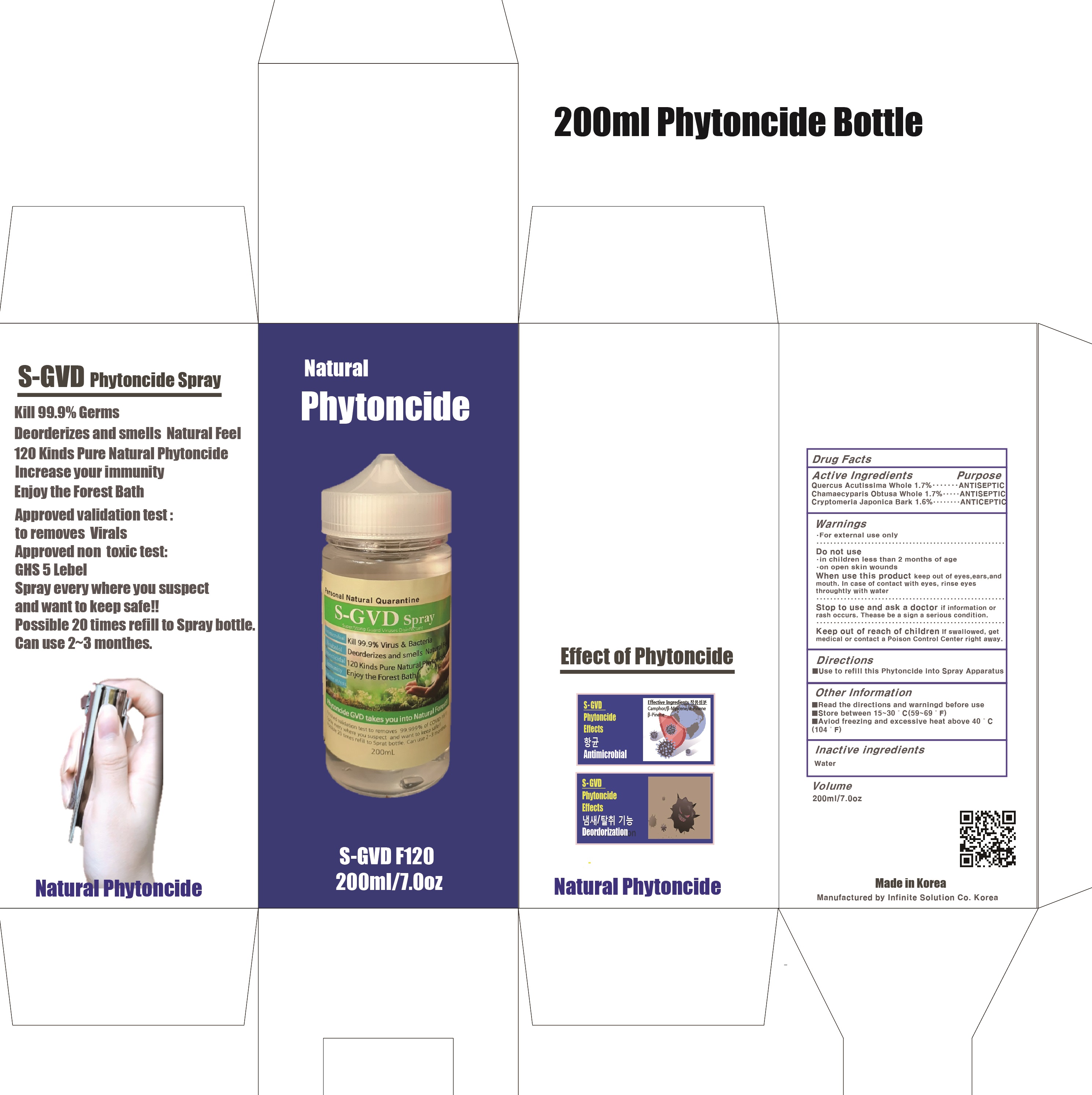
Label: S GVD PHYTONCIDE- quercus acutissima whole, chamaecyparis obtusa whole, cryptomeria japonica bark liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 81744-010-01, 81744-010-02, 81744-010-03, 81744-010-04 - Packager: Infinite Solution Co.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated April 1, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
- INACTIVE INGREDIENT
- PURPOSE
-
WARNINGS
• For external use only
--------------------------------------------------------------------------------------------------------
Do not use
• in children less than 2 months of age
• on open skin wounds
--------------------------------------------------------------------------------------------------------
When using this product keep out of eyes, ears, and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
--------------------------------------------------------------------------------------------------------
Stop use and ask a doctor if irritation or rash occurs. These may be signs of a serious condition. - KEEP OUT OF REACH OF CHILDREN
- Uses
- Directions
- Other Information
- Package Label81744-010-02: S-GVD Phytoncide (200mL, Bottle)
- Package Label81744-010-04: S-GVD Phytoncide (500mL, Bottle)
-
INGREDIENTS AND APPEARANCE
S GVD PHYTONCIDE
quercus acutissima whole, chamaecyparis obtusa whole, cryptomeria japonica bark liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:81744-010 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength QUERCUS ACUTISSIMA WHOLE (UNII: 0TVP714IPW) (QUERCUS ACUTISSIMA WHOLE - UNII:0TVP714IPW) QUERCUS ACUTISSIMA WHOLE 1.7 g in 100 mL CHAMAECYPARIS OBTUSA WHOLE (UNII: 80ZNL1G5XY) (CHAMAECYPARIS OBTUSA WHOLE - UNII:80ZNL1G5XY) CHAMAECYPARIS OBTUSA WHOLE 1.7 g in 100 mL CRYPTOMERIA JAPONICA BARK (UNII: 5VB6Q5926T) (CRYPTOMERIA JAPONICA BARK - UNII:5VB6Q5926T) CRYPTOMERIA JAPONICA BARK 1.6 g in 100 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:81744-010-02 1 in 1 CARTON 03/01/2021 1 NDC:81744-010-01 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:81744-010-04 1 in 1 CARTON 03/01/2021 2 NDC:81744-010-03 500 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 03/01/2021 Labeler - Infinite Solution Co. (695502472) Registrant - Infinite Solution Co. (695502472) Establishment Name Address ID/FEI Business Operations Infinite Solution Co. 695502472 manufacture(81744-010)