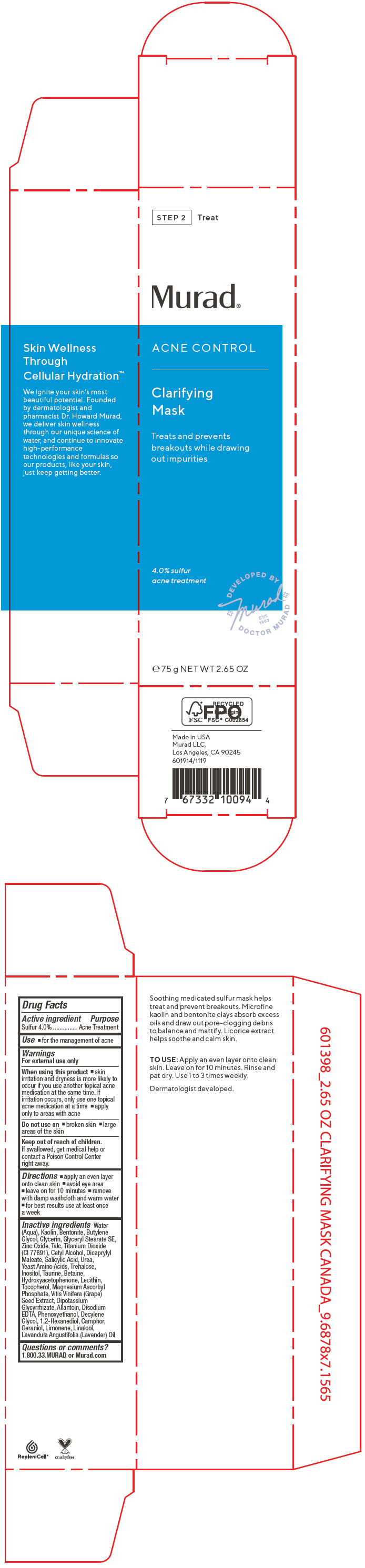
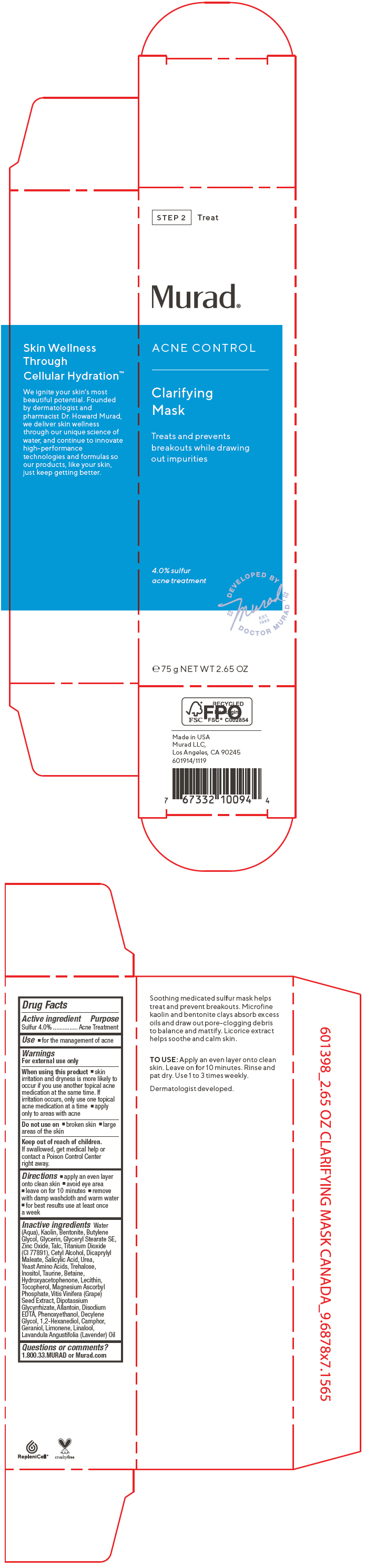
Label: ACNE CONTROL CLARIFYING MASK- sulfur cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 70381-115-01, 70381-115-02 - Packager: Murad, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 2, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
- Warnings
- Directions
-
Inactive ingredients
Water (Aqua), Kaolin, Bentonite, Butylene Glycol, Glycerin, Glyceryl Stearate SE, Zinc Oxide, Talc, Titanium Dioxide (CI 77891), Cetyl Alcohol, Dicaprylyl Maleate, Salicylic Acid, Urea, Yeast Amino Acids, Trehalose, Inositol, Taurine, Betaine, Hydroxyacetophenone, Lecithin, Tocopherol, Magnesium Ascorbyl Phosphate, Vitis Vinifera (Grape) Seed Extract, Dipotassium Glycyrrhizate, Allantoin, Disodium EDTA, Phenoxyethanol, Decylene Glycol, 1,2-Hexanediol, Camphor, Geraniol, Limonene, Linalool, Lavandula Angustifolia (Lavender) Oil
- Questions or comments?
- PRINCIPAL DISPLAY PANEL - 75 g Tube Carton
-
INGREDIENTS AND APPEARANCE
ACNE CONTROL CLARIFYING MASK
sulfur creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70381-115 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SULFUR (UNII: 70FD1KFU70) (SULFUR - UNII:70FD1KFU70) SULFUR 4 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) KAOLIN (UNII: 24H4NWX5CO) BENTONITE (UNII: A3N5ZCN45C) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) GLYCERIN (UNII: PDC6A3C0OX) GLYCERYL STEARATE SE (UNII: FCZ5MH785I) ZINC OXIDE (UNII: SOI2LOH54Z) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) CETYL ALCOHOL (UNII: 936JST6JCN) DIOCTYL MALEATE (UNII: OD88G8439L) SALICYLIC ACID (UNII: O414PZ4LPZ) UREA (UNII: 8W8T17847W) AMINO ACIDS, SOURCE UNSPECIFIED (UNII: 0O72R8RF8A) TREHALOSE (UNII: B8WCK70T7I) INOSITOL (UNII: 4L6452S749) TAURINE (UNII: 1EQV5MLY3D) BETAINE (UNII: 3SCV180C9W) HYDROXYACETOPHENONE (UNII: G1L3HT4CMH) EGG PHOSPHOLIPIDS (UNII: 1Z74184RGV) TOCOPHEROL (UNII: R0ZB2556P8) MAGNESIUM ASCORBYL PHOSPHATE (UNII: 0R822556M5) VITIS VINIFERA SEED (UNII: C34U15ICXA) GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) ALLANTOIN (UNII: 344S277G0Z) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) PHENOXYETHANOL (UNII: HIE492ZZ3T) DECYLENE GLYCOL (UNII: S57M60MI88) 1,2-HEXANEDIOL (UNII: TR046Y3K1G) CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) GERANIOL (UNII: L837108USY) LIMONENE, (+)- (UNII: GFD7C86Q1W) LINALOOL, (+/-)- (UNII: D81QY6I88E) LAVENDER OIL (UNII: ZBP1YXW0H8) Product Characteristics Color WHITE (Off-White to Beige) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70381-115-02 1 in 1 CARTON 07/28/2021 1 NDC:70381-115-01 75 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH FINAL part333D 07/28/2021 Labeler - Murad, LLC (781254792) Establishment Name Address ID/FEI Business Operations Cosway Company, Inc. 052400223 MANUFACTURE(70381-115)