Label: SODIUM TETRADECYL SULFATE injection, solution
- NDC Code(s): 24201-201-01, 24201-201-05
- Packager: Hikma Pharmaceuticals USA Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated June 26, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
DESCRIPTION
Sodium tetradecyl sulfate is an anionic surfactant which occurs as a white, waxy solid. The structural formula is as follows:
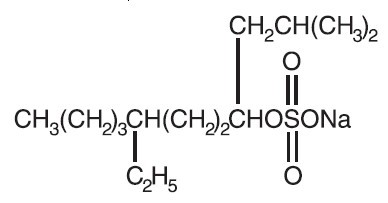
C 14H 29NaSO 4 7-Ethyl-2-methyl-4-hendecanol sulfate sodium salt MW 316.44
Sodium tetradecyl sulfate injection is a sterile nonpyrogenic solution for intravenous use as a sclerosing agent.
3% 60 mg/2 mL (30 mg/mL): Each mL contains sodium tetradecyl sulfate 30 mg, benzyl alcohol 0.02 mL and dibasic sodium phosphate, anhydrous 9 mg in Water for Injection. pH 7.9; monobasic sodium phosphate and/or sodium hydroxide added, if needed, for pH adjustment.
- CLINICAL PHARMACOLOGY
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Sodium tetradecyl sulfate injection is contraindicated in previous hypersensitivity reactions to the drug; in acute superficial thrombophlebitis; valvular or deep vein incompetence; huge superficial veins with wide open communications to deeper veins; phlebitis migrans; acute cellulitis; allergic conditions; acute infections; varicosities caused by abdominal and pelvic tumors unless the tumor has been removed; bedridden patients; such uncontrolled systemic diseases as diabetes, toxic hyperthyroidism, tuberculosis, asthma, neoplasm, sepsis, blood dyscrasias and acute respiratory or skin diseases.
-
WARNINGS
Sodium tetradecyl sulfate injection should only be administered by a healthcare professional experienced in venous anatomy and the diagnosis and treatment of conditions affecting the venous system and familiar with proper injection technique. Severe adverse local effects, including tissue necrosis, may occur following extravasation; therefore, extreme care in intravenous needle placement and using the minimal effective volume at each injection site are important.
Emergency resuscitation equipment should be immediately available. Allergic reactions, including fatal anaphylaxis, have been reported. As a precaution against anaphylactic shock, it is recommended that 0.5 mL of sodium tetradecyl sulfate injection be injected into a varicosity, followed by observation of the patient for several hours before administration of a second or larger dose. The possibility of an anaphylactic reaction should be kept in mind, and the physician should be prepared to treat it appropriately.
Because of the danger of thrombosis extension into the deep venous system, thorough preinjection evaluation for valvular competency should be carried out and slow injections with a small amount (not over 2 mL) of the preparation should be injected into the varicosity. Deep venous patency must be determined by noninvasive testing such as duplex ultrasound. Venous sclerotherapy should not be undertaken if tests such as Trendelenberg and Perthes, and angiography show significant valvular or deep venous incompetence.
The development of deep vein thrombosis and pulmonary embolism have been reported following sclerotherapy treatment of superficial varicosities. Patients should have post-treatment follow-up of sufficient duration to assess for the development of deep vein thrombosis. Embolism may occur as long as four weeks after injection of sodium tetradecyl sulfate. Adequate post-treatment compression may decrease the incidence of deep vein thrombosis.
Arterial Embolism
Stroke, transient ischemic attack, myocardial infarction, and impaired cardiac function have been reported in close temporal relationship with STS administration. These events may be caused by air embolism when using the product foamed with room air (high nitrogen concentration) or thromboembolism. The safety and efficacy of STS foamed with room air has not been established and its use should be avoided.
- PRECAUTIONS
-
DRUG INTERACTIONS
No well-controlled studies have been performed on patients taking antiovulatory agents. The physician must use judgment and evaluate any patient taking antiovulatory drugs prior to initiating treatment with sodium tetradecyl sulfate injection. (See ADVERSE REACTIONS section.)
