
- ▪
- do not use no more than 7 days
- ▪
- children 16 years of age or under: ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
PRINCIPAL DISPLAY PANEL
DISCOUNT DRUG MART
NDC 53943-652-14
Polyethylene Glycol 3350,
Powder for Solution,
Osmotic Laxative
- •
- Relieves Occasional Constipation/Irregularity
- •
- Softens Stool
UNFLAVORED POWDER
14 ONCE-DAILY DOSESNET WT 8.3 OZ (238 g)
- •
- Dissolves in ANY Beverage
DISTRIBUTED BY: DRUG MART-FOOD FAIR
MEDINA, OHIO 44256
SATISFACTION GUARANTEED
IF DISSATISFIED, RETURN UNUSED PORTION AND PACKAGE TO THE STORE WHERE PURCHASED. IF UNABLE TO RETURN TO STORE, SEND REASON FOR DISSATIFACTION, NAME, ADRESS AND PACKAGE TO:
DISCOUNT DRUG MART, 211 COMMERCE DRIVE, MEDINA, OHIO 044256
*This product is not manufactured or distributed by Bayer HealthCare LLC, the distributer of MiraLAX®
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
DISCOUNT DRUG MART
polyethylene glycol 3350 powder, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53943-652 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POLYETHYLENE GLYCOL 3350 (UNII: G2M7P15E5P) (POLYETHYLENE GLYCOL 3350 - UNII:G2M7P15E5P) POLYETHYLENE GLYCOL 3350 17 g in 17 g Product Characteristics Color white (Colorless upon dissolution) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53943-652-30 510 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/17/2023 2 NDC:53943-652-14 238 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 04/17/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022015 04/17/2023 Labeler - DISCOUNT DRUG MART (047741335)