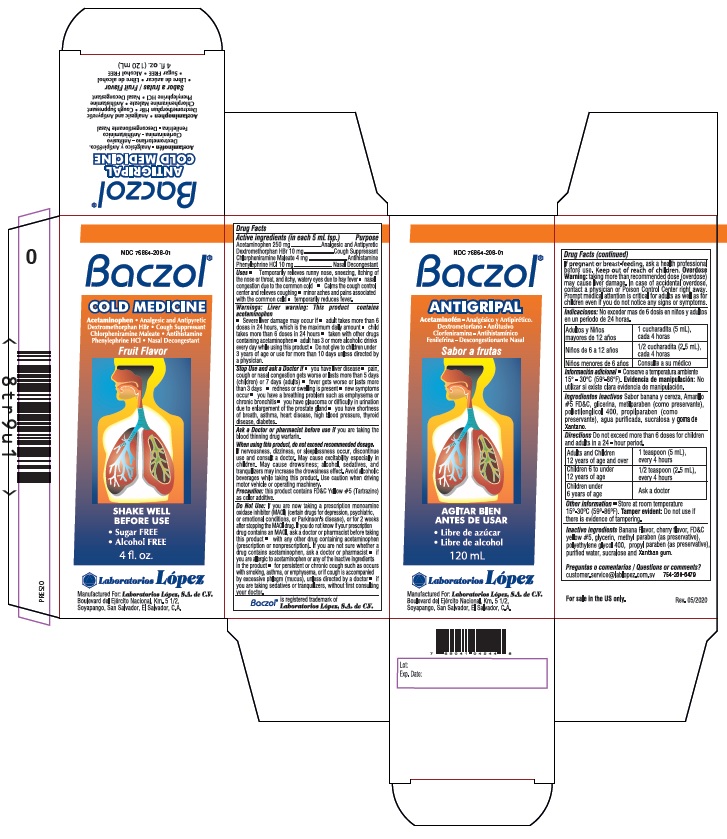
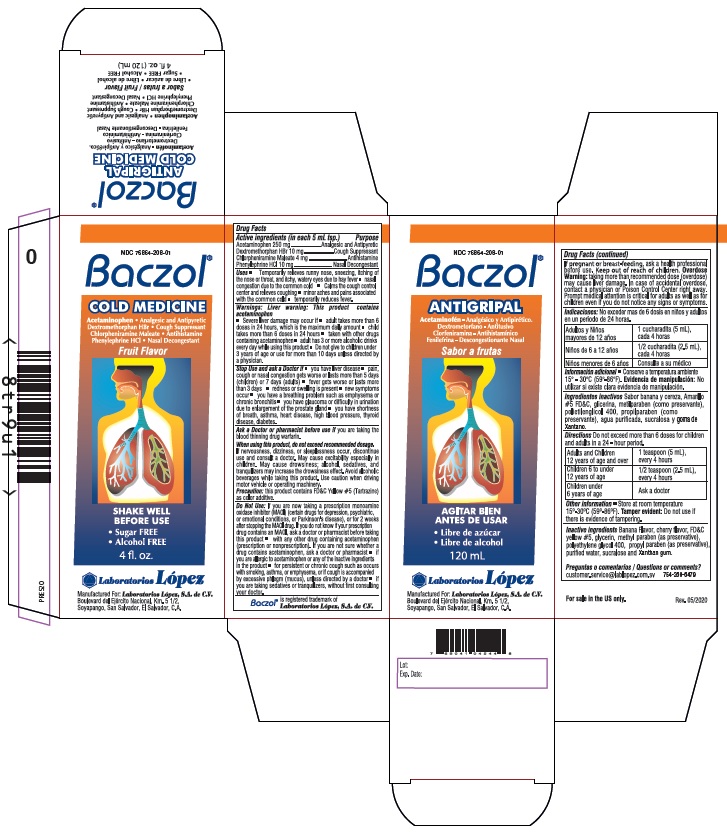
Label: BACZOL COLD MEDICINE- acetaminophen, dextromethorphan hbr, chlorpheniramine maleate, phenylephrine hcl syrup
- NDC Code(s): 76864-208-01
- Packager: Procaps S.A. de C.V.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated August 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
-
INDICATIONS & USAGE
Uses • Temporarily relieves runny nose, sneezing, itching of
the nose or throat, and itchy, watery eyes due to hay fever • nasal
congestion due to the common cold • Calms the cough control
center and relieves coughing • minor aches and pains associated
with the common cold • temporarily reduces fever. -
WARNINGS
Warnings: Liver warning: This product contains
acetaminophen
• Severe liver damage may occur if • adult takes more than 6
doses in 24 hours, which is the maximum daily amount •child
takes more than 6 doses in 24 hours • taken with other drugs
containing acetaminophen• adult has 3 or more alcoholic drinks
every day while using this product • Do not give to children
under 3 years of age or use for more than 10 days unless directed by
a physician. -
SPL UNCLASSIFIED SECTION
Stop Use and ask a Doctor if • you have liver disease • pain, cough or nasal congestion gets
worse or lasts more than 5 days (children) or 7 days (adults) • fever gets worse or lasts more
than 3 days • redness or swelling is present • new symptoms occur • you have a breathing
problem such as emphysema or chronic bronchitis • you have glaucoma or difficulty in urination
due to enlargement of the prostate gland • you have shortness of breath, asthma, heart disease,
high blood pressure, thyroid disease, diabetes. - ASK DOCTOR/PHARMACIST
-
WHEN USING
When using this product, do not exceed recommended dosage. If nervousness, dizziness,
or sleeplessness occur, discontinue use and consult a doctor. May cause excitability especially
in children. May cause drowsiness; alcohol, sedatives, and tranquilizers may increase the
drowsiness effect. Avoid alcoholic beverages while taking this product. Use caution when driving
motor vehicle or operating machinery. - PRECAUTIONS
-
DO NOT USE
Do Not Use:If you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain
drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2
weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an
MAOI, ask a doctor or pharmacist before taking this product • with any other drug containing
acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains
acetaminophen, ask a doctor or pharmacist • if you are allergic to acetaminophen or any of the
inactive ingredients in the product • for persistent or chronic cough such as occurs with smoking,
asthma, or emphysema, or if cough is accompanied by excessive phlegm (mucus), unless
directed by a doctor • if you are taking sedatives or tranquilizers, without first consulting your
doctor. - PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- INACTIVE INGREDIENT
- QUESTIONS
-
Product Labeling
NDC 76864-208-01
Baczol ®
COLD MEDICINEAcetaminophen • Analgesic and Antipyretic
Dextromethorphan HBr • Cough Suppressant
Chlorpheniramine Maleate • Antihistamine
Phenylephrine HCl • Nasal DecongestantFruit Flavor
For sale in the
US only.SHAKE WELL
BEFORE USE• Sugar FREE • Alcohol FREE
4 fl.oz. (120 mL)
Manufactured For: Laboratorios López, S.A. de C.V.
Boulevard del Ejército Nacional, Km. 5 1/2,
Soyapango, San Salvador, El Salvador, C.A.Baczol ® is registered trademark of Laboratorios López, S.A. de C.V.
7 69041 04544 8
Lot: Rev. 05/2020
Exp. Date:

res
-
INGREDIENTS AND APPEARANCE
BACZOL COLD MEDICINE
acetaminophen, dextromethorphan hbr, chlorpheniramine maleate, phenylephrine hcl syrupProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76864-208 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 250 mg in 5 mL DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg in 5 mL CHLORPHENIRAMINE MALEATE (UNII: V1Q0O9OJ9Z) (CHLORPHENIRAMINE - UNII:3U6IO1965U) CHLORPHENIRAMINE MALEATE 4 mg in 5 mL PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 10 mg in 5 mL Inactive Ingredients Ingredient Name Strength FD&C YELLOW NO. 5 (UNII: I753WB2F1M) GLYCERIN (UNII: PDC6A3C0OX) METHYLPARABEN (UNII: A2I8C7HI9T) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color yellow Score Shape Size Flavor BANANA (BANANA, CHERRY) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76864-208-01 1 in 1 CARTON 05/25/2020 1 120 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part341 05/25/2020 Labeler - Procaps S.A. de C.V. (851259341)