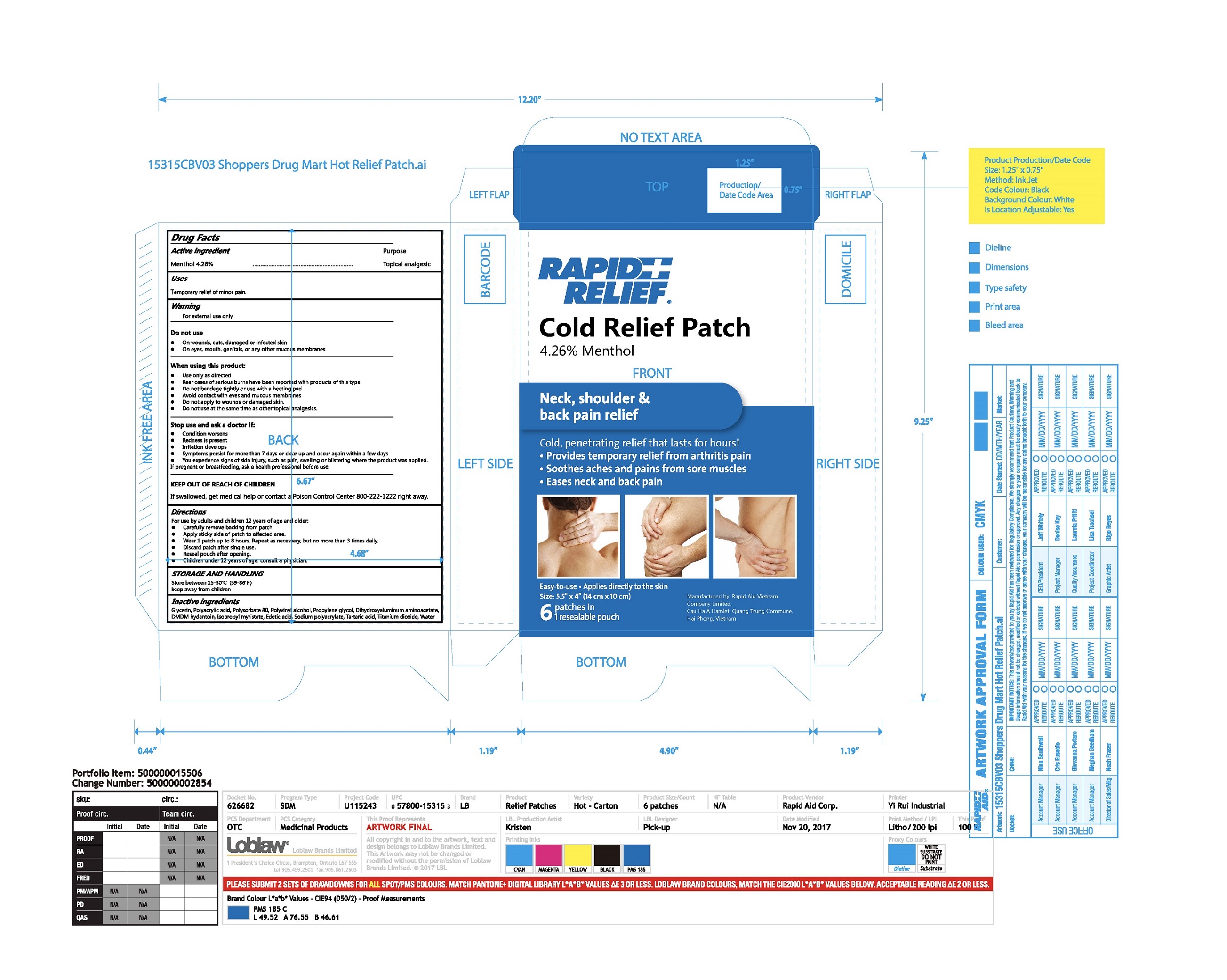
Label: RAPID RELIEF COLD RELIEF PATCH- menthol patch
- NDC Code(s): 83569-001-01
- Packager: RAPID AID VIET NAM CO., LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 6, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient(s)
- Purpose
- Use
- Warnings
- Do not use
- When using this product:
-
Stop use and ask a doctor if:
Condition worsens
Redness is present
Irritation develops
Symptoms persist for more than 7 days or clear up and occur again within a few days
You experience signs of skin injury, such as pain, swelling or blistering where the product was applied.
If pregnant or breastfeeding, ask a health professional before use. - KEEP OUT OF REACH OF CHILDREN
-
Directions
For use by adults and children 12 years of age and older:
Carefully remove backing from patch
Apply sticky side of patch to affected area.
Wear 1 patch up to 8 hours. Repeat as necessary, but no more than 3 times daily.
Discard patch after single use.
Reseal pouch after opening.
Children under 12 years of age: consult a physician. - STORAGE AND HANDLING
- Inactive ingredients
- Package Label - Principal Display Panel
-
INGREDIENTS AND APPEARANCE
RAPID RELIEF COLD RELIEF PATCH
menthol patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83569-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Menthol (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) Menthol 4.26 g in 100 g Inactive Ingredients Ingredient Name Strength POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYACRYLIC ACID (450000 MW) (UNII: KD3S7H73D3) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) Propylene glycol (UNII: 6DC9Q167V3) Glycerin (UNII: PDC6A3C0OX) Water (UNII: 059QF0KO0R) DMDM hydantoin (UNII: BYR0546TOW) Edetic acid (UNII: 9G34HU7RV0) SODIUM POLYACRYLATE (8000 MW) (UNII: 285CYO341L) Dihydroxyaluminum aminoacetate (UNII: DO250MG0W6) Isopropyl myristate (UNII: 0RE8K4LNJS) Tartaric acid (UNII: W4888I119H) Titanium dioxide (UNII: 15FIX9V2JP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83569-001-01 1 in 1 BOX 03/06/2024 1 6 in 1 POUCH 1 4.62 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 03/06/2024 Labeler - RAPID AID VIET NAM CO., LTD (673067008) Registrant - RAPID AID VIET NAM CO., LTD (673067008) Establishment Name Address ID/FEI Business Operations RAPID AID VIET NAM CO., LTD 673067008 manufacture(83569-001)