Heparin should not be included in the same syringe as sodium tetradecyl sulfate injection, since the two are incompatible.
-
CARCINOGENESIS, MUTAGENESIS, IMPAIRMENT OF FERTILITY
When tested in the L5178YTK +/- mouse lymphoma assay, sodium tetradecyl sulfate did not induce a dose-related increase in the frequency of thymidine kinase-deficient mutants and, therefore, was judged to be nonmutagenic in this system. However, no long-term animal carcinogenicity studies with sodium tetradecyl sulfate have been performed.
-
PREGNANCY
Teratogenic Effects
Animal reproduction studies have not been conducted with sodium tetradecyl sulfate injection. It is also not known whether sodium tetradecyl sulfate injection can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. sodium tetradecyl sulfate injection should be given to a pregnant woman only if clearly needed and the benefits outweigh the risks.
- NURSING MOTHERS
- PEDIATRIC USE
-
ADVERSE REACTIONS
Local reactions consisting of pain, urticaria or ulceration may occur at the site of injection. A permanent discoloration may remain along the path of the sclerosed vein segment. Sloughing and necrosis of
tissue may occur following extravasation of the drug. (See WARNINGS section.)
Allergic reactions such as hives, asthma, hay fever and anaphylactic shock have been reported. Mild systemic reactions that have been reported include headache, nausea and vomiting. (See WARNINGS section.)
At least six deaths have been reported with the use of sodium tetradecyl sulfate injection. Four cases of anaphylactic shock leading to death have been reported in patients who received sodium tetradecyl sulfate injection. One of these four patients reported a history of asthma, a contraindication to the administration of sodium tetradecyl sulfate injection. (See WARNINGS section.)
One death has been reported in a patient who received sodium tetradecyl sulfate injection and who had been receiving an antiovulatory agent. Another death (fatal pulmonary embolism) has been reported in a 36-year-old female treated with sodium tetradecyl acetate and who was not taking oral contraceptives.
Cerebrovascular accident, myocardial infarction
To report SUSPECTED ADVERSE REACTIONS, contact Hikma Pharmaceuticals USA Inc. at 1-877-845-0689 or FDA at 1-800 FDA-1088 or www.fda.gov/medwatch.
-
DOSAGE AND ADMINISTRATION
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. Do not use if precipitated or discolored.
Sodium tetradecyl sulfate injection is for intravenous use only. The strength of solution required depends on the size and degree of varicosity. In general, the 1% solution will be found most useful with the 3% solution preferred for larger varicosities. The dosage should be kept small, using 0.5 mL to 2 mL (preferably 1 mL maximum) for each injection, and the maximum single treatment should not exceed 10 mL.
- HOW SUPPLIED
- STORAGE
-
ANIMAL TOXICOLOGY
The intravenous LD 50 of sodium tetradecyl sulfate in mice was reported to be 90 ± 5 mg/kg.
In the rat, the acute intravenous LD 50 of sodium tetradecyl sulfate was estimated to be between 72 mg/kg and 108 mg/kg.
Purified sodium tetradecyl sulfate was found to have an LD 50 of 2 g/kg when administered orally by stomach tube as a 25% aqueous solution to rats. In rats given 0.15 g/kg in drinking water for 30 days, no appreciable toxicity was seen, although some growth inhibition was discernible.
Manufactured for:
Hikma Pharmaceuticals USA Inc.
Berkeley Heights, NJ 07922 USAManufactured by:
Hikma Farmacêutica (Portugal), S.A.
