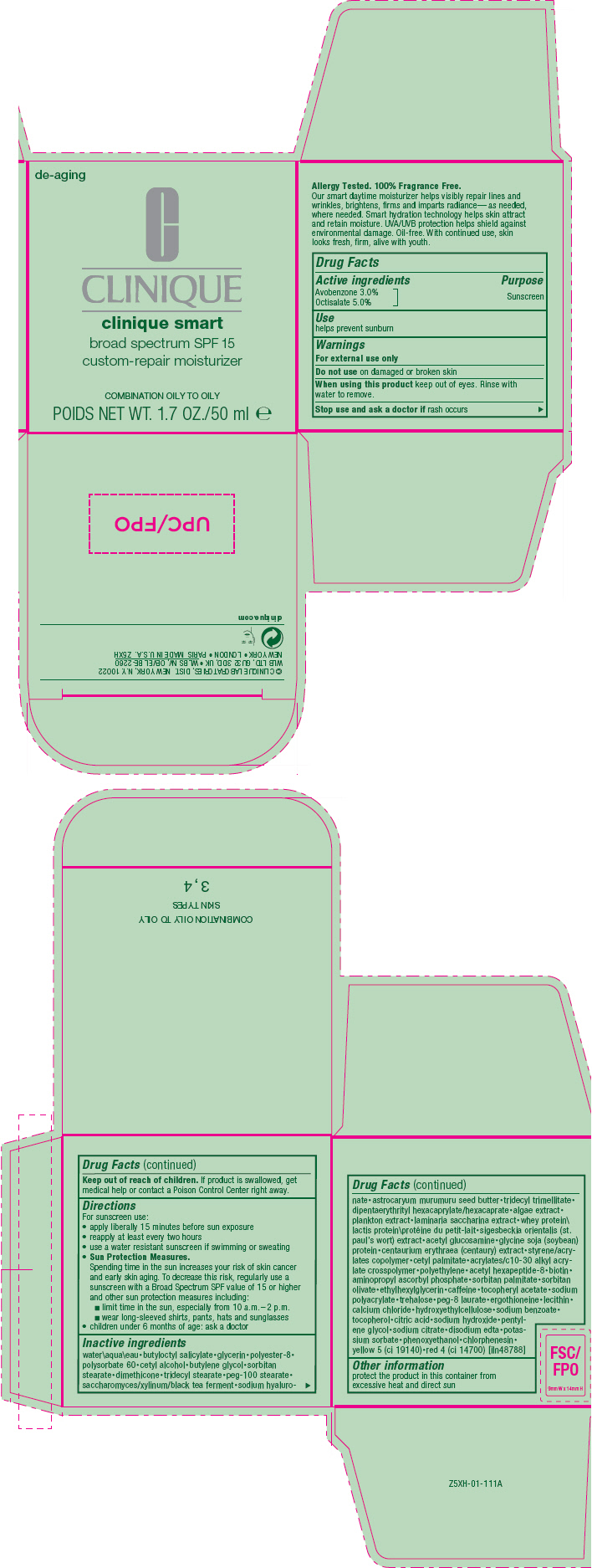
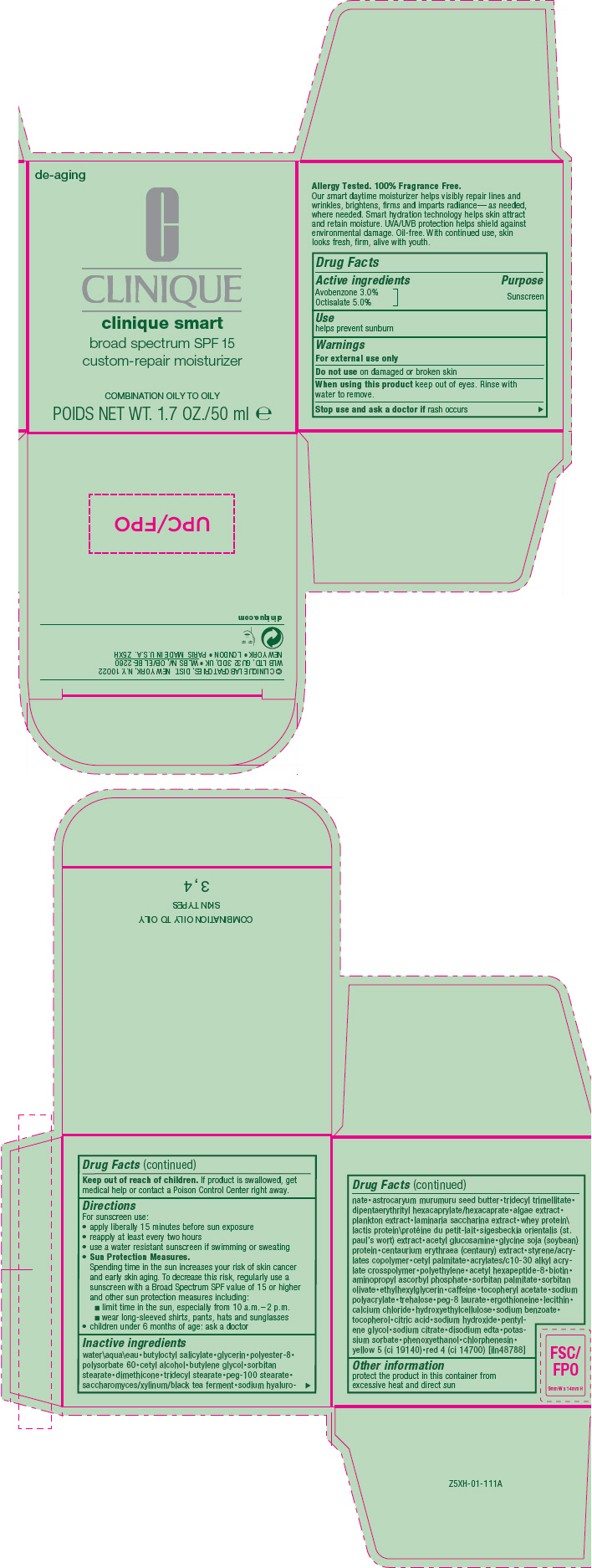
Label: CLINIQUE SMART CUSTOM REPAIR MOISTURIZER BROAD SPECTRUM SPF 15 - COMBINATION/OILY TO OILY- avobenzone and octisalate cream
- NDC Code(s): 49527-050-01
- Packager: CLINIQUE LABORATORIES LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 5, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Use
- Warnings
-
Directions
For sunscreen use:
- apply liberally 15 minutes before sun exposure
- reapply at least every two hours
- use a water resistant sunscreen if swimming or sweating
-
Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:- limit time in the sun, especially from 10 a.m.–2 p.m.
- wear long-sleeved shirts, pants, hats and sunglasses
- children under 6 months of age: ask a doctor
-
Inactive ingredients
water\aqua\eau•butyloctyl salicylate•glycerin•polyester-8• polysorbate 60•cetyl alcohol•butylene glycol•sorbitan stearate•dimethicone•tridecyl stearate•peg-100 stearate• saccharomyces/xylinum/black tea ferment•sodium hyaluro-nate•astrocaryum murumuru seed butter•tridecyl trimellitate• dipentaerythrityl hexacaprylate/hexacaprate•algae extract• plankton extract•laminaria saccharina extract•whey protein\lactis protein\protéine du petit-lait•sigesbeckia orientalis (st. paul's wort) extract•acetyl glucosamine•glycine soja (soybean) protein•centaurium erythraea (centaury) extract•styrene/acrylates copolymer•cetyl palmitate•acrylates/c10-30 alkyl acrylate crosspolymer•polyethylene•acetyl hexapeptide-8•biotin• aminopropyl ascorbyl phosphate•sorbitan palmitate•sorbitan olivate•ethylhexylglycerin•caffeine•tocopheryl acetate•sodium polyacrylate•trehalose•peg-8 laurate•ergothioneine•lecithin• calcium chloride•hydroxyethylcellulose•sodium benzoate• tocopherol•citric acid•sodium hydroxide•pentylene glycol•sodium citrate•disodium edta•potassium sorbate•phenoxyethanol•chlorphenesin• yellow 5 (ci 19140)•red 4 (ci 14700) [iln48788]
