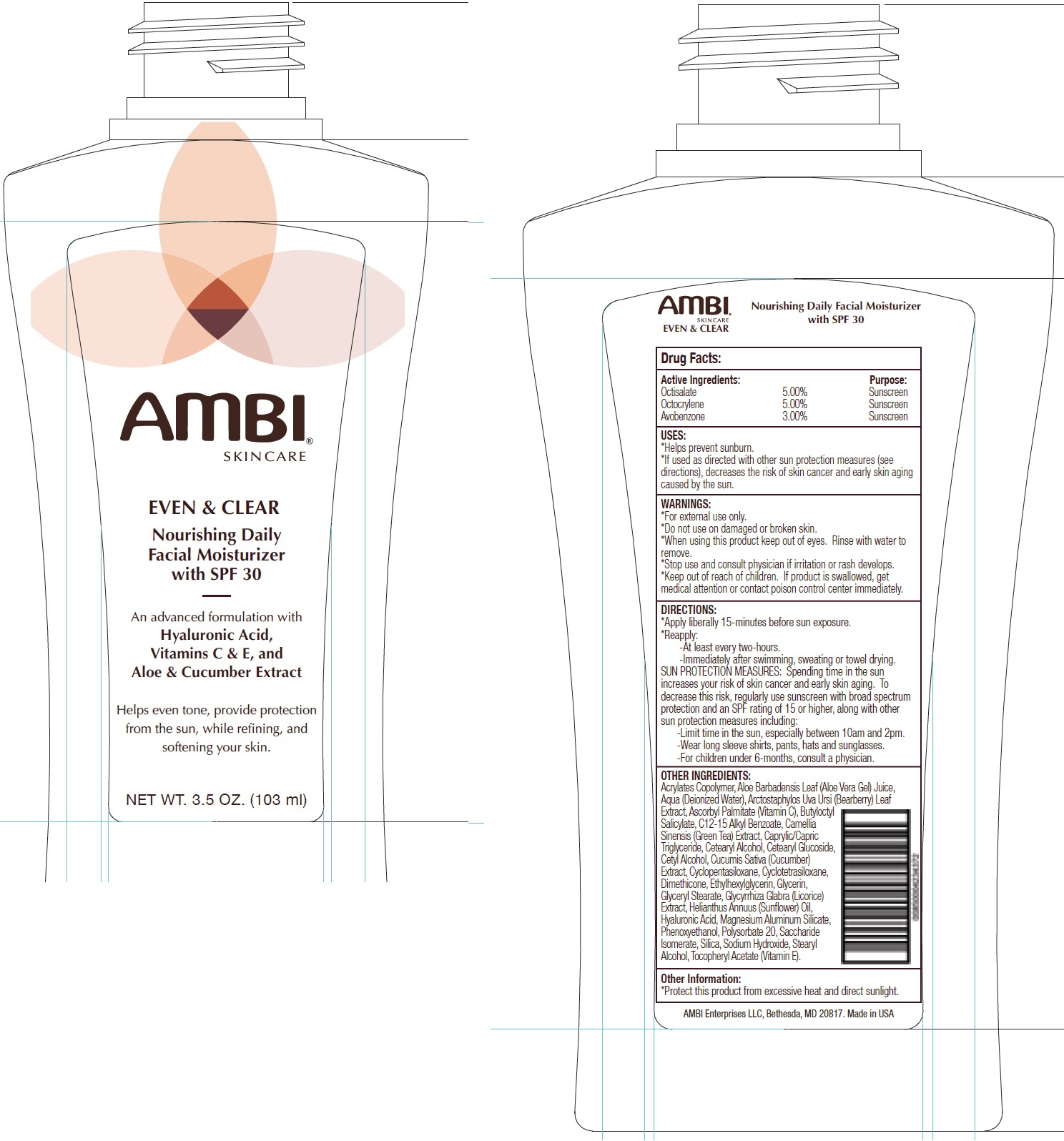
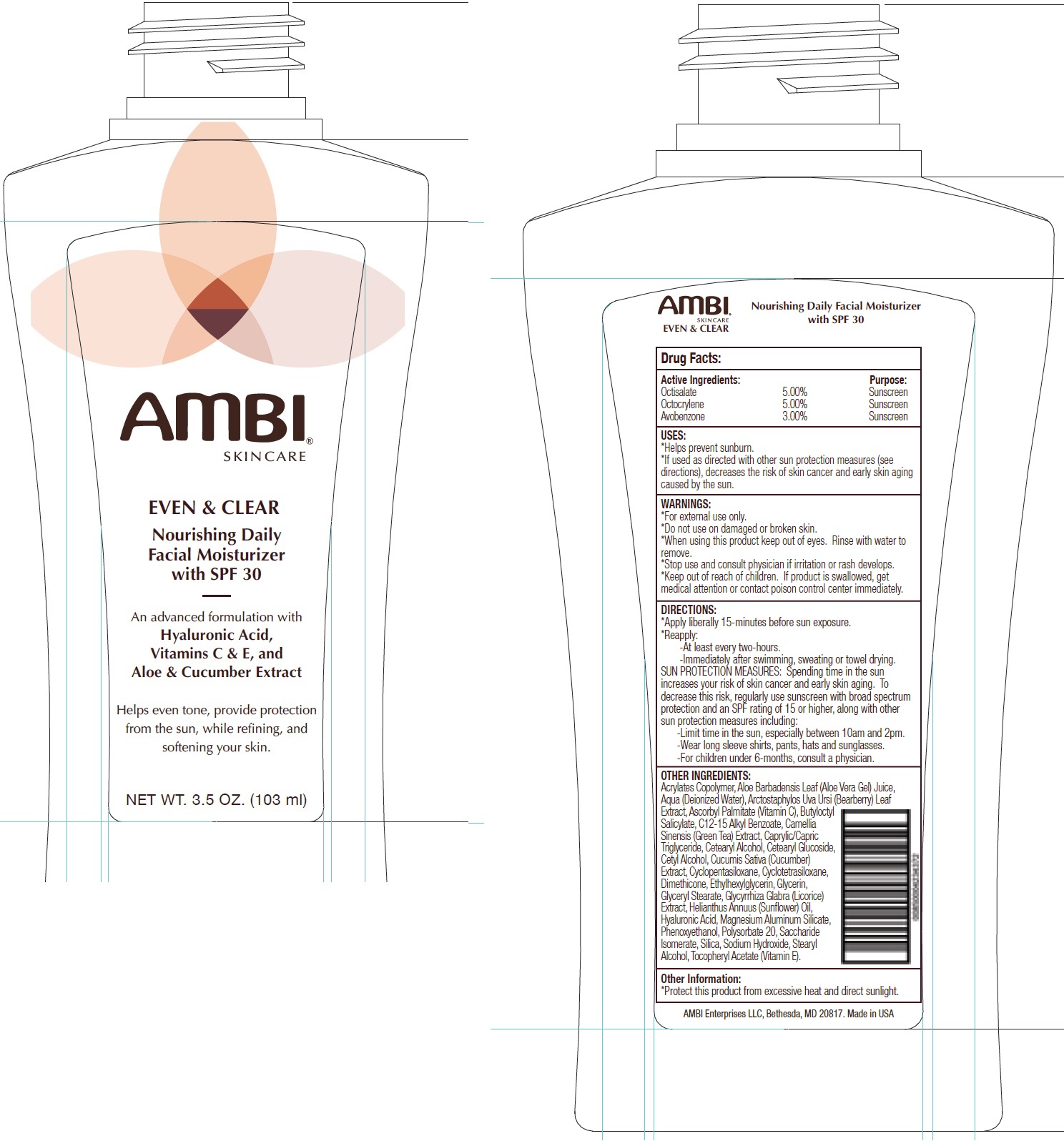
Label: AMBI SKINCARE EVEN AND CLEAR DAILY FACIAL MOISTURIZER SPF 30- octisalate, octocrylene, avobenzone cream
- NDC Code(s): 73453-073-03
- Packager: AMBI Enterprises LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated June 19, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts:
- Active Ingredients:
- USES:
- WARNINGS:
-
DIRECTIONS:
- Apply liberally 15-minutes before sun exposure.
