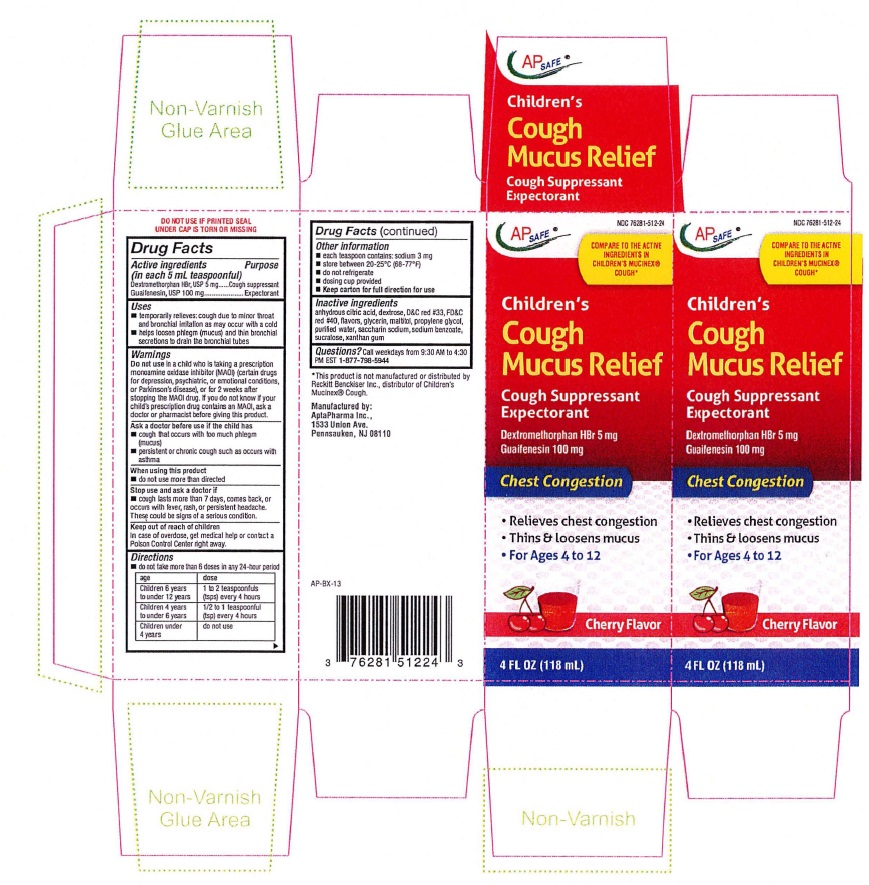
Label: CHILDRENS MUCUS RELIEF COUGH - CHERRY FLAVOR- dextromethorphan hydrobromide, guaifenesin liquid
- NDC Code(s): 76281-512-24
- Packager: AptaPharma Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated January 31, 2024
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Do not use in a child who is taking a prescription monoamine oxidase inhibitor (MAOI) (certain
drugs for depression, psychiatric, or emotional conditions, or Parkinson' s disease), or for 2
weeks after stopping the MAOI drug. If you do not know if your child's prescription drug
contains an MAOI, ask a doctor or pharmacist before giving this product.Ask a doctor before use if the child has
• cough that occurs with too much phlegm (mucus)
• persistent or chronic cough such as occurs with asthmaWhen using this product• do not use more than directed
Stop use and ask a doctor if
• cough lasts more than 7 days, comes back, or occurs with fever, rash, or persistent headache.
These could be signs of a serious condition. - KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
-
CHILDREN'S MUCUS RELIEF COUGH CHERRY
AP SAFE NDC 76281-512-24
Children's
Cough
Mucus ReliefCough Suppressant
ExpectorantCherry Flavor
4 FL OZ (118 mL)DO NOT USE IF PRINTED SEAL UNDER CAP IS TORN OR MISSING
*This product is not manufactured or distributed by
Reckitt Benckiser Inc., distributor of Children's
Mucinex®Cough.Manufactured by:
AptaPharma Inc.,
1533 Union Ave.
Pennsauken, NJ 08110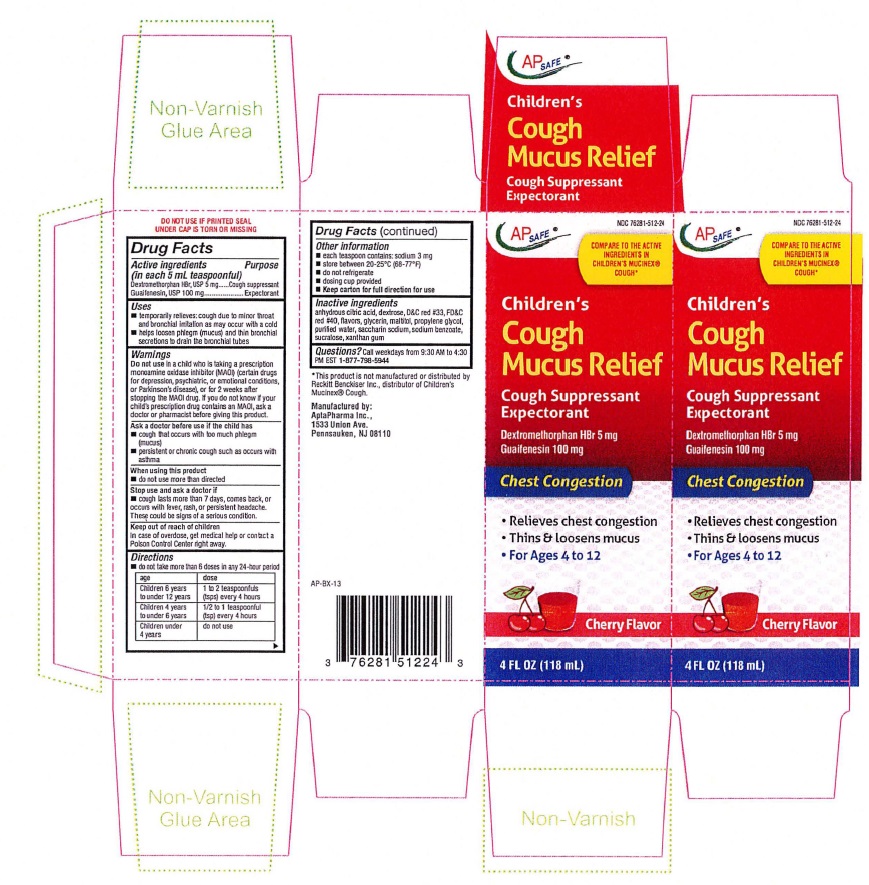

res
-
INGREDIENTS AND APPEARANCE
CHILDRENS MUCUS RELIEF COUGH - CHERRY FLAVOR
dextromethorphan hydrobromide, guaifenesin liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:76281-512 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 5 mg in 5 mL GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 100 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) DEXTROSE (UNII: IY9XDZ35W2) D&C RED NO. 33 (UNII: 9DBA0SBB0L) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) MALTITOL (UNII: D65DG142WK) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SODIUM BENZOATE (UNII: OJ245FE5EU) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color red Score Shape Size Flavor CHERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76281-512-24 1 in 1 CARTON 01/30/2024 1 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 01/30/2024 Labeler - AptaPharma Inc. (790523323) Registrant - AptaPharma Inc. (790523323) Establishment Name Address ID/FEI Business Operations AptaPharma Inc. 790523323 manufacture(76281-512)