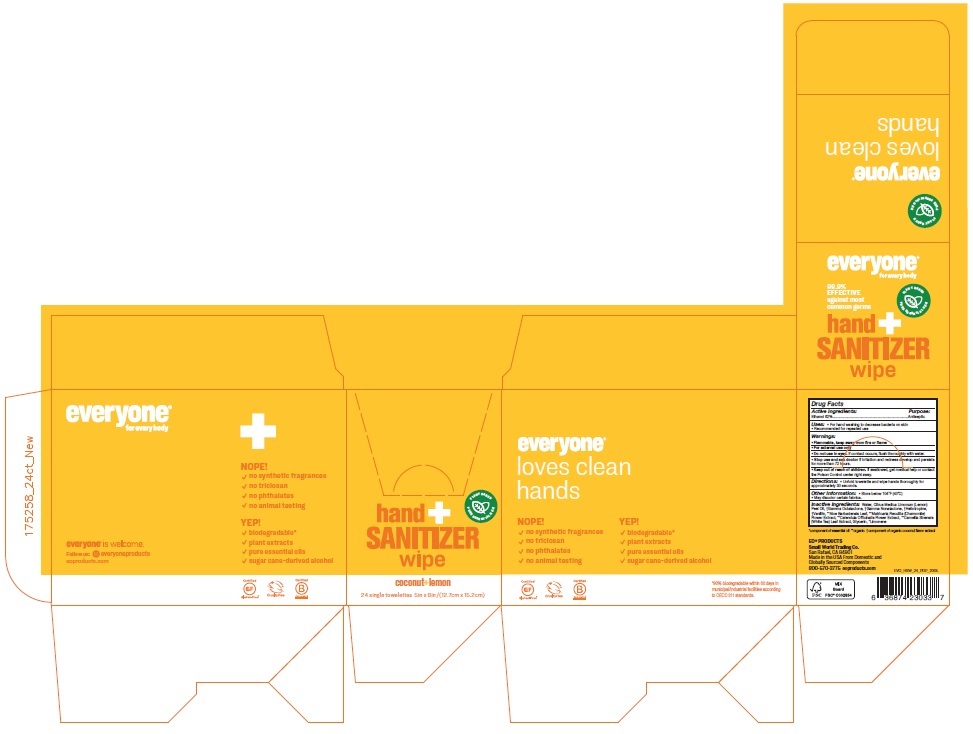
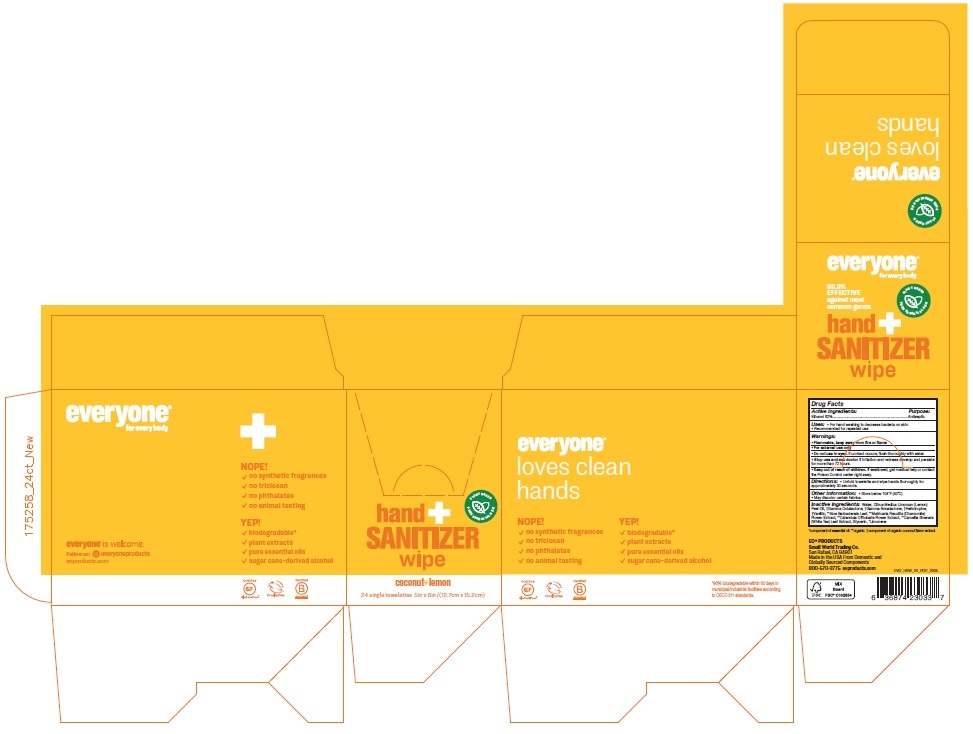
Label: EVERYONE HAND SANITIZER COCONUT LEMON- alcohol cloth
- NDC Code(s): 54748-506-15, 54748-506-24
- Packager: EO Products, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated February 22, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients:
- Purpose:
- Uses:
- Warnings:
- Directions:
- Other information:
-
Inactive ingredients:
Water, Citrus Medica Limonum (Lemon) Peel Oil, †Gamma Octalactone, †Gamma Nonalactone, †Heliotropine, †Vanillin, **Aloe Barbadensis Leaf, **Matricaria Recutita (Chamomile) Flower Extract, **Calendula Officinalis Flower Extract, **Camellia Sinensis (White Tea) Leaf Extract, Glycerin, *Limonene
*component of essential oil
**organic
†component of organic coconut flavor extract
-
SPL UNCLASSIFIED SECTION
for every body
PLANT BASED
PURE ESSENTIAL OILS
99.9% EFFECTIVE against most common germs
Biodegradable wipes gently clean and moisturize hands with aloe, calendula, and chamomile extracts.
coconut+ lemon
NOPE!
√ no synthetic fragrances
√ no triclosan
√ no phthalates
√ no animal testing
YEP!
√ biodegradable*
√ plant extracts
√ pure essential oils
√ sugar-cane-derived alcohol
Certified Gluten-Free®
Cruelty Free
Certified Corporation™
everyone® is welcome.
Follow us: everyoneproducts
eoproducts.com