- Other information
- PRINCIPAL DISPLAY PANEL - 50 ml Jar Carton
-
INGREDIENTS AND APPEARANCE
CLINIQUE SMART CUSTOM REPAIR MOISTURIZER BROAD SPECTRUM SPF 15 - COMBINATION/OILY TO OILY
avobenzone and octisalate creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49527-050 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL Inactive Ingredients Ingredient Name Strength POLYSORBATE 60 (UNII: CAL22UVI4M) GLYCERIN (UNII: PDC6A3C0OX) CETYL ALCOHOL (UNII: 936JST6JCN) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) SORBITAN MONOSTEARATE (UNII: NVZ4I0H58X) DIMETHICONE (UNII: 92RU3N3Y1O) TRIDECYL STEARATE (UNII: A8OE252M6L) PEG-100 STEARATE (UNII: YD01N1999R) HYALURONATE SODIUM (UNII: YSE9PPT4TH) ASTROCARYUM MURUMURU SEED BUTTER (UNII: 12V64UPU6R) TRIDECYL TRIMELLITATE (UNII: FY36J270ES) DIPENTAERYTHRITYL HEXACAPRYLATE/HEXACAPRATE (UNII: 554N82UWVW) AGAR, UNSPECIFIED (UNII: 89T13OHQ2B) TAMARIND SEED (UNII: 6AHP8A7OML) N-ACETYLGLUCOSAMINE (UNII: V956696549) SOY PROTEIN (UNII: R44IWB3RN5) CENTAURIUM ERYTHRAEA (UNII: 57X4TSH58S) CETYL PALMITATE (UNII: 5ZA2S6B08X) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) ACETYL HEXAPEPTIDE-8 (UNII: L4EL31FWIL) BIOTIN (UNII: 6SO6U10H04) AMINOPROPYL ASCORBYL PHOSPHATE (UNII: 290O2PQ83R) SORBITAN MONOPALMITATE (UNII: 77K6Z421KU) SORBITAN OLIVATE (UNII: MDL271E3GR) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) CAFFEINE (UNII: 3G6A5W338E) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) TREHALOSE (UNII: B8WCK70T7I) ERGOTHIONEINE (UNII: BDZ3DQM98W) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM HYDROXIDE (UNII: 55X04QC32I) PENTYLENE GLYCOL (UNII: 50C1307PZG) EDETATE DISODIUM (UNII: 7FLD91C86K) PHENOXYETHANOL (UNII: HIE492ZZ3T) CHLORPHENESIN (UNII: I670DAL4SZ) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) FD&C RED NO. 4 (UNII: X3W0AM1JLX) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) CITRIC ACID ACETATE (UNII: DSO12WL7AU) TOCOPHEROL (UNII: R0ZB2556P8) HYDROXYETHYL CELLULOSE (100 MPA.S AT 2%) (UNII: R33S7TK2EP) CALCIUM CHLORIDE (UNII: M4I0D6VV5M) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) PEG-8 LAURATE (UNII: 762O8IWA10) SODIUM POLYACRYLATE (2500000 MW) (UNII: 05I15JNI2J) CARBOMER INTERPOLYMER TYPE A (ALLYL SUCROSE CROSSLINKED) (UNII: 59TL3WG5CO) BUTYL ACRYLATE/METHYL METHACRYLATE/METHACRYLIC ACID COPOLYMER (18000 MW) (UNII: JZ1374NL9E) SIGESBECKIA ORIENTALIS FLOWERING TOP (UNII: 6UL878YAR7) WHEY (UNII: 8617Z5FMF6) SACCHARINA LATISSIMA (UNII: 68CMP2MB55) WATER (UNII: 059QF0KO0R) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49527-050-01 1 in 1 CARTON 12/01/2015 1 50 mL in 1 JAR; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 12/01/2015 Labeler - CLINIQUE LABORATORIES LLC (044475127) Registrant - Estee Lauder Companies Inc. (790802086) Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics Ltd. 202952982 manufacture(49527-050) Establishment Name Address ID/FEI Business Operations Estee Lauder Cosmetics Ltd. 204132062 pack(49527-050) , label(49527-050) Establishment Name Address ID/FEI Business Operations The Estee Lauder Inc 802599436 manufacture(49527-050) , pack(49527-050) , label(49527-050)