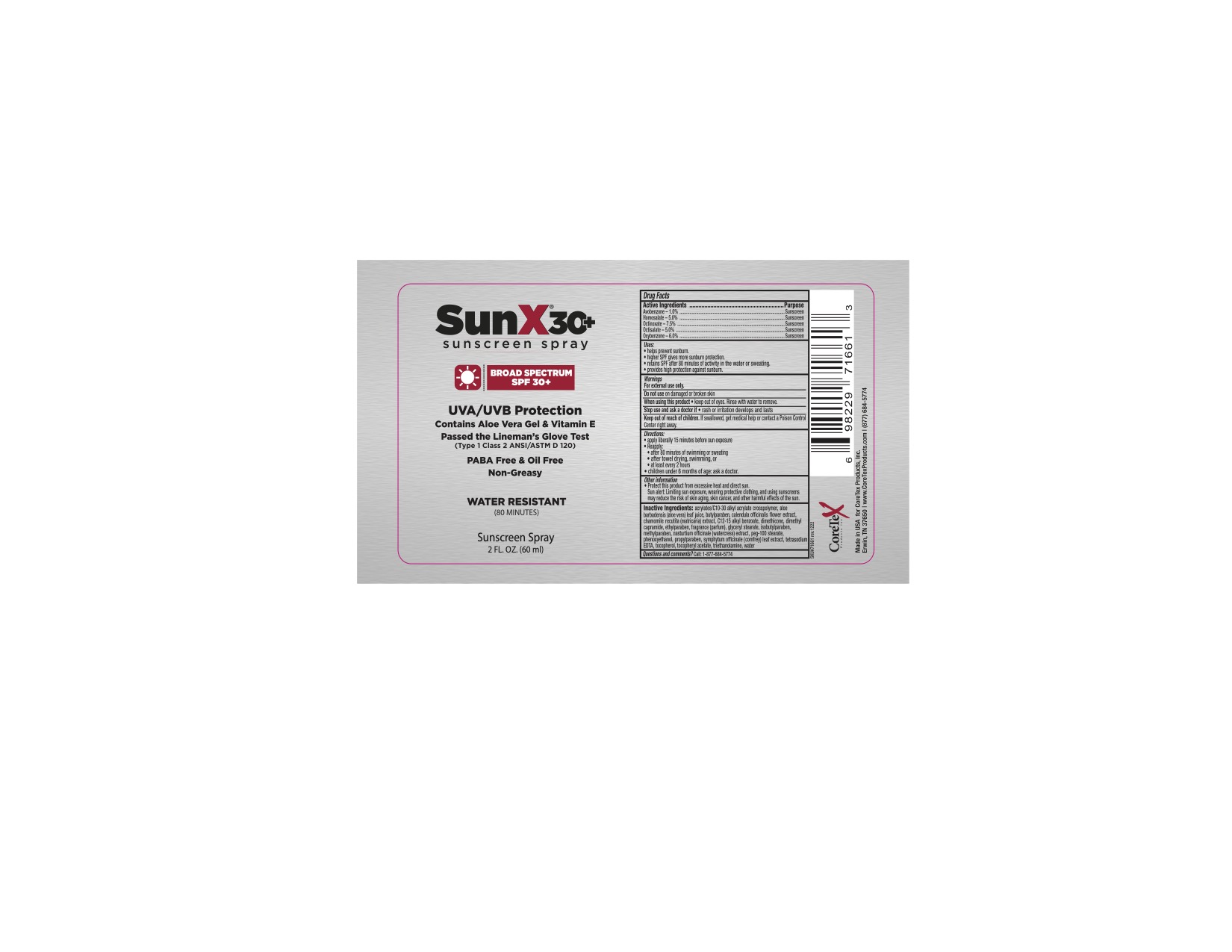
Label: CORETEX SUN X SPF 30 THIN- avobenzone, homosalate, octinoxate, octisalate, oxybenzone liquid
-
NDC Code(s):
65753-101-11,
65753-101-12,
65753-101-22,
65753-101-23, view more65753-101-24, 65753-101-25, 65753-101-26, 65753-101-27
- Packager: CoreTex Products Inc
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 22, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
- Warnings
- Directions:
- Other information
-
Inactive ingredients:
acrylates/C10-30 alkyl acrylate crosspolymer, aloe barbadensis (aloe vera) leaf juice, butylparaben, calendula officinalis flower extract, carbomer, chamomile recutita (matricaria) extract, C12-15 alkyl benzoate, dimethicone, dimethyl capramide, ethylparaben, fragrance (parfum), glyceryl stearate, isobutylparaben, methylparaben, nasturtium officinale (watercress) extract, peg-100 stearate, phenoxyethanol, propylparaben, symphytum officinale (comfrey) leaf extract, tetrasodium EDTA, tocopherol, tocopheryl acetate, triethanolamine, water
- Questions and comments?