- Reapply:
-At least every two-hours. -Immediately after swimming, sweating or towel drying. SUN PROTECTION MEASURES: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use sunscreen with broad spectrum protection and an SPF rating of 15 or higher, along with other sun protection measures including: -Limit time in the sun, especially between 10am and 2pm. -Wear long sleeve shirts, pants, hats and sunglasses. -For children under 6-months, consult a physician.
-
OTHER INGREDIENTS:
Acrylates Copolymer, Aloe Barbadensis Leaf (Aloe Vera Gel) Juice, Aqua (Deionized Water), Arctostaphylos Uva Ursi (Bearberry) Leaf Extract, Ascorbyl Palmitate (Vitamin C), Butyloctyl Salicylate, C12-15 Alkyl Benzoate, Camellia Sinensis (Green Tea) Extract, Caprylic/Capric Triglyceride, Cetearyl Alcohol, Cetearyl Glucoside, Cetyl Alcohol, Cucumis Sativa (Cucumber) Extract, Cyclopentasiloxane, Cyclotetrasiloxane, Dimethicone, Ethylhexylglycerin, Glycerin, Glyceryl Stearate, Glycyrrhiza Glabra (Licorice) Extract, Helianthus Annuus (Sunflower) Oil, Hyaluronic Acid, Magnesium Aluminum Silicate, Phenoxyethanol, Polysorbate 20, Saccharide Isomerate, Silica, Sodium Hydroxide, Stearyl Alcohol, Tocopheryl Acetate (Vitamin E).
- Other Information:
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
AMBI SKINCARE EVEN AND CLEAR DAILY FACIAL MOISTURIZER SPF 30
octisalate, octocrylene, avobenzone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73453-073 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 50 mg in 1 mL AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL Inactive Ingredients Ingredient Name Strength BUTYL ACRYLATE/METHYL METHACRYLATE/METHACRYLIC ACID COPOLYMER (18000 MW) (UNII: JZ1374NL9E) ALOE VERA LEAF (UNII: ZY81Z83H0X) WATER (UNII: 059QF0KO0R) ARCTOSTAPHYLOS UVA-URSI LEAF (UNII: 3M5V3D1X36) ASCORBYL PALMITATE (UNII: QN83US2B0N) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) GREEN TEA LEAF (UNII: W2ZU1RY8B0) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) CETEARYL GLUCOSIDE (UNII: 09FUA47KNA) CETYL ALCOHOL (UNII: 936JST6JCN) CUCUMBER (UNII: YY7C30VXJT) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) CYCLOMETHICONE 4 (UNII: CZ227117JE) DIMETHICONE (UNII: 92RU3N3Y1O) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) GLYCYRRHIZA GLABRA (UNII: 2788Z9758H) HELIANTHUS ANNUUS FLOWERING TOP (UNII: BKJ0J3D1BP) HYALURONIC ACID (UNII: S270N0TRQY) MAGNESIUM ALUMINUM SILICATE (UNII: 6M3P64V0NC) PHENOXYETHANOL (UNII: HIE492ZZ3T) POLYSORBATE 20 (UNII: 7T1F30V5YH) SACCHARIDE ISOMERATE (UNII: W8K377W98I) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM HYDROXIDE (UNII: 55X04QC32I) STEARYL ALCOHOL (UNII: 2KR89I4H1Y) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73453-073-03 103 mL in 1 BOTTLE; Type 0: Not a Combination Product 11/01/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/01/2019 Labeler - AMBI Enterprises LLC (117015229)