Label: DR. AVI FOR HIM- zinc, tongkat ali, oyster meat ,l-arginine hcl, horny goat weed icariins, tribulus extract saponins, butea superba extract ,ginkgo biloba extract ,pine bark extract capsule
- NHRIC Code(s): 13411-871-30
- Packager: Advanced Pharmaceutical Services, Inc. Dba Affordable Quality Pharmaceuticals
- Category: DIETARY SUPPLEMENT
Drug Label Information
Updated April 6, 2021
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Identity
- Warning
- Safe handling and warning
-
Health claim
Dr. Avi’s Formula for HIM contains Tongkat Ali (Eurycoma Longifolia) and other natural extracts that help promote the production of Luteinizing hormone which naturally stimulates the body to synthesize Testosterone rapidly and constantly.*
For some men, male menopause (known as Andropause) can be physically and emotionally straining. Male Andropause occurs when testosterone levels become depleted, resulting in mood changes, loss of energy, low sex drive and reduction of physical agility.*
Dr. Avi’s Formula for HIM helps increase vitality and improve sexual functions by stimulating the producing of testosterone in the body.** These statements have not been evaluated by the Food & Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.
- KEEP OUT OF REACH OF CHILDREN.
- Direction
- Product label
-
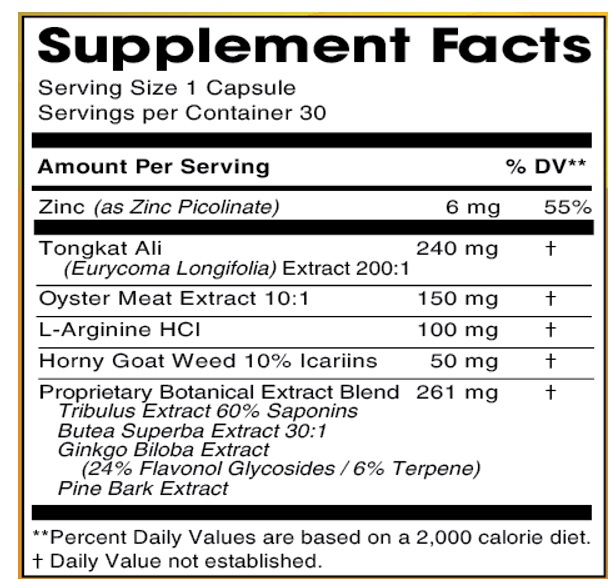
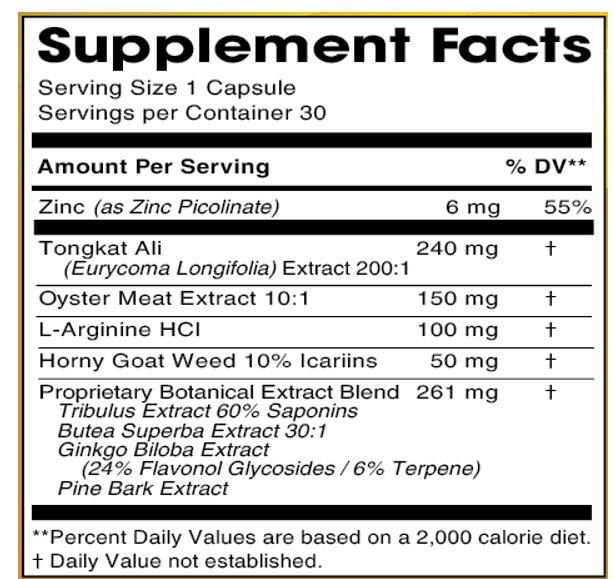
INGREDIENTS AND APPEARANCE
DR. AVI FOR HIM
zinc, tongkat ali, oyster meat ,l-arginine hcl, horny goat weed icariins, tribulus extract saponins, butea superba extract ,ginkgo biloba extract ,pine bark extract capsuleProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:13411-871 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC PICOLINATE (UNII: ALO92O31SE) (ZINC CATION - UNII:13S1S8SF37) ZINC PICOLINATE 6 mg EURYCOMA LONGIFOLIA ROOT (UNII: 43B8E2HG8B) (EURYCOMA LONGIFOLIA ROOT - UNII:43B8E2HG8B) EURYCOMA LONGIFOLIA ROOT 240 mg ARGININE HYDROCHLORIDE (UNII: F7LTH1E20Y) (ARGININE - UNII:94ZLA3W45F) ARGININE HYDROCHLORIDE 100 mg EPIMEDIUM GRANDIFLORUM TOP (UNII: 137PC46F89) (EPIMEDIUM GRANDIFLORUM TOP - UNII:137PC46F89) EPIMEDIUM GRANDIFLORUM TOP 50 mg TRIBULUS TERRESTRIS FRUIT (UNII: QNL076V6EQ) (TRIBULUS TERRESTRIS FRUIT - UNII:QNL076V6EQ) TRIBULUS TERRESTRIS FRUIT 100 mg GINKGO (UNII: 19FUJ2C58T) (GINKGO - UNII:19FUJ2C58T) GINKGO 40 mg MARITIME PINE (UNII: 50JZ5Z98QY) (MARITIME PINE - UNII:50JZ5Z98QY) MARITIME PINE 40 mg BUTEA SUPERBA ROOT (UNII: A9619D9HWY) (BUTEA SUPERBA ROOT - UNII:A9619D9HWY) BUTEA SUPERBA ROOT 81 mg EDIBLE OYSTER (UNII: 7776U7P31L) (EDIBLE OYSTER - UNII:7776U7P31L) EDIBLE OYSTER 150 mg Inactive Ingredients Ingredient Name Strength GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) MAGNESIUM STEARATE (UNII: 70097M6I30) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:13411-871-30 30 in 1 BOTTLE 1 1 in 1 CAPSULE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date dietary supplement 04/06/2021 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color shape size (solid drugs) 23 mm scoring 1 Labeler - Advanced Pharmaceutical Services, Inc. Dba Affordable Quality Pharmaceuticals (187498279)