*90% biodegradable within 60 days in municipal/industrial facilities according to OECD 311 standards.
EO® PRODUCTS
Small World Trading Co.
San Rafael, CA 94901
Made in the USA From Domestic and Globally Sourced Components
800-570-3775 eoproducts.com
- Packaging
-
INGREDIENTS AND APPEARANCE
EVERYONE HAND SANITIZER COCONUT LEMON
alcohol clothProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:54748-506 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 62 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) LEMON OIL (UNII: I9GRO824LL) .GAMMA.-OCTALACTONE (UNII: UHD6M52X0K) .GAMMA.-NONALACTONE (UNII: I1XGH66S8P) PIPERONAL (UNII: KE109YAK00) VANILLIN (UNII: CHI530446X) ALOE VERA LEAF (UNII: ZY81Z83H0X) CHAMOMILE (UNII: FGL3685T2X) CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) GREEN TEA LEAF (UNII: W2ZU1RY8B0) GLYCERIN (UNII: PDC6A3C0OX) LIMONENE, (+)- (UNII: GFD7C86Q1W) Product Characteristics Color Score Shape Size Flavor COCONUT, LEMON Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:54748-506-15 6 in 1 BOX 07/28/2020 1 15 in 1 POUCH 1 4.17 mL in 1 PACKAGE; Type 0: Not a Combination Product 2 NDC:54748-506-24 24 in 1 BOX 07/28/2020 2 1 in 1 POUCH 2 4.17 mL in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 07/28/2020 Labeler - EO Products, LLC (786611210) Establishment Name Address ID/FEI Business Operations EO Products, LLC 786611210 manufacture(54748-506)