Estrada do Rio da Mó, 8, 8A e 8B - Fervença
2705-906 Terrugem SNT, PORTUGALREVISED December 2023
PIN657-WES/1 -
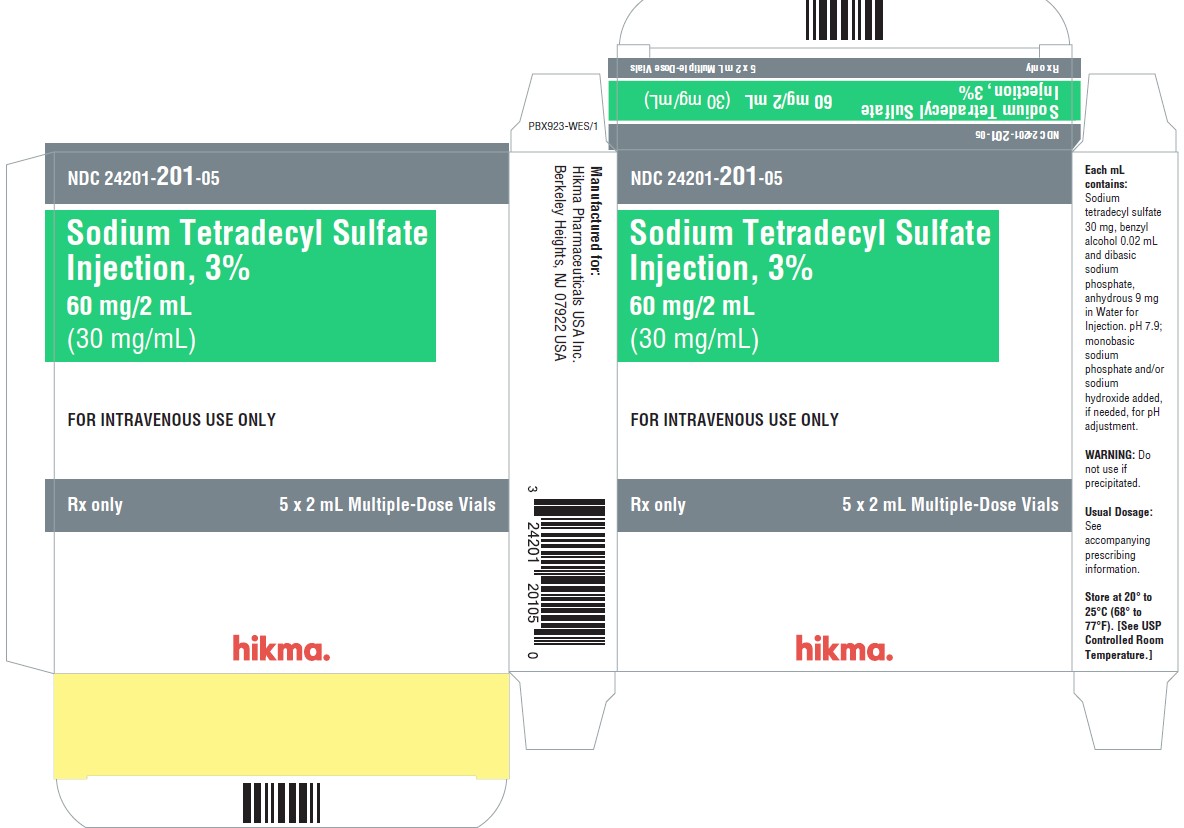
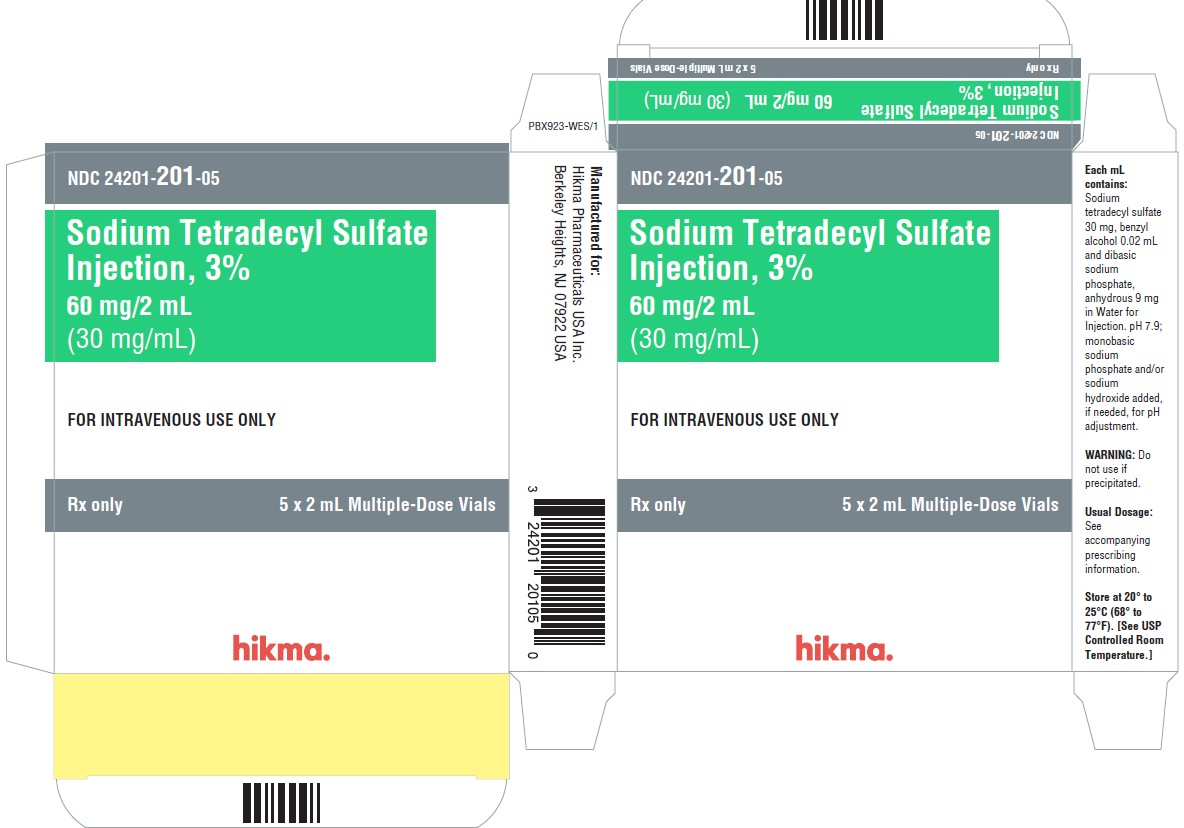
PRINCIPAL DISPLAY PANEL - 30 mg/mL Carton Label
NDC 24201- 201-01
2 mL Multiple-Dose Vial
Sodium Tetradecyl Sulfate 3%
Injection60 mg/2 mL
(30 mg/mL)FOR IV USE ONLY
DO NOT USE IF
PRECIPITATEDRx Only

NDC 24201- 201-05
5 x 2 mL Multiple-Dose Vial
Sodium Tetradecyl Sulfate 3%
Injection60 mg/2 mL
(30 mg/mL)FOR IV USE ONLY
DO NOT USE IF
PRECIPITATEDRx Only
-
INGREDIENTS AND APPEARANCE
SODIUM TETRADECYL SULFATE
sodium tetradecyl sulfate injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24201-201 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM TETRADECYL SULFATE (UNII: Q1SUG5KBD6) (TETRADECYL HYDROGEN SULFATE (ESTER) - UNII:6326W0DRHY) SODIUM TETRADECYL SULFATE 30 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) 0.02 mL in 1 mL SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) 9 mg in 1 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24201-201-05 5 in 1 CARTON 12/09/2019 1 NDC:24201-201-01 2 mL in 1 VIAL, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA209937 12/09/2019 Labeler - Hikma Pharmaceuticals USA Inc. (118707839) Registrant - HIKMA FARMACEUTICA (PORTUGAL), S.A. (452742943)