- Principal Display Panel 101-12
- 101-11
-
INGREDIENTS AND APPEARANCE
CORETEX SUN X SPF 30 THIN
avobenzone, homosalate, octinoxate, octisalate, oxybenzone liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65753-101 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 1 g in 100 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 5 g in 100 mL OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 g in 100 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 5 g in 100 mL OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 6 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) DIMETHYL CAPRAMIDE (UNII: O29Y6X2JEZ) DIMETHICONE (UNII: 92RU3N3Y1O) ALOE VERA LEAF (UNII: ZY81Z83H0X) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) TROLAMINE (UNII: 9O3K93S3TK) PROPYLPARABEN (UNII: Z8IX2SC1OH) PHENOXYETHANOL (UNII: HIE492ZZ3T) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) METHYLPARABEN (UNII: A2I8C7HI9T) ETHYLPARABEN (UNII: 14255EXE39) BUTYLPARABEN (UNII: 3QPI1U3FV8) ISOBUTYLPARABEN (UNII: 0QQJ25X58G) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) CHAMOMILE (UNII: FGL3685T2X) CALENDULA OFFICINALIS FLOWER (UNII: P0M7O4Y7YD) PEG-100 STEARATE (UNII: YD01N1999R) NASTURTIUM OFFICINALE (UNII: YH89GMV676) COMFREY LEAF (UNII: DG4F8T839X) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) TETRASODIUM EDETATE DIHYDRATE (UNII: 3JGX4KKZ4A) FRAGRANCE 13576 (UNII: 5EM498GW35) ACRYLATES CROSSPOLYMER-6 (UNII: 4GXD0Q3OS3) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65753-101-11 59 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 11/22/2019 2 NDC:65753-101-12 118 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 11/22/2019 3 NDC:65753-101-27 16 in 1 BOX 11/22/2018 11/21/2022 3 7 mL in 1 PACKET; Type 0: Not a Combination Product 4 NDC:65753-101-22 25 in 1 CONTAINER 11/22/2019 11/21/2022 4 7 mL in 1 PACKET; Type 0: Not a Combination Product 5 NDC:65753-101-23 50 in 1 CONTAINER 11/22/2019 11/21/2022 5 7 mL in 1 PACKET; Type 0: Not a Combination Product 6 NDC:65753-101-24 50 in 1 CARTON 11/22/2019 11/21/2022 6 7 mL in 1 PACKET; Type 0: Not a Combination Product 7 NDC:65753-101-25 100 in 1 CARTON 11/22/2019 11/21/2022 7 7 mL in 1 PACKET; Type 0: Not a Combination Product 8 NDC:65753-101-26 300 in 1 BOX 11/22/2019 11/21/2022 8 7 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 11/22/2018 Labeler - CoreTex Products Inc (061944620) Establishment Name Address ID/FEI Business Operations CoreTex Products Inc 061944620 pack(65753-101) Establishment Name Address ID/FEI Business Operations Pure Source 080354456 manufacture(65753-101)